
Union Health Minister Shri J P Nadda emphasized the need for India to elevate its regulatory framework for drugs, cosmetics, and medical devices to global standards, aligning with the country's reputation as the 'Pharmacy of the World'. He made these remarks during a comprehensive review meeting held today to assess the regulation practices within the Central Drugs Standard Control Organization (CDSCO) and the Union Health Ministry.
Accompanied by Union Health Secretary Shri Apurva Chandra, Drugs Controller General of India (DCGI) Dr Rajeev Singh Raghuvanshi, and senior officials from CDSCO, Minister Nadda underscored India's global position as a leading producer and exporter of pharmaceuticals. He stressed the necessity for CDSCO to devise a roadmap with clear timelines to achieve international benchmarks in its regulatory mandates.
"In order to maintain our status as the global leader in drugs regulation, our regulatory framework must match our scale of operations and international expectations," stated Minister Nadda. He highlighted the importance of system-based upscaling, emphasizing uniformity, technical advancement, and a forward-thinking approach in regulatory practices.
Minister Nadda also emphasized the critical role of transparency in CDSCO's operations and interactions with the pharmaceutical and medical devices industry. "Transparency in procedures within CDSCO and the industry is crucial for achieving global standards," he affirmed. He called for adherence to principles that ensure products manufactured and exported from India meet the highest global quality standards.
Furthermore, Minister Nadda emphasized the necessity for continuous dialogue between CDSCO and industry stakeholders to address challenges and support compliance with regulatory standards. "We must focus on developing mechanisms that facilitate ease of doing business for the drugs industry while maintaining regulatory rigor," he added.
Concluding the meeting, Minister Nadda reiterated the importance of CDSCO evolving into a user-friendly organization equipped with state-of-the-art facilities comparable to global standards. He emphasized the need for CDSCO to proactively engage with industry partners to understand their concerns and collaborate towards achieving excellence in regulatory practices.
The review meeting chaired by Minister J P Nadda signifies India's commitment to enhancing its regulatory framework in the pharmaceutical sector, aiming to uphold its global reputation as a reliable provider of high-quality drugs and medical products.
Delhi-Meerut Rapid Rail: 3 Crore Passengers Rides in Record Time
May 6, 2026, 5:18 p.m.Delhi Plans Cloth Banks at Metro Stations to Promote Recycling of Used Garments
May 4, 2026, 4:47 p.m.97 Km, 65 Stations: Delhi Govt Prepares Major Metro Expansion Plan for Outer Regions
May 4, 2026, 4:27 p.m.Delhi Bans Advance Fee Collection in Private Schools, Mandates Monthly Payments
May 4, 2026, 4:12 p.m.Gurgaon to see relief from heat as pre-monsoon showers expected from May 2
May 2, 2026, 3:55 p.m.Delhi to launch mobile health vans and mass wedding schemes for construction workers
May 2, 2026, 11:18 a.m.Mahavir Jayanti Observed Nationwide with Devotion; Leaders Highlight Ahimsa and Compassion
March 31, 2026, 3:11 p.m.India-Albania Ties Stronger Than Ever, Says Albania’s Honorary Consul General to India
March 18, 2025, 11:23 p.m.India-Russia Relations Strengthened, PM Modi to Visit Moscow for Annual Summit
July 5, 2024, 12:16 a.m.PM Narendra Modi Calls for Responsible AI at G7 Outreach Session
June 14, 2024, 11:30 p.m.G7 Summit: India’s Prime Minister Narendra Modi in Global Spotlight
June 14, 2024, 10:25 p.m.India-Russia Relations: A Constant Bond, Says Russian Diplomat
April 4, 2024, 11:02 a.m.
Delhi-Meerut Rapid Rail: 3 Crore Passengers Rides in Record Time

Delhi Plans Cloth Banks at Metro Stations to Promote Recycling of Used Garments

97 Km, 65 Stations: Delhi Govt Prepares Major Metro Expansion Plan for Outer Regions

Delhi Bans Advance Fee Collection in Private Schools, Mandates Monthly Payments

Gurgaon to see relief from heat as pre-monsoon showers expected from May 2

Delhi to launch mobile health vans and mass wedding schemes for construction workers

Delhi plans new borewell policy to regulate groundwater use amid shortage concerns

New Academic Shift: DU SOL Brings Foreign Language Courses to Online Platform

Delhi LG Sandhu Attends IPCW’s Samanvay Event Celebrating Academic Achievements

Citizens Come Together to Restore Cleanliness at Historic Mehrauli Archaeological Park

Sudden Weather Shift Brings Rain, Hail and Relief from Heatwave in Delhi-NCR

Delhi Development Authority Launches Sale of 142 Prime Plots to Generate Revenue

Heritage Meets Commerce: Delhi Monuments to Feature Souvenir Shops via PPP Initiative

Delhi Exhibit Explores Philosophical Contrasts Through Contemporary Art

Groundwater Crisis: NGT Forms Panel to Suggest Urgent Conservation Measures

Final Clearance Granted: Noida Airport Eyes Commercial Flights from June 2026

From Colleges to Connaught Place: Delhi’s Anti-Drug Drive Gets Creative Boost

1,300 Pilgrims from Delhi to Undertake Somnath Yatra in Grand Spiritual Initiativ

Government Data Shows Massive Jump in Digital LPG Refill Bookings to 98%

MCD Steps In to Restore Historic Delhi Gate Linked to 18th Century Legacy

Safdarjung, Ayanagar, Najafgarh Emerge as Delhi’s Hottest Areas in New Satellite Survey

Women’s Reservation Bill Takes Centre Stage in Delhi Assembly Special Session

MCD Expands Healthcare Network with New Eye Care Centre in East Delhi

Delhi to Get New Multi-Level Parking Hubs as DDA Opens E-Auction

₹6,270 Crore Development Push for Varanasi: PM Modi Visit Scheduled for April 28

Uttar Pradesh Emerging as Model for Scientific and Sustainable Farming, Says CM Yogi

Delhi Records Year’s Highest Temperature as IMD Issues Heatwave Alert for Next Two Days

Global Collaboration at SGTB Khalsa College: World Bank Group Joins Hands for Student Quiz Event

PM Modi in Kolkata: Boat Ride on Hooghly Reflects Cultural and Spiritual Significance of Ganga

Uttar Pradesh Board Result 2026: Girls Dominate as Lakhs Pass Class 10 and 12 Exams

Massive Rail Revamp: 55,000 km Track Renewal Strengthens Indian Railways Network

Delhi Jal Board Launches e-KYC Drive to Identify Fake Water Connections and Upgrade Services

BJP Names Rohini Councillor Pravesh Wahi as Candidate for Delhi Mayor Poll

Indian Air Force Tests Highway Landing Capability on Purvanchal Expressway

Delhi Govt Pushes for Real-Time Pollution Source Data to Strengthen Air Quality Action

Delhi Jal Board to Conduct 6-Month Door-to-Door Survey to Fix Water Database

Bilaspur to Get Direct Air Connectivity to Delhi from May 3, Boost to Regional Travel

Delhi Hospital Becomes Zero-Waste Model, Sets Benchmark for Sustainable Healthcare

CM Pushkar Singh Dhami Extends Warm Welcome to Devotees on Sacred Char Dham Yatra 2026

Delhi Makes PUC Mandatory for Fuel: Vehicles Without Valid Certificate to Be Denied Petrol & Diesel

APY Scheme Expansion: 9 Crore Indians Join Government Pension Plan for Secure Retirement

Indian artist Sajid Wajid Sheikh awarded prestigious Henry Clews Prize in France

IMD issues warning as Delhi enters heatwave spell from today

Yamuna floodplain under scanner: DDA to finish O-zone drone survey in 2 months

Earth Day 2026: Global Call to Protect Environment Amid Rising Development Challenges

Noida International Airport may need Indian CEO to begin flights

Healthcare push: 108 new PG, DNB seats proposed in Delhi hospitals

Delhi government to track, assess and support care-leavers under new ₹3.5 crore initiative

PM Narendra Modi Urges Civil Servants to Drive ‘Viksit Bharat’ Mission with Renewed Focus

Real-Time Monitoring Boost: Delhi Sets Up Control Room for House-Listing Work

Delhi LG Reviews Fire Service Preparedness, Calls for Stronger Safety Enforcement

Skill Development Initiatives for Minority Youth Gain Momentum with Focus on Modern Job Roles

Delhi Tightens Tree Protection Rules with 24x7 Monitoring System and SOP

BHIM UPI Expands Services, Enables Instant CIBIL Score Access for Users

IMD Warns of Heatwave Across North, Central & Eastern India; Temperatures Set to Rise by 2–4°C

DTC Sees Financial Turnaround, Income Rises by ₹173 Crore in 2025–26

Delhi Schools to Lead Year-Long Anti-Pollution Drive, Turning Questions into Action

MCD Orders Immediate Rollout of Revised Toll Rates Across Delhi

Delhi Plans Semiconductor Push, Draft Policy Aims to Attract Investment and Boost Jobs

NBCC Bags ₹5,779 Crore in Mega E-Auction of 14 Lakh Sq Ft Sarojini Nagar Commercial Space

Delhi NCR Gets First Bat Field Guide; Citizens Join Night ‘Bat Walk’ at Sunder Nursery

MCD Commissioner Reviews Monsoon Preparedness, Orders Faster Execution of Key Works

‘Mini India’ to Debut as DDA Prepares Phase-Wise Opening of Bharat Vandana Park

NDMC Strengthens ‘Broom-Free City’ Mission with Modern Cleaning Machines

Nitish Kumar Allotted Type-8 Bungalow in Lutyens’ Delhi, Security Upgraded to Z+

Delhi CM Flags Off 200 New Electric Buses, Boosts Green Public Transport Push

Census 2027 Begins in Delhi as Houselisting Phase Gets Underway

Specialised Machine-Based Tree Transplantation: Delhi to Select Approved Agencies

Delhi Govt Plans Modern Skill Labs in Four ITIs to Improve Employability

Delhi Weather Update: Hot Days, Partly Cloudy Skies Likely on April 16–17

10 Lakh Vehicles Milestone in Lucknow: Yogi Adityanath Showcases UP’s Industrial Growth

Mukarba Chowk Set for Big Traffic Relief as 3 New Underpasses Near Completion

CBSE 10th results 2026: Strong performance as 23 lakh students pass, 55k cross 95%

NHAI Begins Verification of Vehicle Registration Numbers Linked to FASTags

Delhi MCD Mayor Election to Be Held on April 29

Delhi Govt Plans 5 km Yamuna Flood Barrier to Tackle Recurring Inundation

Saharanpur Wildlife Corridor: PM Modi Reviews Green Infrastructure Along Expressway

UP Wage Revision 2026: New Salary Rates Announced After Noida Industrial Protests Turn Violent

DU Plans Alumni–Corporate Funding Push for Needy Students’ Semester Abroad Dreams

Ambedkar Jayanti: PM Modi Highlights Babasaheb’s Role in Shaping Modern India


Delhi Upgrades Stormwater System with New Precast Drain Technology

Baisakhi Celebrated Nationwide: PM Modi Highlights Farmers’ Contribution and Sikh Heritage

DU May Revamp Undergraduate Course Combinations to Address Vacant Seats

Govt Eyes Formula One Comeback in India Before 2027, Says Sports Minister Mandaviya

Major Infra Push: 19.2-km Yamuna Elevated Road, Metro Expansion Planned in Delhi

Major Boost for Defence: India to Co-Produce Advanced F414 Engines
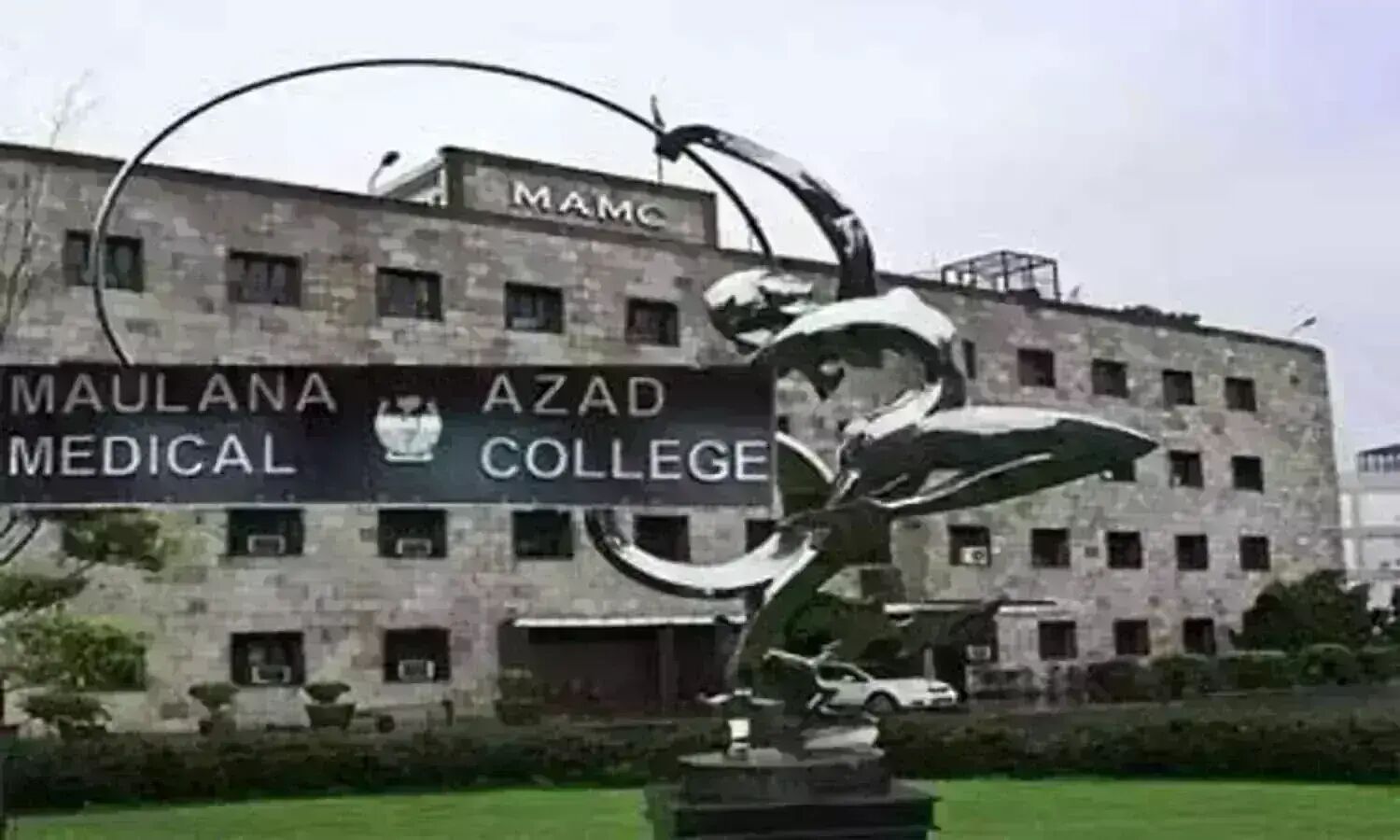
Major Infra Push: Two Hostels to Come Up at Maulana Azad Medical College

PM Narendra Modi to Address ‘Nari Shakti Vandan Sammelan’ at Vigyan Bhawan Today


Schools Introduce Puzzles, Ethical AI Lessons to Boost Student Skills

Delhi Metro’s Double-Decker Flyover Project on Track for Year-End Launch

Guru Dronacharya, IFFCO Chowk Stations May Get Multi-Level Parking Boost

Petrol, Diesel Rates Across India on April 11: Delhi, Mumbai, Kolkata, Bengaluru & More

Big Boost for EV Buyers: Delhi Draft Policy Offers Full Road Tax Exemption

Foreign Secretary Vikram Misri Meets US Officials to Deepen Defence & Tech Ties

CM Rekha Gupta directs agencies to complete pre-monsoon desilting on time, sets June 30 deadline

Delhi: DDA plans seven-day heritage trail in Mehrauli Archaeological Park, Sanjay Van

Delhi University Hosts ICCR Day Celebrations, Highlights Cultural Exchange

ISRO Clears Crucial Gaganyaan Milestone with IADT-02 Test at Sriharikota



NGT Cracks Down: GPS Must for All Water Tankers

VP C.P. Radhakrishnan Addresses IGNOU Convocation, Over 32 Lakh Students Awarded Degrees

Big Relief for Mobile Users: TRAI Plans Flexible, Budget-Friendly Voice & SMS Packs

MCD Plans Revamp of Areas Under Six Major Flyovers

DMRC Launches ₹3.19 Crore Tender for Central Vista Depot Design

Clean Roads, Cleaner Air: MCD Launches ₹1,415-Crore Dust-Control Drive

Pragati Maidan Underpass‑5 Back on Track, Work to Complete by December 2026

BBMB Installs Early Warning System on Beas River to Alert Residents During Water Release

Delhi–Dehradun Expressway Set for April 14 Launch; Travel Time to Halve

Delhi Dedicates 21% of Budget to Green Initiatives; Yamuna Clean-Up Tops List

Delhi Bans Direct LPG Sales from Godowns, Boosts Access with 5-kg Cylinders and Help Desks

MCD Plans Major Push to Bring More Properties Under Tax Net in FY 2026–27

Record-Breaking UPI Growth: 22.64 Billion Transactions in March, PhonePe Leads Market Share

Delhi Police Reunites 155 Missing Persons in March Under ‘Operation Milap’

From Paper to Digital: PM Modi Initiates Census 2027 with Self-Enumeration Drive

Big Financial Changes from April 1: What New Tax Rules, Fuel Prices and Banking Updates Mean for You

Govt Strengthens SHe-Box Portal to Simplify Workplace Sexual Harassment Complaints


CBDT Signs Record 219 Advance Pricing Agreements in FY 2025–26, Crosses 1,000 Mark
PM Modi Inaugurates Second Chip Plant; Vaishnaw Says 6 Semiconductor Units Ready by 2027

Mahavir Jayanti Observed Nationwide with Devotion; Leaders Highlight Ahimsa and Compassion

Ram Navami 2026 Celebrated Nationwide with Devotion and Joy


Delhi CM Rekha Gupta Holds ‘Kheer Ceremony’ Ahead of Budget Session; Political Activity Intensifies

Delhi Assembly Launches AI Chatbot ‘Vidhan Saathi’ for MLAs, First-of-Its-Kind Initiative in India

India Pharma Market Set to Double by 2030, Global Demand and Policy Support Fuel Growth

Rain Brings Relief to Delhi-NCR, IMD Issues Yellow Alert

NACO Holds ‘Suraksha Sankalp Karyashala’ to Boost District-Level HIV Response

PM Narendra Modi Extends Chaitra Navratri Greetings, Links Festival to ‘Viksit Bharat’ Vision

PM Modi Bids Farewell to Retiring Rajya Sabha Members, Calls Their Experience Nation’s Asset

Bharat Tribes Fest 2026 Begins in Delhi Tomorrow; 200+ Stalls to Showcase Tribal Heritage

Ayushman Bharat Scheme Crosses 43.52 Crore Cards; Over 11.69 Crore Treatments Approved

Affordable Housing Push: 116 Lakh Homes Under Construction, 97 Lakh Completed Under PMAY-U

Delhi Budget Session 2026 from March 23–25; Major Infra Projects Get Cabinet Nod

Delhi Govt Approves Free Bus Travel for Transgender Persons in DTC and Cluster Buses
Union Minister Tokhan Sahu Honours Anil Marwah for Unique Delhi Metro Tradition
Union Minister Tokhan Sahu Felicitates Delhi Metro Enthusiast Anil Marwah


Piyush Goyal Says India–US Trade Pact Ensures Full Protection for Farmers

India AI Impact Summit 2026 Kicks Off in New Delhi

PM Modi Unveils ‘Seva Teerth’ for PMO, NSCS and Cabinet Secretariat Complex


Ravi Shankar Prasad Criticises Rahul Gandhi, Alleges Repeated Attempts to Undermine Indian Democracy


Shaina NC Criticises Rohit Pawar for ‘Sensationalising’ Ajit Pawar Plane Crash Remarks

Delhi CM Rekha Gupta Highlights ₹12 Crore Scholarships and Welfare Schemes for Labourers

Sudhanshu Trivedi Accuses Congress of Appeasement, Warns Against Understanding With Jamaat-e-Islami

BJP MP Nishikant Dubey Targets Rahul Gandhi Over Respect for Parliament

India Confirms Consular Access to 16 Detained Sailors in Iran; Eight Released

Manjinder Singh Sirsa Condemns Rahul Gandhi’s Remarks, Seeks Action from Lok Sabha Speaker

Athawale Slams Rahul Gandhi’s Remarks on PM Modi, Defends Prime Minister’s Parliamentary Role



Rahul Gandhi a Leader of Pessimism, Not Opposition, Says BJP Spokesperson Shehzad Poonawalla


Pakistan Avoiding Match Shows Fear of Defeat, Says BJP MP Shashank Mani; Defends Union Budget

Union Budget 2026-27 a ‘Next-Generation’ Budget, Says Delhi CM Rekha Gupta

Shehzad Poonawalla Targets INDI Alliance Over Stand on Illegal Immigration

PM Modi Welcomes UAE President Sheikh Mohamed bin Zayed Al Nahyan with Warmth & Symbolic Gifts

Nitin Nabin Takes Charge as BJP National President, Pledges Commitment to Party Ideology

PM Modi Labels “Urban Naxalism” as a Growing International Threat

BJP Alleges TMC Threats Led to Death of Booth Level Officer in West Bengal

BJP-Led Mahayuti’s Civic Poll Sweep Reflects Trust in PM Modi, Says Shahzad Poonawala

India Closely Monitoring Impact of US Tariffs on Iran Trade, Says MEA

Tarun Chugh Accuses AAP Leaders of Disrespecting Sikh Traditions at Maghi Mela in Punjab



Congress Boycotts Somnath Swabhiman Parv, BJP Alleges Decades-Old Politics of Appeasement

India Will Not Tolerate Interference in Internal Matters, Says BJP Leader Gaurav Vallabh

Tripura Student Murder Case Should Go to Fast-Track Court, Says BJP MP Brij Lal

BJP MP Yogender Chandoliya Assures Protection of Aravali, Cites Supreme Court Stay



CM Rekha Gupta Calls for Greater Inclusion and Support for Divyangjans

Shehzad Poonawalla Questions TMC Over Action Against MP Accused of ‘Lowering Parliament’s Dignity’


Minister Shekhawat Slams Opposition for ‘Politicising’ Vande Mataram During Lok Sabha Debate

Delhi | Govt Reviewing Night Shelter Systems After Coolie Camp Tragedy: Minister Ashish Sood

EAM Jaishankar Highlights Biosecurity Gaps, Calls for Stronger Global South Role at BWC Conference

PM Modi Hosts Indian Blind Women’s Cricket Team, Winners of the Inaugural T20 World Cup


Delhi | Defence Minister Rajnath Singh Praises India’s Defence Innovators at Swavlamban 2025 Seminar

Jaishankar Meets Afghan Minister in New Delhi, Reaffirms India’s Support for Afghan People

Centre Announces Special Trains for 350th Martyrdom Anniversary of Guru Tegh Bahadur Ji


Tarun Chugh Says Public Has Rejected Arrogance, Reaffirmed Faith in Modi–Nitish Leadership



PM Modi Emphasizes Need for Justice in Local Languages, Lauds Supreme Court’s Translation Initiative

PM Modi in Bettiah: NDA Governance Has Ensured Safety, Dignity, and Empowerment for Women in Bihar


Delhi Govt Organises Dev Deepawali, Promotes Unity Through Celebration of All Festivals


Railways to Develop 76 Passenger Holding Areas Nationwide for Better Crowd Management and Comfort

Delhi CM Rekha Gupta Says Bihar’s Development Possible Only Under NDA Rule


Indian Railways Begins Broadcast of Chhath Songs at Stations to Celebrate the Festival

Delhi Conducts First Successful Cloud Seeding Trial, Says Minister Manjinder Singh Sirsa

Rajnath Singh Highlights Defence Reforms, Civil-Military Integration at Book Launch in Delhi

Ashwini Vaishnaw – A Minister Who Cares

Manoj Tiwari Responds to AAP’s Remarks, Says Delhi’s Air Quality Improving Under BJP’s Efforts


Union Minister JP Nadda Attends Cultural Event Organised by Embassy of Mongolia

Union Minister Mansukh Mandaviya Highlights India’s Rising Success in Global Sports

BJP’s Sambit Patra Accuses Mamata Banerjee of Threatening Riots over SIR in Bengal



PM Modi Inaugurates ₹34,200 Crore Development Works in Bhavnagar, Sets Vision for Maritime Growth

Union Railways Minister Ashwini Vaishnaw Flags Off Three New Amrit Bharat Express Trains

BJP MP Bansuri Swaraj Offers Mangala Aarti at Jhandewalan Temple

Amit Shah and JP Nadda Mark Deendayal Upadhyay’s Birth Anniversary with Sapling Plantation in Delhi

JPC Chairperson PP Chaudhary Highlights Economic Benefits of ‘One Nation, One Election’

BJP MP Sambit Patra Highlights Public Relief from GST 2.0 Reforms

Union Minister Gajendra Singh Shekhawat Hails ABVP’s Big Win in DUSU Elections

Rajnath Singh Pays Tribute to Lal Bahadur Shastri’s Leadership During 1965 War

Global Envoys from 72 Nations Gather in Delhi, Showcasing India’s Growing Global Influence

Nation Marks PM Modi’s 75th Birthday with Seva Pakhwada and Blood Donation Drives

Union Minister Ashwini Vaishnaw Extends Greetings to PM Modi, Highlights Mega Blood Donation Drive

Delhi CM Rekha Gupta Attends Engineer’s Day Celebration

India to Host AI Impact Summit in February 2026, Announces Union Minister Ashwini Vaishnaw

Delhi CM Inaugurates India’s First Hotline Maintenance Van for Uninterrupted Power Supply

Minister Kapil Mishra on Launch of Book ‘BJP Delhi Win – Modi Ki Guarantee’

Kiren Rijiju Hails C.P. Radhakrishnan’s Victory as ‘Conscience Vote’ in Vice Presidential Election

PM Modi Praises CP Radhakrishnan’s Candidature for Vice President

FM Nirmala Sitharaman Slams Congress Over GST, Tobacco Tax Proposal

Kapil Mishra Condemns Remarks Against PM Modi’s Late Mother, Says Public Outraged

Delhi Minister Kapil Mishra Highlights Cultural Outreach, Criticizes Rahul Gandhi’s Remarks

Delhi Police use water cannons on BJP protest over remarks against PM Modi and his late mother

Delhi CM Rekha Gupta Condemns Derogatory Remarks Against PM Modi and His Mother

Amit Shah Felicitates Security Personnel for Successful Operation Mahadev in Pahalgam

BJP MP Jagdambika Pal Accuses Tejashwi Yadav of Misleading People on Election Commission

PM Modi at ET World Leaders Forum 2025: India Poised to Drive Global Growth

BJP MP Yogender Chandolia Defends Delhi CM Rekha Gupta, Slams AAP Over ‘Resentment’ Claims

BJP MP Bansuri Swaraj Slams TMC MPs Over Conduct in Parliament
NDA to File Vice Presidential Nomination of C.P. Radhakrishnan on August 20

PM Modi to Inaugurate Rs 11,000 Crore Highway Projects to Ease Delhi Traffic



BJP’s Manan Kumar Mishra Criticises Rahul Gandhi Over EC March, Says Allegations Lack Proof

BJP’s Gaurav Bhatia Slams Rahul Gandhi Over EC Allegations, Calls Claims ‘Baseless’

Delhi Minister Condemns Ban on Indian Attire in Restaurant, Calls it ‘Cheap Mentality’

Sambit Patra Slams Rahul Gandhi's Allegations on Election Commission as "Selective Outrage"

Rahul Gandhi Has No Credibility, Must Apologise Again: BJP MP Sambit Patra

BJP MP Shashank Mani Urges Opposition to Follow Procedure Amidst Parliament Stalemate

Rahul Gandhi Should Resign as LoP, Says Manjinder Singh Sirsa Over Jaitley Remark
Government Ready for Discussion, But SIR Debate Depends on Parliamentary Rules: Kiren Rijiju


Congress Acting as PR Agency of Pakistan, Says BJP MP Tejasvi Surya After PM Modi’s Speech

‘Operation Sindoor is India’s New Normal’: BJP MP Baijayant Panda Defends India's Global Standing

Jharkhand Govt Renames Atal Mohalla Clinics; BJP Calls It a Disrespect to Atal Bihari Vajpayee

Delhi CM Rekha Gupta Inaugurates 24 New Arogya Mandirs, Sets Target of 1100 by March 2026

Rahul Gandhi Should Avoid Divisive Remarks on Pakistan Issues: BJP MP Gulam Ali Khatana

Govt Ready for Debate on Operation Sindoor, Opposition Avoiding Discussion: BJP’s Shazia Ilmi

BJD MP Sasmit Patra Hopes for Clarity on Operation Sindoor Discussion Timing

Union Home Minister Highlights Government's Vision for 2036 Olympics and Sports Development

EU-India Free Trade Agreement Talks Progressing Well: MEA Spokesperson Randhir Jaiswal



Sambit Patra Slams Congress Over Aadhaar-Voter ID Debate, Calls It ‘Double Standards’

Defence Minister Rajnath Singh Chairs First NCC Alumni Association Governing Council Meet

Anil Antony, New Nagaland Inductees Meet BJP President JP Nadda in Delhi

BJP’s Syed Zafar Islam Slams Owaisi, Accuses Him of Provoking Muslims

Virat Kant Joins BJP, Tarun Chugh Calls It a ‘Masterstroke’

Kiren Rijiju Criticizes AIMIM-RJD Alliance Talk, Reaffirms India’s Secular Fabric

BJP MP Manan Kumar Mishra Slams Mahagathbandhan Talks with AIMIM


NDMC to Promote Art and Culture as Part of 'Viksit Delhi' Vision: Kuljeet Singh Chahal

Delhi CM Rekha Gupta: Government to Act on End-of-Life Vehicles Amid Pollution Concerns


BJP MP Bansuri Swaraj: “Emergency Was the Murder of Democracy; We Must Learn from History”


Shivraj Singh Chouhan Highlights Agricultural Roadmap After Nationwide Campaign

Amit Shah Announces Launch of PM Modi’s Book on Emergency at ‘Aapatkaal Ke 50 Saal’ Event

BJP Marks 50 Years Since Emergency, Calls It ‘Murder of Democracy’

India’s FTAs Are Industry-Backed and Nation-Benefiting: Piyush Goyal

Delhi to Celebrate Yoga Day at 11 Locations with 20,000 Participants: CM Rekha Gupta

BJP MP Kangana Ranaut Highlights Women-Centric Reforms, Slams Political Nepotism

BJP MP Ajeet Gopchade Proposes Border Management Authority, Receives Positive Response from PM Modi

Delhi CM Rekha Gupta Unveils Plan to Deploy Advanced Road-Cleaning Machines to Tackle Air Pollution

BJP’s Ajay Alok Slams Pawan Khera Over Remarks on Air India Plane Crash

Union Minister Ramdas Athawale Confirms Death of Vijay Rupani in AI-171 Plane Crash

PM Modi Hosts International Delegations at 7, Lok Kalyan Marg

Delhi CM Rekha Gupta Highlights Benefits of Triple-Engine Govt, Announces ₹870 Crore Grant to MCD



Delhi Minister Manjinder Singh Sirsa Condemns Punjab CM Bhagwant Mann’s Remarks


President Droupadi Murmu Highlights Strong Bilateral Ties on Historic Diplomatic Anniversary



Tarun Chugh Hails PM Modi’s Leadership, Says India Now Symbol of Inclusive, Self-Reliant Growth

Nalin Kohli Reacts to Impeachment Motion Against Justice Yashwant Varma, Calls for Full Transparency

BJP MP Deepak Prakash Slams Kharge, Calls PM Modi's 11-Year Tenure a 'Golden Age'

BJP's Pradeep Bhandari Slams Congress, Says 'Emergency in Rahul Gandhi's Camp'

Delhi CM Rekha Gupta Announces New Tourist Circuit with Modern Attractions


BJP MP Baijyant Panda Highlights Unity Ahead of Global Outreach on Terrorism

Delhi Govt Partners with TERI to Raise Environmental Awareness Among Children

BJP MP Praveen Khandelwal Slams Mallikarjun Kharge Over Remarks on Military Operations


Union Home Minister Amit Shah Launches New OCI Portal to Enhance User Experience


BJP’s Shehzad Poonawalla Slams Congress Over Shashi Tharoor Delegation Row

Amit Shah Inaugurates New Multi-Agency Centre at North Block, Hails Operation Sindoor

BJP MP Jagdambika Pal Praises Indian Army's Response to Terror Attacks

Delhi Minister Sirsa Slams Atishi, Blames AAP for Pollution Crisis

India Puts Indus Water Treaty in Abeyance: Gajendra Singh Shekhawat Reaffirms PM Modi’s Stand

Union Minister Ramdas Athawale Supports PM Modi’s Tough Stance on Pakistan

Delhi CM Rekha Gupta Responds to AAP Leader Atishi on India-Pakistan Tensions

India’s Retaliation Under Operation Sindoor Leaves Deep Impact: Shahnawaz Hussain

Delhi CM Rekha Gupta Launches Solar Power Initiative to Boost Clean Energy

Vice Admiral AN Pramod Highlights India’s Robust Naval Surveillance and Defence Strategy

Air Marshal AK Bharti on Operation Sindoor: Future Conflicts Will Demand New Strategies

Delhi CM Rekha Gupta Expresses Solidarity Amid India-Pakistan Tensions

Shehzad Poonawalla Slams Congress for 'Supporting Pakistan Narratives' Amid Tensions

Indian Armed Forces on High Alert Amid Pakistan Troop Movements: Wing Commander Vyomika Singh

Pakistan Misused Civilian Air Routes to Evade Detection, Says Colonel Sofiya Qureshi

PM Modi Holds Key Security Meeting with Top Defence Officials at 7, LKM

BJP Spokesperson Jaiveer Shergill Says India Giving Pakistan 'Dose' to End Terror Addiction

BJP Leader Shahnawaz Hussain Praises Army’s Action, Says Terrorists and Their Masters Are Trembling

Union Minister Shivraj Singh Chouhan: Terrorists Targeted, Not Civilians; Assures Food Security

BJP’s RP Singh Says Pakistan Is Seeking Aid After One-Day War, Warns of Befitting Reply

Rajnath Singh Hails Precision of Operation Sindhoor, Says Nine Terrorist Camps Destroyed


CM Rekha Gupta Calls Meeting with Manohar Lal Khattar a Turning Point for City's Development

Rajnath Singh Warns Against Testing India's Patience, Says Nation Ready for 'Quality Action'

Amit Shah Holds Key Meeting with Border State Leaders on National Security

Shehzad Poonawalla Slams Congress for Questioning Operation Sindoor

Rajnath Singh Says India Follows Lord Hanuman's Principle in Defence Actions

Sonal Goel IAS, a Trailblazer in Public Service and Women Empowerment
Delhi Plans 1000 Water Sprinkler Vehicles to Fight Air Pollution

Delhi Government Shares New Plan to Fight Pollution in 2025-26


Amit Shah Chairs High-Level Review Meeting on Implementation of New Criminal Laws

Angolan President Hails Historic State Visit to India After 38 Years

BJP’s Sambit Patra Alleges Senior Congress Leader Secretly Stayed in Pakistan

Ministry of Tribal Affairs and BPCL to Set Up 75 Space Labs in EMRS Schools with ISRO’s Support

Minister Manjinder Singh Sirsa Welcomes Ban on 16 Pakistani YouTube Channels

BJP MP Ravi Shankar Prasad Slams Opposition Leaders Over Statements on Pahalgam Terror Attack

BJP MP Nishikant Dubey Slams Tariq Hamid Karra Over Call for Talks with Pakistan

BJP MP Ravi Shankar Prasad Criticizes Robert Vadra Over Remarks on Pahalgam Terror Attack

BJP MP Nishikant Dubey Slams Karnataka CM Siddaramaiah Over Remarks on Pahalgam Attack

BJP’s Ravi Shankar Prasad Slams Congress Leaders Over Remarks on Pahalgam Terror Attack
BJP MP Kamaljeet Sehrawat Condemns Pahalgam Terror Attack, Expresses Faith in PM Modi's Leadership

Raja Iqbal Singh Elected Delhi Mayor, Vows Development and Cleanliness

India Revokes Most Visas Issued to Pakistani Nationals Amid Security Concerns
Delhi CM Rekha Gupta Backs New Report Aimed at Empowering Women in Sports

BJP MLA Satish Upadhyay Reacts to Pahalgam Terror Attack

Govt to Take Stricter Action Post-Pahalgam Attack, Says Kiren Rijiju

Senior Ministers Depart After High-Level All-Party Meeting on Terror Attack

Kiren Rijiju Says All Parties United Behind Government's Strong Stand on Terrorism

BJP Leader Tarun Chugh Condemns Pahalgam Terror Attack, Says “Terrorism Can Never Break India”

Shivraj Singh Chouhan Condemns Pahalgam Terror Attack, Vows Justice

CM Rekha Gupta Pays Last Respects to Navy Lieutenant Vinay Narwal




BJP Leader Nalin Kohli Criticizes Robert Vadra Over Statement on Terror Attack

BJP MP Praveen Khandelwal Highlights Significance of US Vice President JD Vance's Visit to India

BJP’s Tarun Chugh Slams Mamata Banerjee, Accuses Her of Vote Bank Politics and Misrule in Bengal

Union Minister Arjun Ram Meghwal Urges Public to Stay Alert Amid Rumours on Waqf Amendment Act

Union Minister Arjun Ram Meghwal Confirms Progress of JPC on ‘One Nation, One Election’

BJP MP Yogender Chandolia Slams Rahul Gandhi Over Remarks on Election Commission Made Abroad

BJP MP Praveen Khandelwal Slams Quick Commerce & E-Commerce Practices at National Conclave

Murshidabad Violence: BJP’s Mukhtar Abbas Naqvi Slams TMC Government for 'Protecting Looters'

Robert Vadra Summoned by ED in Shikohpur Land Scam; BJP's Gaurav Bhatia Calls Him 'Land Mafia'

Delhi Cabinet Approves Continuation of Power Subsidy for Farmers, Riot Victims, Lawyers

No Ban on Autorickshaws, Says Delhi Transport Minister Amid EV Policy Buzz

Robert Vadra Summoned by ED; BJP's Nalin Kohli Questions Relevance of Documents Submitted

ED Files Prosecution Complaint Against Rahul Gandhi, Sonia Gandhi in National Herald Case

New Train Linking Ambedkar Nagar to Delhi Launched Ahead of Ambedkar Jayanti, Says Vaishnaw

Special Train Services Inaugurated Connecting Kota and Delhi on Ambedkar Jayanti

Leaders Across Party Lines Pay Tribute to Dr. B.R. Ambedkar on Jayanti at Parliament’s Prerna Sthal

Leaders Across Party Lines Pay Homage to Dr B.R. Ambedkar at Prerna Sthal on Ambedkar Jayanti

Nation’s Top Leaders Pay Tribute to Dr B.R. Ambedkar at Prerna Sthal on Ambedkar Jayanti

Leaders Across Party Lines Pay Tribute to Dr B.R. Ambedkar on Ambedkar Jayanti


Delhi Minister Parvesh Verma Flags Poor Conditions in Paalam, Vows Probe into School Infrastructure

Piyush Goyal Hails Italy-India Strategic Ties, Urges Action on Joint Roadmap 2025–29

Piyush Goyal at India-Italy Forum: “India First” Driving Trade Talks, Startups Key to Global Growth

BJP National President JP Nadda paid floral tributes to Dr. Syama Prasad Mookerjee and Pandit

Delhi CM Rekha Gupta Announces 50-Acre Park in Uttam Nagar

Minister Parvesh Verma: Road Recarpeting Drive Underway to Prevent Pollution and Waterlogging

Manoj Tiwari: Extradition of Tahawwur Rana Shows Modi Govt’s Zero Tolerance Against Terror


Delhi CM Rekha Gupta on Yamuna Cleanup: “Work is Ongoing in a Time-Bound Manner”

Delhi to Get One of Its Most Beautiful Parks in Uttam Nagar, Says BJP MLA Pawan Sharma

PM Modi Attends Navkar Mahamantra Program Barefoot, Chooses to Sit Among Public


Murshidabad Violence ‘State-Sponsored’, Says BJP’s Sukanta Majumdar; Targets Mamata Banerjee

Waqf Amendment Act: BJP Minority Morcha Chief Says 'Allah's Work Done Through PM Modi'

BJP MLA Shikha Roy Slams Mahua Moitra and Saurabh Bharadwaj Over Fish Market Allegations in CR Park


India’s Electronic Exports See Sixfold Growth, Says Union Minister Ashwini Vaishnaw





Union Minister Hardeep Singh Puri Responds to Rahul Gandhi's Statement on Economic Issues

Union Minister Hardeep Singh Puri Announces LPG Price Hike

Minister Ashwini Vaishnaw Updates on AI Mission Progress and GPU Facility Expansion

Electronics and IT Minister Ashwini Vaishnaw Highlights Growing Creator Economy Ahead of WAVES 2025

Delhi Minister Manjinder Singh Sirsa Criticizes AAP’s Actions in Punjab's Education System

PM Modi Acknowledges BJP Karyakartas on Party’s Sthapana Diwas


BJP MP Manoj Tiwari Defends Waqf Amendment Bill, Calls It Constitutional and Humane


BJP MP Arun Govil Appeals to West Bengal Government on Ram Navami Procession Issue

BJP MP Arun Govil Urges Strict Action on Mahadev App Controversy

CM Rekha Gupta Promises Clean Water and Health Benefits for Delhiites

Kiren Rijiju Praises Historic 17-Hour Debate on Waqf (Amendment) Bill in Rajya Sabha

Both Lok Sabha and Rajya Sabha Pass Waqf (Amendment) Bill, Chairman Calls It a ‘Big Reform’

Waqf Amendment Bill Marks a New Dawn for India, Says BJP MP Shashank Mani

Waqf Amendment Bill a Triumph for Equality, Says BJP MP Damodar Agarwal

Waqf Amendment Bill: Giriraj Singh Slams Opposition, Highlights Benefits for Muslims

Prime Minister Narendra Modi and PM Paetongtarn Shinawatra Hold Joint Press Meet in Bangkok

Union Minister Sanjay Seth Hails Waqf Amendment Bill, Says It Brings Happiness to 140 Crore People

BJP MP Manoj Tiwari Hails Passage of Waqf (Amendment) Bill 2024, Calls it a Victory for Constitution


Union Defence Minister Rajnath Singh Flags Off Three Mountaineering Expeditions

Union Minister Kiren Rijiju Speaks on Waqf Amendment Bill 2025 in Rajya Sabha


Rajya Sabha MP Rekha Sharma Defends Waqf Amendment Bill, Criticizes Congress for Vote-Bank Politics

BJP MP Kiran Choudhry Defends Waqf Amendment Bill, Criticizes Opposition for Misleading the Public


Union Minister Ramdas Athawale Defends Waqf Amendment Bill, Calls Out Political Criticism



Union Minister Ashwini Vaishnaw Highlights India's Economic Growth Strategy on CBI Foundation Day

Union Minister Ramdas Athawale Defends Waqf Amendment Bill, Urges Muslim Community to Support

BJP MP Kiran Choudhry Defends Waqf Amendment Bill, Criticizes Congress for Objections

BJP MP Ravi Kishan Defends Waqf Amendment Bill, Highlights Benefits for Poor Muslims

Jagdambika Pal Defends Waqf Amendment Bill Amidst Controversy and Protests

BJP Leader Shehzad Poonawalla Reacts to Kerala Catholic Bishops' Support for Waqf Amendment Bill

BJP President Rajeev Chandrasekhar Reacts to Mohanlal's Movie 'L2: Empuraan'




BJP MP Rao Rajendra Singh Criticizes SP MP Ramji Lal Suman’s Remarks on Rana Sanga

BJP MP Ravi Shankar Prasad Criticizes SP MP Ramji Lal Suman's Remarks on Rana Sanga

Union Minister Giriraj Singh Criticizes Mamata Banerjee’s Remarks During UK Visit


BJP MP Manoj Tigga Criticizes TMC Government Over Attack on Arjun Singh’s Residence


BJP MP Deepak Prakash Criticizes Jharkhand Government Over Rising Crime Rates

BJP MP Ravi Kishan Responds to Sambhal SDM's Statement on Public Namaz Practices

Delhi BJP MLA Calls for Action on Traffic Congestion, Supports Closure of Meat Shops During Navratri

BJP MP Kamaljeet Sehrawat Supports Delhi Budget, Calls it a Roadmap for Development

BJP MP Bhartruhari Mahtab Hails 'One Nation, One Election' Meeting as "Fruitful"

BJP Leader Shehzad Poonawalla Slams Kanhaiya Kumar's Remarks on Bihar

AIMPLB Announces Nationwide Agitation Against Waqf Amendment Bill, BJP MP Responds

BJP Leader Gaurav Bhatia Calls for Sonia and Rahul Gandhi's Resignation Over Bofors Issue

BJP MLA Satish Upadhyay Hails Delhi Budget 2025-26 as a Game-Changer

BJP MLA Arvinder Singh Lovely Praises Delhi Budget 2025-26


Delhi Minister Manjinder Singh Sirsa Accuses AAP of Looting DTC, Cites CAG Report


BJP MLA Kailash Gahlot Inspects Passenger Terminal Construction Project in Delhi

President Draupadi Murmu Praises AIIMS for 69 Years of Excellence in Medical Education and Research


President Droupadi Murmu and Union Health Minister JP Nadda Attend 49th Annual Convocation of AIIMS

MEA Official Reaffirms India’s Stance on Ukraine-Russia Conflict


Delhi Minister Parvesh Sahib Singh Vows to Revamp System, Ensures Strict Action Against Negligence

BJP MP Ravi Kishan Slams West Bengal CM, Predicts BJP Victory in Upcoming Elections

BJP MP Tejasvi Surya Criticizes DMK’s Political Strategy Ahead of 2026 Tamil Nadu Elections

BJP MP Jagdambika Pal Criticizes Karnataka Assembly’s Rejection of Waqf (Amendment) Bill, 2024



BJP MP Pradeep Purohit Clarifies Statement on PM Modi's Past Life as Chhatrapati Shivaji Maharaj

President Droupadi Murmu Meets Students from Ladakh and Jammu & Kashmir at Rashtrapati Bhavan

Union Sports Minister Mansukh Mandaviya Launches Khelo India Para Games 2025

India-Albania Ties Stronger Than Ever, Says Albania’s Honorary Consul General to India

Strengthening Ties: Estonia-India Relationship Set to Grow, Says Estonia’s Secretary General

PM Modi and New Zealand PM Christopher Luxon Strengthen Ties, Vow Joint Fight Against Terrorism

BJP West Bengal President Sukanta Majumdar Discusses Strategy for 2026 Assembly Elections

BJP MP PP Chaudhary Updates on 'One Nation One Election' JPC Meeting
BJP MP Bansuri Swaraj Praises Delhi CM Rekha Gupta’s Commitment to City’s Development


BJP MP Ravi Kishan Calls Holi and 'Jumma' Controversy a Political Drama Ahead of Bihar Elections


BJP MP Dr. Ashoka Kumar Yadav Condemns Darbhanga Mayor Anjum Ara's Statement on Holi and Jumma


Amit Shah Highlights Modi Government’s Commitment to North-East Development

BJP Leader Tarun Chugh Defends Education Policy, Criticizes Opposition

BJP MP Ravi Kishan Slams Opposition Over Political Statements Ahead of Bihar Elections

Nityanand Rai Backs Amit Shah’s Statement on Mata Sita Temple in Bihar


BJP MP Anurag Thakur Praises Team India’s Dominance in Champions Trophy 2025 Victory

BJP MP Jagadambika Pal Highlights Extensive Consultations on Waqf Amendment Bill

BJP MP Manoj Tiwari Celebrates India’s Champions Trophy 2025 Victory
BJP MP Praveen Khandelwal Defends Dharmendra Pradhan, Calls for Parliamentary Decorum

BJP MP Dinesh Sharma Criticizes TMC Over Voter List Controversy, Predicts Mamata Banerjee’s Defeat

Delhi Government Approves ‘Mahila Samridhi Yojana’; BJP Welcomes Move









BJP MP Hema Malini Demands Security for Krishna Bhakts in Bangladesh

PM Modi and BJP Leaders Attend Sneh Milan Programme at C.R. Patil's Residence

PM Modi Highlights Citizen-Centered Approach of New Criminal Laws for Swift and Empowering Justice


BJP National Vice President Baijayant Jay Panda Optimistic About Delhi Election Outcome

BJP MP Manoj Tiwari Announces New Appointments in Home Guards, Including Civil Defence Personnel

BJP Leaders Present Documents on Waqf Property Claims in Karnataka at JPC Meeting

BJP MP Jagdambika Pal Raises Concerns Over Waqf Property Claims in Karnataka


PM Modi to Visit Rajasthan Twice in December: Key Focus on Investment and Governance

Union Education Minister Launches Three AI Centres of Excellence

BJP Criticizes Kerala Assembly's Resolution Against Waqf Amendment Bill

BJP Confident of Two-Third Majority in Jharkhand Assembly Elections, Focuses on Demographics

BJP Confident of Strong Performance in Wayanad By-Elections, Criticizes Rahul Gandhi's Tenure

EAM Dr. S. Jaishankar Highlights Support for Migrant Workers

Defence Minister Rajnath Singh Meets Australian Defence Secretary in New Delhi

BJP MP Sudhanshu Trivedi Accuses Congress Leaders of Land Grabbing in Karnataka

Government Pledges Justice Amid Ongoing Tensions in Bahraich, Says Brij Bhushan Singh

Union Minister Manohar Lal Khattar Critiques Opposition Leader Amid Dussehra Celebrations

President Murmu and PM Modi Felicitated at Dussehra Celebration in Delhi

BJP Responds Strongly to Congress President Kharge's Statement on Electoral Results

Delhi Celebrates Dussehra: President Droupadi Murmu and PM Modi Attend Celebrations at Red Fort

Defence Minister Rajnath Singh Embarks on Two-Day Visit to Sikkim

BJP Criticizes AAP Over Allegations of Political Manipulation in Delhi

Maharashtra to Establish Centers of Excellence in Emerging Technologies

BJP Criticizes Congress' Fact-Finding Committee on Haryana Poll Loss

Haryana BJP President Mohan Lal Badoli Criticizes Bhupinder Singh Hooda, Talks Future Leadership

Union Home Minister Amit Shah Remembers Ratan Tata at Delhi Event

Former Cricketer Kapil Dev Pays Tribute to Ratan Tata: A Legacy Remembered

BJP MP Nishikant Dubey Remembers Ratan Tata: A Loss for Jharkhand

Union Minister Anupriya Patel Celebrates BJP's Historic Victory in Haryana

BJP MP Manoj Tiwari Comments on Haryana Assembly Election Results

BJP Leader Gaurav Bhatia Praises Election Commission, Critiques Opposition's EVM Stance

BJP President Raises Questions Over Sealing of CM Arvind Kejriwal's Residence

BJP MP Manoj Tiwari Comments on ED Raid at AAP MP Sanjeev Arora’s Residence

Union Minister Giriraj Singh Speaks on Haryana and J&K Assembly Elections

Maharashtra CM Eknath Shinde Highlights Decline in Naxal Activity Amid Ongoing Security Efforts

Maharashtra CM Defends Ladli Behna Yojana Amid Opposition Criticism

BJP Criticizes Arvind Kejriwal's Housing Decisions Amid Political Tensions

BJP Condemns Congress Amid Rs 5,600 Crore Drug Seizure in Delhi

Giriraj Singh Criticizes Congress Over Treatment of MP Kumari Selja

India’s Defence Minister Highlights Evolving Strategic Partnerships in the Indo-Pacific


BJP Condemns Foreign-Funded NGOs, Accuses Opposition of Economic Sabotage

BJP Hails Inclusion of Ancient Languages in Classical List as Cultural Milestone

Major Drug Seizure in Delhi Sparks Political Debate

BJP Leader Mukhtar Abbas Naqvi Responds to Dinesh Gundu Rao's Comments on Savarkar

BJP MP Raises Alarm Over Drug Seizures, Questions Congress Connections

BJP Accuses Congress of Colluding with Drug Peddlers


BJP MP Ravi Shankar Prasad Urges Congress to Unite for Stability in Manipur

BJP Slams Karnataka CM Siddaramaiah Over Allegations of Corruption

Modi Energizes Haryana Voters, Calls for Third BJP Term in State

BJP MP Manoj Tiwari Criticizes CM Atishi's Road Inspection Efforts

BJP Leader Shehzad Poonawalla Highlights Cultural Contrast Between Parties

President Droupadi Murmu Calls for Respect Towards Healthcare Professionals

Union Minister Hardeep Singh Puri Responds to Rahul Gandhi’s Remarks on Turban and Kada

BJP Condemns Violence Against Bihar Students in West Bengal, Calls for Accountability

Union Minister Athawale Highlights Empowerment of Women at ASDC Annual Conference 2024

BJP Claims Rifts in Congress Leadership as Himachal Pradesh Political Landscape Shifts

BJP Raises Corruption Allegations Against Karnataka CM Siddaramaiah

BJP MP Manoj Tiwari Criticizes Delhi CM Atishi Over Leadership Concerns

BJP Spokesperson Critiques AAP Leadership, Compares It to Lalu-Rabri and Manmohan-Sonia Models

BJP Spokesperson Criticizes Delhi CM Atishi, Draws Parallels to Manmohan Singh

Union Home Minister Amit Shah Addresses Rally in Tohana, Criticizes Rahul Gandhi

BJP MP Accuses Kejriwal of Political Drama Amid Corruption Charges

BJP MP Critiques Farooq Abdullah's Remarks on Kashmir Stability

BJP Leader Criticizes Kejriwal's Leadership Amid Corruption Allegations

BJP President Virendraa Sachdeva Accuses Rahul Gandhi of Hypocrisy Over Reservation Policy

Chinese Ambassador Xu Feihong Highlights Positive Momentum in China-India Relations

Lok Sabha Speaker Om Birla Stresses Importance of News Credibility at Sansad TV Conclave




Amit Shah Highlights Revitalization of Cooperative Sector at National Conference

BJP MP Manoj Tiwari Advocates for 'One Nation, One Election' to Accelerate Development

Tragic Building Collapse in Karol Bagh Claims Four Lives, Injures Over 14

BJP MP Bansuri Swaraj Criticizes Kejriwal’s Resignation Announcement as a “Moral Cover”

BJP Leader Ashok Tanwar Criticizes Kejriwal's Resignation Announcement Amidst Corruption Allegations

BJP Leader Shehzad Poonawalla Slams INDI Alliance and Kejriwal’s Resignation Move

Delhi BJP Criticizes Kejriwal’s Resignation Announcement as a "Shady Stunt"

Delhi Celebrates 'Hindi Diwas' with Emphasis on Language Unity and Technological Advancements

MEA Spokesperson Provides Update on India-China Relations Amid Ongoing Discussions

Union Minister Piyush Goyal Defends PM Modi’s Ganesh Puja Visit Amid Opposition Criticism

PM Modi Celebrates Milestones and Future Potential at Asia Pacific Civil Aviation Conference


BJP Leader Ravi Shankar Prasad Accuses Rahul Gandhi of Insulting India During Foreign Visits

Defence Minister Rajnath Singh Highlights Surge in Tourism in Border Areas

Union Minister Shivraj Singh Chouhan Submits Flood Damage Report to Amit Shah


BJP Criticizes Rahul Gandhi's Views on Regional Differences and Constitution

Union Home Minister Amit Shah Launches Nationwide Cybercrime Awareness Campaign

BJP Condemns Rahul Gandhi's Statements on Sikhs, Accuses Congress of Spreading Division

Delhi Celebrates First Foundation Day of Indian Cybercrime Coordination Centre


Union Minister Kiren Rijiju Criticizes Rahul Gandhi for International Criticism of India

Union Minister Giriraj Singh Criticizes Arvind Kejriwal and Congress over Corruption Allegations


BJP MP Bansuri Swaraj Urges Public to Join Party to Support PM Modi’s Vision for a Stronger India


Smriti Irani Criticizes AAP, Highlights BJP’s Commitment to the Poor During Delhi Membership Drive

BJP MP Criticizes Omar Abdullah's Remarks on Afzal Guru, Calls for Supreme Court Action

Union Minister Piyush Goyal Highlights Enhanced India-Singapore Relations and Global Trade Concerns

BJP MP Sambit Patra Raises Questions on RG Kar Medical College Rape-Murder Incident


BJP Councillor Raja Iqbal Singh Claims Victory Amidst MCD's Inaction

Union Minister Giriraj Singh Champions Indian Textile Industry and BharatTech Initiative

Union Minister Nitin Gadkari Advocates for Stainless Steel in Coastal Bridge Construction

BJP Confident of Victory in MCD Standing Committee Elections, Says MP Yogender Chandolia

Prime Minister Narendra Modi Congratulates Paris Paralympic Games Medalists

BJP Leader Shehzad Poonawalla Criticizes Mamata Banerjee Following CBI Arrest

Union Minister Shivraj Singh Chouhan Criticizes West Bengal's Anti-Rape Bill

PM Modi Inaugurates New Vande Bharat Trains, Boosts India’s Railway Modernization Efforts

PM Modi Celebrates Supreme Court’s Legacy and Its Role in Upholding Constitutional Values

PM Modi Stresses Modernization of District Judiciary to Achieve a Viksit Bharat

PM Modi Hails Vadhvan Port Project Ahead of Maharashtra Visit

BJP's Shehzad Poonawalla Criticizes Mamata Banerjee Over RG Kar Medical College Rape-Murder Case

EAM Dr. S. Jaishankar Highlights NEP 2020’s Role in Strengthening India-UK Education Ties

Hungary's Balázs Orbán Praises EAM Dr. S. Jaishankar’s Role in Global Diplomacy

BJP’s Central Election Committee Convenes for Haryana Assembly Polls



Railways Announces 45,000 Job Vacancies in First Half of 2024, Says Union Minister Ashwini Vaishnaw

India and Chile Strengthen Ties at Second Joint Commission Meeting


Boxer and BJP Leader Vijender Singh Advocates for Sports and Supports Vinesh Phogat

Delhi BJP President Criticizes AAP Over Corruption Allegations Amid CBI Probe

BJP Leader Tarun Chugh Criticizes Congress-National Conference Alliance Ahead of J&K Assembly Polls

Union Sports Minister Encourages Nationwide Participation on National Sports Day

BJP Criticizes Congress-National Conference Alliance for J&K Assembly Polls

BJP Leader Shehzad Poonawalla Criticizes Mamata Banerjee's Government Over Handling of Sandeep Ghosh

ISRO Chairman S Somanath Unveils Vision for Space Exploration on First National Space Day

Union Health Minister JP Nadda Launches National Medical Register Under National Medical Commission

Union Minister Hardeep Singh Puri Praises PM Modi’s Diplomatic Mission to Ukraine

BJP Leader Smriti Irani Criticizes Congress-National Conference Alliance, Questions Their Agenda

PM Modi and Polish PM Tusk Elevate Ties to Strategic Partnership During Warsaw Talks

Union Minister Ramdas Athawale Criticizes Bharat Bandh and Congress Stance on SC Reservations


BJP MP Saumitra Khan Criticizes Kolkata Police and Mamata Banerjee Over RG Kar Medical College Case

BJP Leader Shehzad Poonawalla Condemns Rahul Gandhi's Response to RG Kar Medical College Case


Union Finance Minister Nirmala Sitharaman Reviews Performance of Public Sector Banks

BJP Leader Shehzad Poonawalla Calls for Mamata Banerjee’s Resignation Amid Controversy

Students Celebrate Raksha Bandhan with Prime Minister Modi: A Heartwarming Tradition


BJP MP Aparajita Sarangi Defends Party's Stance on Women's Rights Amid Controversy

BJP MP Sambit Patra Questions Congress Leaders Over Alleged Double Standards in Corruption Cases




BJP MP Sudhanshu Trivedi Criticizes Mamata Banerjee's Latest Statement as Vote Bank Politics

BJP MP Sudhanshu Trivedi Criticizes Mamata Banerjee Over Delay in CBI Case Transfer

Sumit Antil, 2024 Paralympic Flag Bearer, Eyes Success with 25+ Medal Target

India-Japan 2+2 Foreign-Defence Ministers Meeting Scheduled for August 20 in Delhi

BJP Confident of Victory in Upcoming Jammu and Kashmir Assembly Elections
Former BJP Spokesperson Nupur Sharma Joins Protest Against Atrocities on Minorities in Bangladesh

Prime Minister Modi Unveils Vision for India’s Future in 78th Independence Day Address

Modi Hoists Tricolour at Red Fort to Mark India’s 78th Independence Day

Union Sports Minister Mansukh Mandaviya Felicitates Olympic Bronze Medalist Aman Sehrawat in Delhi

BJP Leader Shehzad Poonawalla Criticizes Delhi Government’s Independence Day Arrangements

EAM Dr. S. Jaishankar Engages with German Bundestag MPs in Delhi, Highlights Strong Bilateral Ties

Prime Minister Narendra Modi to Lead 78th Independence Day Celebrations from Red Fort

BJP MP Bansuri Swaraj Criticizes INDI Alliance, Cites Recent Incidents of Violence and Silence

Union Minister Nitin Gadkari Reviews Progress of 51 National Highway Projects in Odisha


BJP MP Anil Baluni Announces Progress on Road Construction in Garhwal

BJP Leader Shazia Ilmi Welcomes CBI Investigation into RG Kar Medical College Rape-Murder Case

EAM Dr. S. Jaishankar Hoists Tricolour at Residence for 'Har Ghar Tiranga' Campaign

Delhi BJP MP Criticizes West Bengal Government's Handling of RG Kar Medical College Incident


Prime Minister Narendra Modi Conducts Aerial Survey of Landslide-Affected Areas in Wayanad, Kerala

Union Minister Ashwini Vaishnaw Unveils Major Initiative to Boost Farmers' Income

BJP MP Bansuri Swaraj Leads Cleanliness Drive, Aims to Transform Area into a Park

BJP Leader Gaurav Bhatia Criticizes Congress Over Karnataka Farmers' Crisis

BJP Leader Gaurav Bhatia Accuses Congress of Bias in MUDA Scam

Lok Sabha Speaker Om Birla Addresses Orientation Program for New MPs


Union Minister JP Nadda Congratulates Indian Hockey Team for Bronze Medal at Paris Olympics

BJP MP Dinesh Sharma Criticizes Opposition Stance on Waqf (Amendment) Bill, 2024

BJP President Virendraa Sachdeva Responds to Sisodia's Bail: "Bail Doesn't Mean Innocence"

Delhi: Private Complaint Filed Against Karnataka CM Over MUDA 'Scam'

BJP MP Manoj Tiwari Accuses Opposition of Provoking Muslim Community Over Waqf (Amendment) Bill

BJP MP Arun Govil Defends Waqf (Amendment) Bill, Rejects Religious Motives

MEA Confirms High-Level Talks on Bangladesh Between EAM Jaishankar and UK Foreign Secretary

MEA Updates on Indian Nationals in Bangladesh Amidst Ongoing Situation

India to Attend Swearing-In Ceremony of Bangladesh's New Interim Government

Uttarakhand CM Dhami Supports Waqf (Amendment) Bill, Defends Land Encroachment Drive

National Capital Region Planning Board Unveils Ambitious RRTS Plan for NCR-2032

BJP MP Basavaraj Bommai Responds to Salman Khurshid's 'Bangladesh' Remark: India’s Unity Undermined


Union Minister Piyush Goyal Urges Renewed Focus on BIMSTEC Free Trade Agreement Negotiations

BJP MP Anurag Thakur Slams Congress for Salman Khurshid’s 'Bangladesh' Remark

BJP MP Anurag Thakur Criticizes Opposition Leaders for Accepting Mangoes from Pakistan


BJD MP Sasmit Patra Responds to EAM Dr. Jaishankar's Statement on Bangladesh Crisis



BJP MP Bansuri Swaraj Commemorates Late Mother Sushma Swaraj with Tree Planting Campaign

Tamil Nadu BJP President Thanks EAM Dr. S. Jaishankar for Support to Fishermen Community

Tamil Nadu Fishermen Representatives Meet External Affairs Minister Dr. S. Jaishankar in Delhi

Delhi BJP President Calls for Kejriwal's Resignation Following High Court Ruling

Delhi: EAM S. Jaishankar Addresses Fishermen's Concerns After Meeting Tamil Nadu Delegation

Delhi High Court Dismisses Kejriwal's Plea; BJP MP Calls for His Resignation

Delhi: Former Minister Mukhtar Abbas Naqvi Supports Central Government's Potential Waqf Board Reform

Delhi: BJP Leader Advocates for Bill to Regulate Waqf Board Powers


BJP Leader Shazia Ilmi Demands Accountability Over Deaths at Asha Kiran Shelter Home

EAM Dr. S. Jaishankar Highlights Robust US-India Relationship Amid Global Changes

Union Minister Giriraj Singh Criticizes Rahul Gandhi as Leader of Opposition

BJP MP Dinesh Sharma Criticizes Rahul Gandhi's Recent Actions and Statements

Lok Sabha Speaker Om Birla Congratulates Swapnil Kusale on Olympic Bronze Medal

BJP MP Kangana Ranaut Criticizes Rahul Gandhi and Congress Party

Union Health Minister JP Nadda Advocates for Decentralized AIIMS Expansion

Union Minister Ashwini Vaishnaw Highlights Major Achievements and Future Plans for Indian Railways

Government Advances Coal Production with Strategic Mine Vesting Orders

Prime Minister Modi Hosts Japanese Delegation, Strengthens India-Japan Partnership

Prime Minister Modi Congratulates Swapnil Kusale on Historic Olympic Bronze Medal

BJP MP Manoj Tiwari Criticizes Delhi Government, Calls for Change

Vietnam Prime Minister Pham Minh Chinh Expresses Gratitude for Indian Condolences and Support

Prime Ministers Modi and Pham Minh Chinh Hold High-Level Delegation Meeting at Hyderabad House

Prime Minister Modi and Vietnam’s Pham Minh Chinh Forge New Agreements in Delhi

Basavaraj Bommai Questions Congress' Response to Anurag Thakur's Remarks on Caste Census

Ramdas Athawale Encourages Adhir Ranjan Chowdhury to Join NDA or RPI Amidst Congress Resignation

BJP MP Sudhanshu Trivedi Criticizes INDI Alliance Over Caste-Based Census Controversy

BJP MP Sudhanshu Trivedi Criticizes Rahul Gandhi Over Congress's Historical Stance on Caste Issues

Amit Shah Questions Kerala Government’s Response, Highlights India’s Early Warning System in Crisis


Rijiju Slams Congress Over Caste Remarks Following Anurag Thakur's Lok Sabha Statements

BJP Criticizes INDIA Bloc Rally at Jantar Mantar, Accuses Opposition of Misplaced Priorities

Union Minister Piyush Goyal Criticizes UPA Era as a 'Failed Experiment' at Conference

Union Minister Piyush Goyal Projects India’s Rise to Third Position in Global GDP Rankings

PM Modi Criticizes Previous Governments at 'Journey Towards Viksit Bharat' Conference

PM Modi Highlights Rs. 2 Lakh Crore PM Package at 'Journey Towards Viksit Bharat' Conference

PM Modi Calls for Investor-Friendly Charters at 'Journey Towards Viksit Bharat' Conference

Delhi BJP President Virendra Sachdeva Criticizes INDIA Alliance Amid Jantar Mantar Protest

BJP MP Sudhanshu Trivedi Questions Opposition's Stance on Caste Census

PM Modi Champions National Anti-Narcotics Helpline 'Manas' in Latest Mann Ki Baat Broadcast


Union Sports Minister Mansukh Mandaviya Highlights Khelo India’s Role in Supporting Athletes

BJP MP Praveen Khandelwal Criticizes AAP's Handling of Old Rajinder Nagar Incident

EAM S Jaishankar Highlights Synergy of QUAD and Bilateral Relations at Tokyo Meeting

Prime Minister Modi Chairs 9th Governing Council Meeting of NITI Aayog






Union Minister Pralhad Joshi Accuses Karnataka CM of Involvement in Scandals, Calls for CBI Probe




Union Minister JP Nadda Commemorates Kargil Vijay Diwas, Highlights Valor and Sacrifice


Union Minister Jitendra Singh Highlights Unity and Resolve on Kargil Vijay Diwas

Union Minister JP Nadda Honors Kargil War Heroes at 'Marshal Rally' in Delhi

President Droupadi Murmu Turns Educator: Engages Students at Dr. Rajendra Prasad Kendriya Vidyalaya

Union Minister Pralhad Joshi Alleges Scams Involving Karnataka CM: Demands CBI Probe

Former Karnataka CM Basavaraj Bommai Alleges Scam in Valmiki Board Funds, Opposition Protests

Defence Minister Rajnath Singh Pays Tribute to Late Nguyen Phu Trong at Vietnam Embassy

UK-India Technology Security Initiative Launched to Boost Strategic Partnership


Union Budget 2024 Emphasizes Job Creation and State Benefits, Asserts BJP MP Sukanta Majumdar


Assam CM Himanta Biswa Sarma Defends Union Budget 2024-25 Amid Opposition Criticism


BJP Leader Praveen Shankar Kapoor Files Defamation Case Against AAP's Atishi

Union Minister Shivraj Singh Chouhan Praises Budget for Transformative Impact on 'Viksit Bharat'

BJP Leader Rajeev Chandrasekhar Praises Budget as Stepping Stone to 'Viskit Bharat'


Union Minister Hardeep Singh Puri Highlights Fiscal Health and Development Initiatives in Budget

Union Minister Hardeep Singh Puri Criticizes Opposition, Defends Budget Allocations

BJP Leader Gaurav Bhatia Defends Government's Decision on RSS Participation for Government Employees

Rajasthan BJP President CP Joshi Meets Home Minister Amit Shah in Parliament

EAM Dr S Jaishankar Highlights Strengthened India-Egypt Relations at National Day Celebrations

BJP MP Ravi Shankar Prasad Criticizes Mamata Banerjee's Stance on Refugee Policy and CAA

PM Narendra Modi Inaugurates 46th World Heritage Committee Session on Guru Purnima

Prime Minister Narendra Modi to Inaugurate 46th Session of World Heritage Committee in New Delhi

BJP MP Anurag Thakur Accuses Rahul Gandhi and Congress of Hypocrisy Regarding Constitution

BJP MP Bansuri Swaraj Accuses AAP Leader Somnath Bharti of Spreading Lies Following Electoral Defeat

BJP Leader Shazia Ilmi Questions CM Kejriwal’s Health Regimen


Prime Minister Narendra Modi Felicitated by BJP National President JP Nadda

Haryana CM Nayab Singh Saini Meets PM Modi, Discusses Development and Future Plans

President Droupadi Murmu Confers Ati Vishisht Seva Medal on Lt Gen Vikas Lakhera

Union Home and Cooperation Minister Amit Shah to Visit Ranchi, Jharkhand

Union Home Minister Stresses 24x7 Operations and Upgradation of Multi-Agency Centre (MAC)

Union Home Minister Amit Shah Reviews Multi Agency Centre Operations

BJP Leader Dushyant Kumar Gautam Defends UP Police Directive Amid Kanwar Yatra Controversy

BJP Leader Rajeev Chandrasekhar Urges Swift Restoration of Microsoft Services Amid Outage

BJP Leader Shehzad Poonawalla Criticizes Congress Silence on Women's Issues

Union Home Minister Amit Shah Champions Efforts Towards a Drug-Free India

PM Modi Acknowledges BJP Workers' Efforts in Election Victory



BJP Leader Shehzad Poonawalla Criticizes Samajwadi Party's Remarks on UP Government

Chhattisgarh Deputy CM Announces Operation Against Naxals: 12 Naxalites Killed

Union Minister Ashwini Vaishnaw Urges Opposition to Choose Words Wisely

Amit Shah Chairs 7th Apex Level Narco-Coordination Center Meeting

NITI Aayog to Unveil Report on India's Electronics Sector and Global Value Chains

Union Minister Piyush Goyal Engages in Key Bilateral Talks at G7 Trade Ministers' Meeting in Italy




BJP Leader Shehzad Poonawalla Accuses INDI Alliance of Economic Mismanagement Across States

Halwa Ceremony Marks Commencement of Union Budget 2024 Preparation Process

BJP MLA Vijender Gupta Calls for Special Delhi Assembly Session Amidst Public Concerns

BJP MLA Ajay Mahawar Criticizes Delhi Government Over Electricity Rates and Governance

Himachal Pradesh CM Sukhvinder Singh Sukhu Highlights NHAI Meeting Agenda for Development Projects

Title: Union Minister Alleges Unlawful Acquisition of Land by Karnataka CM's Wife

Assam Governor Meets PM Modi, Discusses Flood Relief Efforts

Union Minister Nitin Gadkari and Rajasthan CM Bhajanlal Sharma Review National Highway Projects

BJP Leader Shehzad Poonawalla Accuses AAP of Sins and Propaganda

BJP MP Manoj Tiwari Warns Delhi Power Companies Amid Protests

BJP Leader Shehzad Poonawalla Alleges Manipulation in Arvind Kejriwal's Jail Term

BJP Accuses AAP of Political Maneuvering Ahead of Kejriwal's Court Appearances

Union Home Minister Amit Shah to Chair Apex Level Narco-Coordination Meeting in Delhi

Union Minister Jyotiraditya Scindia Charts Path to Make India a Global Communication Leader

India-Germany Foreign Office Consultations Foster Bilateral Cooperation

BJP Protests Alleged Electricity Billing Scam in Delhi, Demands Inquiry

BJP Leader Shazia Ilmi Criticizes Opposition's Response to Election Defeats

Union Minister Giriraj Singh Advocates Sports Participation and Funding Allocation

BJP MP Manoj Tiwari Encourages Indian Athletes Ahead of Paris Olympics Marathon

Amit Shah Emphasizes Employment and Connectivity in 'Vibrant Villages Programme' Review


Delhi BJP Chief Alleges Kejriwal's Declining Political Influence Amid Corruption Allegations

Supreme Court Grants Interim Bail to Delhi CM Arvind Kejriwal Amidst Corruption Allegations
BJP Organizes Protest Against Electricity Bill Hike; Delhi BJP Chief Detained

Haryana CM Nayab Singh Saini Welcomes Congress Defections to BJP Amidst Political Shifts



Delhi BJP Chief Raises Allegations Against Arvind Kejriwal Amid Legal Tensions

PM Modi Hosts BIMSTEC Foreign Ministers, Advocates Regional Cooperation

Supreme Court Grants Interim Bail to Delhi CM Arvind Kejriwal Amid Controversy

Prime Minister Narendra Modi Engages with Economists in Pre-Union Budget Meeting

BJP MP Manoj Tiwari Urges Action as Munak Canal Barrage Breaks, Floods North Delhi Residential Areas

BJP MP Yogender Chandolia Takes Action Amidst Water Crisis in North West Delhi

BJP MP Manoj Tiwari Alleges Arvind Kejriwal's Involvement in Delhi Liquor Policy Case

EAM Dr S Jaishankar Highlights BIMSTEC's Strategic Imperatives at 2nd Foreign Ministers' Retreat

EAM Dr S Jaishankar Calls for Renewed Commitment at 2nd BIMSTEC Foreign Ministers' Retreat

EAM Dr S Jaishankar Hosts 2nd BIMSTEC Foreign Ministers’ Retreat in New Delhi

Uttarakhand CM Pushkar Singh Dhami Commended for Uniform Civil Code Implementation

Uttarakhand Chief Minister Pushkar Singh Dhami Celebrates Passage of Uniform Civil Code Bill

President Droupadi Murmu Launches Prestigious Football Tournaments at Rashtrapati Bhavan


Former AAP Minister Raaj Kumar Anand Joins BJP in Delhi

Piyush Goyal Hails PM Modi's Historic Recognition with Russia's Highest Civilian Honour


PM Modi Hails India’s Remarkable Progress and Aspirations Towards Development

PM Modi Unveils Ambitious Vision for India's Future in Third Term Address

Olympic Gold Medalist Abhinav Bindra Meets Union Home Minister Amit Shah in Delhi

Delhi BJP Chief Alleges Corruption and Mismanagement in Education System

BJP's L Murugan Criticizes DMK Government Over Atrocities Against Dalits in Tamil Nadu

BJP Leader VP Duraisamy Raises Concerns Over Law and Order in Delhi

Delhi BJP Chief Alleges Massive Mismanagement in Delhi's Education System

Prime Minister Narendra Modi Receives Grand Welcome in Moscow, Russia for Bilateral Summit

Delhi BJP President Virendraa Sachdeva Vows Change, Criticizes Kejriwal Government

Delhi BJP President Virendraa Sachdeva Alleges Corruption, Calls for Investigation into AAP Minister

BJP MP Manoj Tiwari Challenges AAP's Saurabh Bharadwaj on Investigation Stance

BJP Leader Arvinder Singh Lovely Calls for Policy Clarity Amid Teacher Transfer Controversy in Delhi
BJP MP Kamaljeet Sehrawat Raises Concerns Over Late-Night Teacher Transfers in Delhi

Delhi BJP President Virendraa Sachdeva Launches Ambitious 'Vijay Sankalp' Campaign for 2024 and 2025

PM Modi Embarks on Diplomatic Visit to Russia and Austria, Aims to Strengthen Bilateral Ties

BJP Leader Radhika Khera Criticizes Rahul Gandhi's Leadership Approach

BJP MP Manoj Tiwari Responds to Lalu Yadav’s Statement
Delhi BJP President Virendraa Sachdeva: Dr. Syama Prasad Mookerjee’s Enduring Ideals

Delhi BJP Leaders Pay Tribute to Dr. Syama Prasad Mookerjee on His Birth Anniversary

BJP MP Bansuri Swaraj Commemorates Dr Syama Prasad Mookerjee's Birth Anniversary in Delhi

Delhi BJP President Accuses AAP Government Amid Water Crisis and Flood Concerns

BJP Leader Devinder Kumar Maniyal Discusses J&K Strategy with Union Home Minister Amit Shah

PM Modi Encourages Indian Olympic Contingent, Promises Support at Red Fort Event

Andhra Pradesh CM N Chandrababu Naidu Meets BJP Chief and Union Minister JP Nadda in Delhi

Andhra Pradesh CM N Chandrababu Naidu Meets Defence Minister Rajnath Singh in Delhi

Title: PM Modi Motivates Paris 2024 Olympics Contingent, Discusses 2036 Hosting Bid

Railway Minister Ashwini Vaishnaw Announces Expansion Plans for Indian Railways

EAM Dr S Jaishankar Discusses Bilateral Meeting with Chinese Counterpart Wang Yi

EAM Dr S Jaishankar Provides Insights on Recent Bilateral Engagements


EAM Dr S Jaishankar Affirms Importance of India-Russia Annual Summit Ahead of PM Modi's Visit

India-Russia Relations Strengthened, PM Modi to Visit Moscow for Annual Summit

India and Australia Strengthen Defence Cooperation in Indo-Pacific Dialogue

PM Modi Meets T20 World Cup-Winning Team: A Memorable Celebration

PM Modi Accuses Congress of Prioritizing 'Save the Corrupt Andolan' Over National Development

BJP Nominates Bansuri Swaraj, Daughter of Late Sushma Swaraj, to Contest New Delhi Seat


Tragedy Strikes at Delhi's Hathras Stampede: BJP MP Demands Accountability

PM Modi Condemns Paper Leak, Vows Stringent Measures for Culprits


BJP Leader Shehzad Poonawalla Accuses Rahul Gandhi of Misinformation on Hinduism, Sparks Controversy

BJP MP Jagadish Shettar Raises Concerns Over Karnataka Government's Stability and Alleges Corruption

BJP MP Bansuri Swaraj Files Notice Against Rahul Gandhi for Alleged Misinformation in Lok Sabha

BJP MP Ravi Shankar Prasad Slams Opposition for Disrupting PM Modi's Speech in Lok Sabha


Prime Minister Narendra Modi Reminisces on India's Past Scams, Promises a Corruption-Free Future

PM Modi Accuses Rahul Gandhi of Starting Drama to Gain Sympathy

BJYM Protest Against Rahul Gandhi's Remarks Draws Attention in Delhi

Delhi BJP President Condemns Rahul Gandhi's Remarks, Calls Them Insulting to Hindus

Chirag Paswan Highlights Concerns on Parliamentary Traditions at NDA Meeting

HM Amit Shah Clarifies Implementation of New Criminal Laws with First Cases in Gwalior and Delhi


Union Minister Chirag Paswan Addresses Key Issues on NEET and Criminal Laws

Union Minister Pabitra Margherita Hails 'Mann ki Baat' as Global Phenomenon

Union Home Minister Amit Shah Inaugurates Sardar Patel Sahkar Bhavan at KDCC Bank's 76th AGM

Delhi BJP MP Bansuri Swaraj Praises PM Modi's 'Mann Ki Baat' Call for Tree Planting

Delhi CM Arvind Kejriwal Sent to Judicial Custody Amidst CBI Case, BJP President Reacts



Odisha CM Mohan Charan Majhi and Deputy CMs Meet BJP President JP Nadda in Delhi

BJP MP Manoj Tiwari Criticizes AAP Government Over Drain Cleaning in Delhi

BJP Delhi President Criticizes CM Arvind Kejriwal, Calls for Accountability Amidst Legal Proceedings



BJP MP Manoj Tiwari Criticizes Delhi Government Over Waterlogging Issues

BJP MP Accuses AAP Government of Neglect Amid Waterlogging Woes in Delhi


External Affairs Minister Dr. S. Jaishankar Discusses India-Malaysia Enhanced Strategic Partnership

Railways Minister Ashwini Vaishnaw Collaborates with Odisha CM for Rath Yatra Preparations

Union Minister Dr. Jitendra Singh Launches Bhuvan Panchayat Portal for Rural Land Mapping


BJP MP Tapir Gao Urges Reflection on India's Emergency Era for Future Democratic Vigilance

BJP MP Ravi Shankar Prasad Criticizes Congress Over Emergency, Questions Intentions

Sam Pitroda Re-appointed as Chairman of Indian Overseas Congress Amidst BJP Criticism

BJP Condemns Samajwadi Party MP RK Chaudhary's Remarks on Sengol, Asserts Cultural Insensitivity

President Droupadi Murmu Advocates Modernization of Armed Forces and Defence Reforms

President Droupadi Murmu Commends Indian Athletes Ahead of Paris Olympics, Eyes 2036 Hosting Bid

President Droupadi Murmu Highlights India's Achievements and Democratic Values in Parliament Address

BJP Leader CR Kesavan Condemns Samajwadi Party MP RK Chaudhary's Controversial Remarks

President Droupadi Murmu Delivers Inspiring Address to Parliament, Outlines India's Path Forward

BJP MP Trivendra Singh Rawat Praises President's Address, Criticizes Opposition's Rhetoric

Union Minister Ramdas Athawale Commends President's Address, Dismisses Opposition Criticism

Punjab Governor Meets Prime Minister Narendra Modi in Delhi

Speaker Om Birla Addresses Lok Sabha on 1975 Emergency: Highlights and Reflections

BJP MP Kangana Ranaut Criticizes Opposition at NDA Protest on 50th Anniversary of Emergency

Union Minister Chirag Paswan Condemns Emergency on its 50th Anniversary at NDA Protest

BJP MP Sambit Patra Remarks on NDA Protest on 50th Anniversary of Emergency

BJP MP Arun Govil Expresses Confidence Ahead of Lok Sabha Speaker Election

External Affairs Minister Dr S Jaishankar Meets Kuwaiti Ambassador, Discusses Bilateral Relations

BJP MP Hema Malini Endorses Om Birla for Re-election as Lok Sabha Speaker

BJP MP Suresh Gopi Comments on Kerala Assembly's Name Change Resolution and Emergency

Union Minister Ramdas Athawale Confident of NDA Victory in Lok Sabha Speaker Elections



BJP MP Sanjay Jaiswal Urges Opposition for Unanimous Election of Speaker

PM Modi and Goa CM Dr. Pramod Sawant Strategize Goa's Developmental Roadmap


Union Ministers Unveil Ambitious Plans in Launch of 4th Tranche Auction for Critical Minerals

Prime Minister Narendra Modi and Ministers Sworn In as 18th Lok Sabha Convenes


BJP MP B. Y. Raghavendra Defends Constitution Amid INDIA Bloc Protests in Delhi

BJP MP Manoj Tiwari Calls for Responsible Opposition Role in Parliament


Union Minister Athawale Criticizes Opposition's Protest with Constitution in Parliament Premises


Delhi BJP President Raises Concerns Over Removal of Vocational Teachers; Urges LG's Intervention

BJP MP Manoj Tiwari Raises Vocational Teachers' Issue with Delhi LG; Alleges Conspiracy

BJP MP Bansuri Swaraj Raises Concerns Over Removal of Part-Time Vocational Teachers in Delhi

Union Health Minister JP Nadda Meets President and Vice President

Union Finance Minister Nirmala Sitharaman Chairs 53rd GST Council Meeting in Delhi

Union Minister Giriraj Singh Emphasizes Yoga's Global Impact on International Day of Yoga

Defence Minister Rajnath Singh Offers Prayers at Banke Bihari Temple in Mathura

PM Modi Celebrates International Day of Yoga in Srinagar, Jammu & Kashmir

Arvind Kejriwal’s Bail Hearing and BJP MP Manoj Tiwari’s Statement

BJP MP Manoj Tiwari Addresses Delhi’s Water Crisis

BJP Leader Alleges DMK Involvement in Kallakurichi Hooch Tragedy

Union Education Minister Dharmendra Pradhan Assures Action Amid NEET Exam Investigation


Union Health Minister JP Nadda Emphasizes Citizen-Friendly Approach During Visit to Health Ministry

Union Health Minister JP Nadda Visits Various Offices of Nirman Bhawan in Delhi

Delhi Water Crisis: BJP Accuses AAP of Corruption and Water Tanker Mafia Involvement

EAM Dr S Jaishankar Meets Sri Lankan President Ranil Wickremesinghe in Colombo

BJP MP Accuses Kejriwal Government of Neglecting Delhi’s Water Crisis

US Congressional Delegation Meets Prime Minister Narendra Modi in New Delhi

Vadhavan Port Project: Economic Stimulus and Strategic Advancement

Union Cabinet Approves Development of Major Port at Vadhavan, Maharashtra

BJP Leader Accuses AAP of Fueling Delhi Water Crisis, Alleges Corruption

Cabinet Approves New Scheme to Enhance Forensic Infrastructure Across India

BJP Stages Protest Against AAP Over Water Crisis in Delhi

External Affairs Minister Dr. S. Jaishankar to Visit Sri Lanka for High-Level Talks

Indian Navy Chief Meets PM Modi, Assures Readiness and Nation-Building Initiatives

PM Modi Announces Return of 'Mann Ki Baat' Radio Broadcast After Election Hiatus

Delhi Water Management Debate: BJP President Criticizes AAP Minister Saurabh Bhardwaj's Claims

Delhi Water Crisis: BJP MP Ramvir Singh Bidhuri Criticizes CM Kejriwal's Promises


Delhi BJP President Accuses AAP Government of Creating Water Crisis, Alleges Mismanagement

BJP Leader Anil Antony Criticizes Congress Over Controversial Post Mocking PM-Pope Meeting


BJP MP Praveen Khandelwal Challenges Delhi Government Over Persistent Water Crisis

BJP MP Bansuri Swaraj Criticizes Delhi Government's Management of Water Crisis

Union Home Minister Amit Shah Chairs High-Level Meeting Amid Escalating Tensions in Manipur

BJP Leader Shehzad Poonawalla Criticizes AAP and Congress Over Delhi Water Crisis


India’s Cultural and Tourism Sector: A Strategic Pillar for Economic Advancement

Delhi Water Crisis: A Closer Look at Union Minister Gajendra Singh Shekhawat's Comments

PM Narendra Modi Calls for Responsible AI at G7 Outreach Session

BJP Condemns Congress Leader Mallikarjun Kharge's Criticism of NDA Government

President Droupadi Murmu Celebrates ‘Raja Parb’ at Rashtrapati Bhavan

BJP MPs Receive Mortal Remains of Kuwait Fire Victims at Palam Airport

BJP Alleges Water Wastage Amid Acute Shortage in Delhi

G7 Summit: India’s Prime Minister Narendra Modi in Global Spotlight

Defence Minister Rajnath Singh Heads to Visakhapatnam to Review Indian Navy’s Eastern Fleet

BJP Leader Accuses Congress of Allying with Radical Islamist Groups

Delhi Water Crisis: BJP MP Bansuri Swaraj Accuses AAP of Negligence

Prime Minister Narendra Modi Departs for Italy to Attend G7 Outreach Summit

G Kishan Reddy Assumes Office as Union Minister of Coal and Mines

Defence Minister Rajnath Singh: A Vision for a Self-Reliant India

Delhi’s Water Crisis: A Tug of War Between Politics and Basic Needs

India’s Power Sector Shines: Union Minister Announces Sufficient Power Supply Across the Country

BJP’s Sukanta Majumdar Challenges Rahul Gandhi’s Dual Seat Victory

PM Narendra Modi Reviews Kuwait Fire Incident Upon Return from Andhra Pradesh and Odisha

Indian Government Responds to Kuwait Fire Incident

BJP’s Triumph in Ayodhya and Tragedy in Kuwait: A Dual Perspective

PM Modi Extends Condolences and Announces Relief for Victims of Kuwait Fire Tragedy

Delhi Water Crisis: BJP MP Manoj Tiwari Criticizes AAP’s Atishi

Delhi BJP Chief Raises Alarm Over Water Shortage and ‘Tanker Mafia’

Delhi Water Crisis: BJP Leader Accuses AAP of Involvement in Water Scam

Nitin Gadkari Assumes Charge as Minister of Road Transport and Highways

BJP MLA Mohan Charan Majhi to be the New Chief Minister of Odisha

Delhi Water Crisis: BJP Accuses Government of Negligence



Railways Minister Ashwini Vaishnaw Lauds PM Modi’s Contributions to Indian Railways

India’s Growing Influence on the Global Stage: A Look at Modi 3.0’s Foreign Policy

Bhupender Yadav Assumes Office as Minister of Environment, Forest and Climate Change

Ashwini Vaishnaw Takes Charge as I&B Minister, Expresses Gratitude to PM Modi

New Leadership in the Ministry of Textiles

Hardeep Singh Puri Set to Retain Ministry of Petroleum and Natural Gas

Ashwini Vaishnaw: A Triple Portfolio Ministerial Appointment

Satish Chandra Dubey Appointed as MoS in Ministry of Coal and Mines

Government Prioritizes Farmer Welfare: Union Minister Shivraj Singh Chouhan

Delhi BJP President Accuses AAP Minister of Misleading the Public

Uttar Pradesh CM Yogi Adityanath Meets Union Ministers in Delhi


Union Minister Dr S Jaishankar Meets Bangladesh Prime Minister Sheikh Hasina in Delhi

Nirmala Sitharaman Sworn in as Union Cabinet Minister in Modi Government

JP Nadda Joins Union Cabinet in Modi-led NDA Government

Nitin Gadkari Sworn in as Union Cabinet Minister in Modi-led NDA Government

Amit Shah Sworn in as Union Cabinet Minister in PM Narendra Modi’s NDA Government

Rajnath Singh Sworn in as Cabinet Minister in Prime Minister Narendra Modi’s Cabinet

Narendra Modi Sworn in as Prime Minister for Third Consecutive Term

Historic Day in India: Narendra Modi Sworn in as Prime Minister for the Third Time

Narendra Modi Set to Take Oath as Prime Minister for the Third Consecutive Term

Bangladesh PM Sheikh Hasina Arrives in India for PM Modi’s Swearing-In Ceremony

Manoj Tiwari Foresees Positive Changes as Narendra Modi Prepares for Third Term as Prime Minister

BJP MP Bhola Singh Arrives in Delhi, Confirms Narendra Modi’s Third Term as Prime Minister

Delhi BJP President Virendraa Sachdeva Responds to Alliance Partners’ Support for PM Modi’s Vision

Narendra Modi Appointed as Prime Minister of India by President Droupadi Murmu

Opposition Dismisses Modi's Assertion on EVMs, Calls for Electoral Reforms



BJP President JP Nadda Celebrates NDA's Electoral Triumphs and India's Transformation

Bhajanlal Sharma, Rajasthan CM, Foresees Continued Progress Under Modi’s Third Term

BJP MP Rajnath Singh Expresses Confidence in PM Modi’s Leadership

Nitin Gadkari Backs Narendra Modi as Leader of Lok Sabha, BJP, and NDA Parliamentary Party

Amit Shah Endorses Narendra Modi as Leader of Lok Sabha and BJP: A Continuation of BJP's Dominance

HD Kumaraswamy Supports Proposal to Name Narendra Modi as Leader of NDA Parliamentary Party

NDA Parliamentary Party Meeting: A Boost of Confidence for Andhra Pradesh

Nitish Kumar Endorses Modi’s Leadership at NDA Parliamentary Party Meeting

Naidu Lauds Modi’s Leadership at NDA Parliamentary Party Meeting

Rajasthan CM Bhajanlal Sharma in Delhi for NDA Meeting

India’s Market Cap Crosses $5 Trillion: A Milestone Achievement

India’s Market Capitalization Skyrockets under BJP Leadership, says Piyush Goyal

Uttar Pradesh CM Yogi Adityanath Heads to Delhi for Crucial BJP Meeting

BJP’s B.Y. Raghavendra Hails Historic Victory in Shimoga Lok Sabha Seat

BJP Leader Pradeep Kumar Singh Envisions a ‘Viksit’ India

Athawale Confident of BJP’s Comeback in Upcoming UP Assembly Elections

BJP MP Radha Mohan Singh Expresses Confidence in Modi’s Leadership and the JD(U)-BJP Alliance

Supreme Court Verdict on Water Dispute Reveals AAP’s “Dirty Politics”, Claims Delhi BJP President

Sri Lankan President to Attend PM Narendra Modi’s Swearing-In Ceremony

BJP Leader Arjun Munda Confident in Party's Victory, Foresees Third Term for PM Modi

Prime Minister Narendra Modi Launches ‘Ek Ped Maa Ke Naam’ Campaign on World Environment Day

Vice-President Jagdeep Dhankhar Hosts Prime Minister Narendra Modi in Delhi

Union Cabinet Unanimously Supports PM Modi’s Leadership

Crucial NDA Meeting Commences at Prime Minister’s Residence

New Government Formation in India: BJP-led NDA to Take Charge

BJP’s Ramvir Singh Bidhuri Foresees a Two-Thirds Victory in Delhi Assembly Elections

BJP Sweeps Delhi: Wins All Seven Lok Sabha Seats

BJP’s Bansuri Swaraj Triumphs in New Delhi: A Soldier of PM Modi

BJP’s Harsh Malhotra Triumphs in East Delhi, Credits Victory to People’s Verdict

PM Modi Acknowledges Election Commission for Successfully Conducting the World’s Largest Election

BJP’s Bansuri Swaraj Triumphs in New Delhi Constituency

PM Modi’s Vision for a ‘Viksit Bharat’

BJP’s Manoj Tiwari Triumphs in North-East Delhi: A Promise of Change

BJP Sweeps Lok Sabha Elections 2024: A Day of Celebration at Party HQ in Delhi

BJP’s Manoj Tiwari Expresses Confidence in Party’s Victory in North East Delhi

BJP’s North West Delhi Candidate Yogendra Chandolia Confident of Victory

Jubilations in Delhi as BJP Takes the Lead in Election Count

BJP's Sudhanshu Trivedi Condemns Opposition's Alleged Attack on Democracy

BJP's Sudhanshu Trivedi Suggests Opposition Facing Inevitable Electoral Defeat

Remembering Sushil Kumar Modi: A Tribute to the Late Bihar Deputy CM

BJP’s Victory: A Mandate for PM Modi, says Shaina NC

Pre-Election Meeting Between Bihar CM and PM Modi Sparks Interest

Bihar’s Chief Minister Meets Prime Minister Ahead of 2024 Lok Sabha Election Results

BJP Sweeps Northeast States, Congress Must Introspect, Says Shehzad Poonawalla

Market Confidence Soars as Sensex and Nifty Surge Ahead of Election Results

BJP Leaders Meet at Party Chief JP Nadda’s Residence, Discuss Counting Day Preparations

Delhi BJP Chief, Virendraa Sachdeva, Expresses Confidence in Exit Polls

PM Modi’s Unwavering Commitment to Service: Union Minister Hardeep Singh Puri

Delhi BJP Chief Slams Arvind Kejriwal Over Exit Poll Comments

Exit Polls 2024: BJP-led NDA Predicted to Win Big

North East Delhi Exit Polls 2024: BJP’s Manoj Tiwari Takes the Lead, Predicts Axis My India

Smooth Voting Process: PM Modi Thanks Election Commission for Meticulous Planning

Beyond Politics: PM Modi's Spiritual Trip Echoes Across India

BJP-Led NDA Predicted to Win: Exit Polls 2024

Union Minister Hardeep Singh Puri Questions Congress’s Lok Sabha Poll Predictions


BJP Leader Shaina NC Defends PM Modi's Meditation Amid Opposition Criticism
BJP Spokesperson Condemns Opposition's Criticism of Prime Minister's Meditation in Kanyakumari

Diplomatic Relations Strengthened: Rashtrapati Bhavan Welcomes New Ambassadors

ChatGPT Delhi LG Blasts Government Over Water Crisis: Alleges Irresponsibility and Mismanagement

Delhi Government’s Inefficiency to Blame for Water Shortage, Says LG VK Saxena


Delhi BJP President Accuses Arvind Kejriwal of Corruption Scandal

Delhi BJP Holds Demonstration March Over Water Crisis
Delhi Water Crisis: BJP Candidate Accuses AAP Government of Mismanagement


BJP Women Workers Protest Against AAP Over Water Crisis in Delhi

India’s Economic Surge: A Testament to Modinomics

Delhi Water Crisis: BJP Leader Accuses AAP Government of Promoting ‘Tanker Mafia’

DRDO Achieves Milestone with Successful Flight Test of RudraM-II Missile

AAP Government’s Inaction Amid Soaring Temperatures Draws Criticism from BJP President

BJP Leader Gaurav Bhatia Questions Congress’s Silence on 1962 China Invasion

Controversy Surrounds Punjab Minister Balkar Singh Amidst Allegations of Sexploitation

The Alleged Obscene Video of Punjab Minister Balkar Singh: A Call for Accountability and Justice

Political Tensions Rise: BJP’s Bansuri Swaraj and AAP’s Atishi Summoned in Defamation Case

Delhi Court Summons AAP’s Atishi in Defamation Case Filed by BJP Leader

Manoj Tiwari Challenges Congress: ‘No One Can Stop Me from Visiting Punjab’

Tribute to Vinayak Damodar Savarkar: A Beacon of Indian Freedom Struggle

Celebrating One Year of the New Parliament Building: A Milestone in India’s Democratic Journey

BJP Leader Shazia Ilmi Criticizes AAP Over Punjab Minister’s Controversy

The Unfolding Drama: AAP, Swati Maliwal, and the Alleged Assault

The Political Rhetoric: A Tale of ‘Mujra’, ‘Poison’, and ‘Hitler’s Death’


Delhi Elections: BJP Candidate Manoj Tiwari Criticizes Opponent Kanhaiya Kumar

BJP Candidate Ramvir Singh Bidhuri Casts Vote, Expresses Confidence in PM Modi’s Leadership

BJP Candidate Ramvir Singh Bidhuri Casts Vote in South Delhi

BJP Leader Kuljeet Singh Chahal Casts Vote in Delhi for Lok Sabha Elections 2024

Delhi Votes: BJP’s Manoj Tiwari Casts His Vote, Criticizes Opposition

Delhi CM Arvind Kejriwal: A Chief Minister Behind Bars

BJP Leader Shazia Ilmi’s Response to AIMIM Chief Asaduddin Owaisi’s Statement

India’s Role in an Uncertain World: A Perspective from External Affairs Minister S Jaishankar

Delhi BJP President Accuses Arvind Kejriwal of Corruption

BJP Spokesperson Jaiveer Shergill’s Statement on AAP’s Anti-Women Policies

Political Turmoil in India: The Allegations Against Bibhav Kumar

BJP Leader Shehzad Poonawalla Raises Concerns Over Swati Maliwal Assault Case

BJP's Shehzad Poonawalla Slams West Bengal Government Over OBC Certificate Cancellation

BJP's Praveen Khandelwal Confident of Victory in Chandni Chowk, Highlights High-profile Support

BJP's North West Delhi Candidate Asserts Confidence in Victory, Calls Upon High-profile Supporters




BJP National President JP Nadda Criticizes Opposition, Predicts Election Results

Central Government’s Development Work in Delhi: A Report

Delhi: BJP Leader Arvinder Singh Lovely Criticizes Coalition Governments

Delhi Water Crisis: BJP Leader Shazia Ilmi Accuses AAP of Mismanagement

BJP’s Confidence Soars in Delhi, Predicts Victory in All Seven Seats

BJP to Sweep Delhi Elections, Claims Goa CM Pramod Sawant

Political Tug-of-War: Rahul Gandhi Accuses PM Modi of Creating Two Indias, BJP Retorts

Defence Minister Rajnath Singh Recalls Emergency Era

BJP Leader Shivraj Singh Chouhan Criticizes Congress and Nehru, Praises Modi and Shah

BJP Leader Shivraj Singh Chouhan Questions Delhi CM Arvind Kejriwal’s Silence


BJP National President JP Nadda Accuses AAP of Having Ties with Anti-National Forces

Delhi Chief Minister Arvind Kejriwal’s Arrest Sparks Controversy

BJP Leader Shaina NC Criticizes Nana Patole’s Statement on ‘Bhagwa’ Clothes

India Rises to Second Position in Mobile Production, Says BJP National President JP Nadda

BJP MP Sudhanshu Trivedi Questions Delhi CM’s Residence Protocol

Delhi BJP President Accuses CM Kejriwal of Ignoring Misconduct at His Residence

Rajasthan CM Bhajan Lal Sharma Criticizes Delhi CM Arvind Kejriwal
Start-Up Boom in India: A Digital Revolution

BJP Candidate Manoj Tiwari’s Development-Based Campaign in North East Delhi

India Finalizes Long-Term Agreement on Chabahar Port: A New Era of Connectivity

Education in India: A Shift Towards Innovation and Entrepreneurship

India Mourns the Loss of Iranian President Dr. Seyed Ebrahim Raisi

PM Modi Criticizes Opposition at Delhi Rally

Union Minister Ashwini Vaishnaw Foresees Third Term for PM Modi

Delhi Political Drama: Kejriwal’s Challenge and BJP’s Counter

Delhi BJP President Accuses CM’s Residence of Harboring Criminals
Piyush Goyal Responds to AAP’s Allegations in Swati Maliwal Assault Case

BJP’s Nadda Refutes AAP’s Allegations, Calls Out Kejriwal’s Silence

BJP National President JP Nadda Questions Mamata Banerjee’s Intentions

Union Minister Ashwini Vaishnaw Rallies for BJP in Cuttack

Delhi Political Turmoil: BJP Leader Shehzad Poonawalla Criticizes AAP and Congress

South Delhi Rally: Uttarakhand CM Pushkar Singh Dhami Rallies for BJP Candidate Ramvir Singh Bidhuri

Assault Case of AAP Rajya Sabha MP Swati Maliwal: BJP Leader Shazia Ilmi Speaks Out

India’s Economic Pathway: A Glimpse into the Future

BJP’s “400 Paar” Goal: Aiming for Stability, Not Constitutional Change, Says Amit Shah

BJP’s Vision for a Secure India: Aiming for “400 Paar”

Union Finance Minister Comments on AAP MP Swati Maliwal ‘Assault’ Case

Union Minister Anurag Thakur Criticizes Delhi CM Arvind Kejriwal

Delhi BJP Mahila Morcha Stands in Solidarity with Swati Maliwal

BJP Leader Manoj Tiwari Accuses Arvind Kejriwal of Corruption

Indian Prime Minister Condemns Attack on Slovak Counterpart

Delhi BJP President Accuses AAP of Being Anti-Women

BJP Spokesperson Shehzad Poonawalla Criticizes Kejriwal and Congress Over Assault Incident

BJP Spokesperson Shehzad Poonawalla Questions AAP-Congress Alliance

BJP’s Manoj Tiwari Campaigns in South Delhi, Boosts Party Morale

Political Tensions Rise as Union HM Dismisses Delhi CM’s Claims

Union Home Minister Amit Shah’s Take on Maharashtra Politics

BJP Government Committed to Reclaim PoK, Says Union Home Minister Amit Shah

EAM Dr S Jaishankar Calls for Strict Action Against Misinformation on Armed Forces

Women BJP Workers Protest Near Delhi CM’s Residence Over Assault Case

Assam CM Himanta Biswa Sarma’s Bold Predictions for Future Indian Politics

Delhi BJP President Calls for Investigation into Vibhav Kumar’s Actions

BJP Spokesperson Shehzad Poonawalla Criticizes Rahul Gandhi’s Statement

BJP Leader Shazia Ilmi Questions AAP’s Silence on Serious Allegations

Uttarakhand CM Meets Union Finance Minister, Invites Her to Chardham Yatra 2024

BJYM President Nominates Vice President for Public Debate with Rahul Gandhi

Goyal’s Accusations Against Thackeray Stir Up Maharashtra Politics

PM Modi Mourns the Loss of Sushil Modi, a Stalwart of BJP in Bihar

Prime Minister Narendra Modi to Hold First Rally in Delhi Ahead of Lok Sabha Election

Delhi BJP Chief Criticizes Kejriwal’s Poll Promises

Delhi BJP Chief Criticizes Kejriwal’s Interim Bail, Calls it ‘Not a Clean Chit’

Arbitration: A Cornerstone of Modern Dispute Resolution, Says EAM Dr Jaishankar

Arbitration: A Key Facet of Make In India, Says EAM Dr Jaishankar

BJP Leader Manoj Tiwari’s Bold Predictions for Delhi Elections

BJP Leader Rajeev Chandrasekhar Criticizes Congress Leaders’ Statements

Union Minister Rajeev Chandrasekhar Accuses Congress of Apologizing for Pakistan-Based Terror

Union Minister Ashwini Vaishnaw Foresees a Third Term for PM Modi

Union Minister Ashwini Vaishnaw Rallies for BJP Candidate Anirban Ganguly in Kolkata

Population Shift in India: A Study by EAC-PM and Reactions

Delhi BJP President Accuses Congress of Weakening National Unity

Delhi BJP President Detained Following Protest Against Congress

DRDO Chairman Advocates for Sustainable and Green Infrastructure in Defence Systems

BJP Leader Accuses Congress of Insulting Adivasi Community

Controversial Remarks Spark Political Firestorm in India

Controversial Remarks by Sam Pitroda Stir Political Tensions in India

Union Minister Rajeev Chandrasekhar Condemns Sam Pitroda’s Controversial Remarks

Union Minister Rajeev Chandrasekhar Responds to Controversial Remarks

AI to Transform Lives, India to Become World’s Third-Largest Economy: EAM Dr S Jaishankar

Digitization in India: A Leap Towards Transparency and Efficiency

India’s Stance on Global Challenges: A New Era of Firmness and Fairness

Former Congress Leader Radhika Khera Joins BJP, Cites Injustice in Congress

PM Modi Accuses Congress and INDI Alliance of Anti-National Sentiments

PM Modi's Bold Vision: NDA's Aspiration for Andhra Pradesh

Union Minister Hardeep Singh Puri Welcomes New Members to the Party

Arvinder Singh Lovely Joins BJP, Cites Congress’s Fragmentation as Reason

BJP Candidate Anil K Antony Foresees Historic Performance in National Election

BJP Candidate Bansuri Swaraj Receives Warm Welcome in Ramesh Nagar

BJP Spokesperson Accuses Congress of Anti-Hindu Sentiment

Uttarakhand CM Pushkar Singh Dhami Rallies Support for BJP Candidate in South Delhi
Rahul Gandhi’s Candidature: A Foregone Conclusion?

Delhi BJP President Responds to Kejriwal’s Arrest

Rahul Gandhi’s Candidature from Raebareli Sparks Political Debate

Rahul Gandhi’s Candidature from Raebareli: BJP’s Shehzad Poonawalla Reacts

Prime Minister Narendra Modi Arrives in Kolkata, West Bengal for Public Meetings

PM Modi's Reservation Challenges: Catalyst for National Discourse on Social Equity


BJP Leader Rohan Gupta Accuses Congress of Spreading Lies on Reservation

Political Tensions Rise in Delhi Amid Controversial Statements

BJP Spokesperson Accuses Congress of Dividing Hindu Society

BJP’s Manoj Tiwari Confident of Record-Breaking Win in North East Delhi

BJP Spokesperson Gaurav Bhatia Criticizes Congress National President Mallikarjun Kharge

Rupali Ganguly and Ameya Joshi Join BJP, Meet Party National President JP Nadda

BJP to Sweep Delhi Elections, Predicts Virendraa Sachdeva

Defence Minister Rajnath Singh Rallies for BJP Candidate Manoj Tiwari in Delhi

Uttarakhand CM Pushkar Singh Dhami Rallies for BJP Candidate Harsh Malhotra in East Delhi

Union Minister Hardeep Singh Puri Addresses Reservation Issue

BJP Candidate Bansuri Swaraj Pledges Implementation of Ayushman Bharat in Delhi

BJP Candidate Rajeev Chandrasekhar Questions Congress’ Stand on Crime Against Women

Prime Minister Modi Unveils Ambitious Vision for India's Sporting Future and National Security

EAM Dr. S Jaishankar Engages with IAF’s No. 3 WASP Participants

BJP and Allies Cross 100 Seats, Aims for ‘400 Paar’, Says Union Home Minister Amit Shah

PM Modi Accuses Congress of Sponsoring Terrorism, Sparks Political Firestorm

Prime Minister Narendra Modi Warns Against Fake Videos in Satara Rally

BJP MP Brij Lal Accuses Congress and INDI Alliance of Being Anti-Constitutional

Delhi North West BJP Candidate Yogender Chandoliya’s Bold Statement on Arvind Kejriwal

Union Home Minister Amit Shah Criticizes INDI Alliance’s Rotational PM Proposal

PM Modi Advocates for Uniform Civil Code, Criticizes Congress for Communal Politics

India’s Untapped Potential: A Vision for the Future

Nation Mourns the Loss of MP V Sreenivasa Prasad, PM Modi Pays Tribute

Ujjwal Nikam: A Renowned Public Prosecutor Steps into Politics

BJP Leader Shaina NC Accuses Congress of Double Speak and Appeasement Politics

Sikh Community Members Join BJP in Delhi

Delhi High Court’s Remarks Stir Controversy: BJP MP Manoj Tiwari Criticizes CM Arvind Kejriwal

PM Modi’s Contributions to Punjab and Sikh Community Highlighted by BJP National President JP Nadda

Delhi High Court’s Remarks Stir Political Debate

Delhi BJP President Calls for Kejriwal’s Resignation


Youth of Porbandar Rally Behind Modi, Mandaviya: Union Minister Ashwini Vaishnaw

Manoj Tiwari, BJP Lok Sabha Candidate, Holds Roadshow in Yamuna Vihar, Delhi

PM Modi Criticizes RJD Rule in Munger, Bihar

Delhi Breathes a Sigh of Relief as Rain Showers Cool the City

Uttar Pradesh Deputy CM Accuses Congress and INDI Alliance of Robbing SC, ST, and OBC Rights

Prime Minister Narendra Modi Calls for ‘Viksit Bharat’ in Agra Rally

YouTuber Manish Kashyap Joins BJP, Pledges to Advocate for the Poor

YouTuber Manish Kashyap Joins BJP, Pledges to Strengthen Bihar

YouTuber Manish Kashyap Joins BJP in Delhi

Prime Minister Narendra Modi Addresses Vijay Sankalp Rally in Morena

BJP Leader Shehzad Poonawalla Criticizes INDI Alliance


PM Modi Lauds Growth of Coalition for Disaster Resilient Infrastructure at International Conference

PM Modi Advocates for Resilient Infrastructure Amid Rising Natural Disasters

PM Modi Advocates for Global Cooperation in Disaster Resilience Efforts

PM Narendra Modi Advocates for Shared Resilience and Support for Vulnerable Island States

PM Narendra Modi Lauds CDRI’s Focus on Global South and India’s G20 Presidency

Rainfall Brings Respite to Delhi, Alters Weather Dynamics

PM Modi’s Gratitude and Festive Spirit Reverberate in Rajasthan on Hanuman Jayanti

Amit Shah Accuses Congress of Appeasement Politics
Union Minister Hardeep Singh Puri Responds to Unopposed Election in Surat

Prime Minister Modi Extends Hanuman Jayanti Wishes, Invokes Spirit of Dedication and Devotion

BJP Leader Shaina NC Accuses Congress of Divisive Politics, Praises PM Modi’s Inclusive Approach

BJP National General Secretary Dushyant Kumar Gautam Criticizes Congress’s Ideology

BJP National Spokesperson Predicts Massive Victory in Upcoming Elections

Delhi BJP Chief Virendraa Sachdeva Visits Ghazipur Landfill as Fire Continues to Rage

Delhi Landfill Crisis: BJP Chief Calls for Investigation and Action

BJP Leader Calls Out Delhi Government Over Ghazipur Landfill Fire Crisis

Poonawalla Demands CBI Probe into Hubballi Incident, Slams Congress

PM Modi’s Vision for Women Empowerment in India

Tajinder Singh Bittu Joins BJP, Citing Deviation of Congress Party from Core Issues

PM Modi Expresses Concern Over Marathwada’s Agricultural Struggles, Criticizes MVA and Congress

PM Modi’s 2024 Election Strategy: A Developed and Self-Reliant India

Law and Order Breakdown in Karnataka: BJP Leader Shehzad Poonawalla Speaks Out

Tajinder Singh Bittu Shifts Allegiance to BJP

Union Minister Anurag Thakur Responds to Sachin Pilot’s Statement

BJP National President JP Nadda Calls for Active Participation in Lok Sabha Elections

Prime Minister Narendra Modi’s Commitment to Progress in Uttar Pradesh and India


PM Modi Applauds Amroha's Dholak Heritage, Embraces Cultural Diversity at Uttar Pradesh Rally

PM Modi Criticizes Opposition at Amroha Rally

Union Minister Anurag Thakur Calls on Youth to Vote in Upcoming Polls

Union Home Minister Amit Shah Urges Voters to Participate in First Phase of Elections

PM Modi's Personal Letters Cement Unity and Vision Ahead of 2024 Lok Sabha Polls

BJP Gears Up for 2024 Lok Sabha Election: A Comprehensive Overview

Digital Revolution Reaches Giu Village: PM Modi Lauds Connectivity Milestone

EAM Jaishankar Discusses Current Situation in Afghanistan with UN Assistance Mission Chief

Communal Tensions in West Bengal: BJP Leader Shaina NC Criticizes State Government

Unrest in West Bengal: BJP Leader Raises Concerns Over Safety

BJP Leader Shehzad Poonawalla Criticizes Congress on Kanker Naxal Encounter

PM Narendra Modi Emotionally Moved by Surya Tilak on Ram Lalla

PM Modi Announces Free Treatment for Elderly Under Ayushman Yojana

PM Narendra Modi's Rally in Nalbari, Assam: A Beacon of Transformation

Historic Celebration of Lord Ram's Birthday in Ayodhya

PM Modi Unveils Ambitious Housing Scheme in Nalbari Rally, Pledging Homes for Millions

Prime Minister Narendra Modi Charms Assam with Traditional Music at Nalbari Rally

Historic Ram Navami Celebration in Ayodhya Following Ram Temple's 'Pran Pratistha'

Nation Mourns the Loss of Squadron Leader Dalip Singh Majithia

Political Paradox: Congress’ Contradictory Alliances, Claims BJP Leader Shehzad Poonawalla

Prime Minister Modi Accuses RJD of Misrule, Labels it as 'Jungle Raj' in Bihar


Prime Minister Narendra Modi Advocates for Regional Aspirations and Healthy Competition Among States

PM Modi’s Rally in Tamil Nadu: A Beacon of Women Empowerment

PM Responds to Opposition’s Allegations

PM Modi’s Assurance at Palakkad Rally: BJP’s Sankalp Patra and Ayushman Bharat Scheme



Prime Minister Narendra Modi Dives into Virtual Reality Gaming with Top Indian Gamers

Prime Minister Narendra Modi Meets Top Indian Gamers in Delhi



“DMK Stands for Deceit, Malpractice, and Kleptomania,” Claims BJP Spokesperson Gaurav Bhatia

Delhi Political Tussle: AAP Alleges Conspiracy by BJP-led Central Government, BJP Retorts

PM Modi Addresses Public Rally in Udhampur, J&K

BJP Candidate Praveen Khandelwal Confident of Victory in Chandni Chowk

PM Modi's Karauli Speech: Pledges for Poverty Alleviation and Agricultural Growth

Karauli, Rajasthan: PM Modi's Rally Sparks Political Momentum Ahead of Crucial Elections

Union Minister Hardeep Singh Puri Counters RJD's Misa Bharti's Remarks, Highlights Alleged Scams


Former Congress Leader Rohan Gupta Criticizes INDIA Alliance Following BJP Joining

Former Congress Leader Rohan Gupta Joins BJP, Citing Disillusionment and Ideological Shift

Rohan Gupta Joins BJP, Criticizes AAP’s Contradictions

BJP Leader Shahnawaz Hussain Celebrates Eid-ul-Fitr at Parliament Street Mosque

PM Modi’s Vision for India: A Developed Nation by 2047

PM Modi Addresses Newsweek: India as a Global Economic Growth Engine

Delhi HC Declares Kejriwal’s Arrest Legal, AAP Faces Internal Strife

PM Modi’s Stirring Speech in Ramtek: A Critique of INDIA Alliance

PM Modi Rallies in Maharashtra: A Call to Uphold Hindu Dharma’s ‘Shakti’

BJP Leader Shehzad Poonawalla Criticizes Congress’s ‘Family First’ Mindset

PM Modi Criticizes Family Parties at Rally in Mettupalayam, Coimbatore

BJP Demands Resignation of Delhi CM Arvind Kejriwal Amidst Corruption Allegations

Delhi High Court Dismisses CM Arvind Kejriwal’s Plea, BJP Spokesperson Responds
BJP MP Manoj Tiwari Accuses AAP Leader Arvind Kejriwal of Criminal Activity

Delhi High Court Rejects Arvind Kejriwal’s Bail Plea



Union Minister Piyush Goyal Foresees India as ‘Vishwaguru’ in the New Year

BJP Leader Manoj Tiwari Criticizes Congress Manifesto, Raises Concerns Over Muslim Personal Law

PM Modi’s Rally in Bastar: A Commitment to the Welfare of the Poor

PM Modi’s Bastar Rally: A Commitment to the Underprivileged

PM Modi’s Rally in Bastar: A Testament to Chhattisgarh’s Support

Prime Minister Narendra Modi Addresses Public Rally in Bastar, Chhattisgarh

BJP Leader Criticizes Congress Party’s Manifesto

Delhi BJP President Calls for Investigation into Delhi Jal Board Scam

BJP National President JP Nadda Criticizes Congress’ Lok Sabha Polls Manifesto

Tiwari Confident of BJP’s Victory in UP, Cites Public Support for PM Modi

BJP Spokesperson Criticizes Karnataka Home Minister’s Statement

BJP Spokesperson Shehzad Poonawalla Criticizes TMC Government

PM Modi’s Remarks on Congress Manifesto: Resonance of Muslim League and Leftist Ideologies

PM Modi’s Address in Saharanpur: A Celebration of BJP’s Growth and Dedication to National Policy

PM Modi’s Rally in Saharanpur: A Decade of Transformation

PM Narendra Modi’s Rally in Saharanpur: A Vision for Inclusive Development

PM Narendra Modi’s Rally in Saharanpur: A Tribute to ‘Shakti’

Prime Minister Narendra Modi to Address Public Rally in Saharanpur, Uttar Pradesh

PM Modi’s Churu Visit: A Declaration of India’s New Stance Against Terrorism

PM Modi in Churu: A Renewed Commitment to India’s Security

ECI Issues Notice to AAP Leader Atishi, BJP’s Manoj Tiwari Responds

PM Modi Advocates for Triple Talaq Law in Rajasthan

Delhi’s Liquor Scam: BJP MP Manoj Tiwari Calls for Transparency

PM Modi Asserts BJP’s Dedication to its ‘Sankalp Patra’


Delhi BJP President Virendraa Sachdeva Accuses AAP of Liquor Scam

BJP Manifesto Committee Convenes in Delhi

Delhi BJP President Criticizes Opposition Party

Gourav Vallabh, Former Congress Leader, Joins BJP in Delhi

Union Minister Ashwini Vaishnaw Sworn in as Rajya Sabha MP

BJP MP Sudhanshu Trivedi Criticizes Congress for Disrespecting Women in Politics


India-Russia Relations: A Constant Bond, Says Russian Diplomat

BJP MP Manoj Tiwari Comments on AAP’s Celebrations Over Sanjay Singh’s Bail

Vijender Singh, Boxer and New BJP Member, Pledges to Serve the Nation

Vijender Singh, Boxer and Former Congress Leader, Joins BJP

Political Tensions Rise in Delhi: Defamation Notice Sent Amidst Allegations

Delhi Political Tensions Rise: BJP Chief Virendraa Sachdeva Issues Legal Notice to AAP Leader Atishi

India Rejects China’s Naming of Arunachal Pradesh Locations

Supreme Court Grants Bail to AAP MP Sanjay Singh: BJP Responds

Delhi BJP Chief Declines Interest in CM Post

BJP Leader Gaurav Bhatia Accuses Arvind Kejriwal of Involvement in Liquor Scam

PM Modi’s Rally in Rudrapur: A Stand for National Unity and Democratic Principles

‘Namo Drone Didi’: Boosting Women’s Empowerment in Uttarakhand

Political Tensions Rise: AAP Leader’s Arrest Claim Sparks Controversy

BJP Spokesperson Criticizes Delhi CM Arvind Kejriwal’s Politics

Delhi Politics Heats Up: AAP Leader Atishi Faces ED Allegations

BJP and RLJP Strengthen Alliance Ahead of Bihar Elections

Kendrapara MP Anubhav Mohanty Joins BJP, Cites National Development as Key Reason

BJP MP Manoj Tiwari’s Sensational Claims on Arvind Kejriwal

PM Modi’s Upcoming Public Rallies and Road Shows

PM Modi Outlines the Next Decade at RBI’s 90th Anniversary

RBI’s 90th Anniversary: PM Modi Stresses on Financial Reforms

Ayodhya Predicted to be the Epicenter of Global Religious Tourism

PM Modi’s Third Term Preparations Underway: Major Decisions Expected in First 100 Days

PM Modi Announces Preparations for Third Term at Meerut Rally

Lok Sabha Election 2024: PM Modi’s Rally in Meerut Sets the Tone


BJP Leader Shaina NC Criticizes INDI Alliance, Calls for a Befitting Reply

INDIA Alliance Rally: A Platform for Political Conversion?

PM Modi to Kickstart Election Campaign in Meerut

Congress Stages Nationwide Protest Over Rs 1,800 Crore Income Tax Notice

Delhi’s BJP MP Manoj Tiwari Criticizes Current State of Affairs


Union Minister Hardeep Singh Puri Criticizes Delhi Government and CM Arvind Kejriwal

Union Minister Hardeep Singh Puri Inaugurates Election Office in Delhi

Green Horizons: PM Modi’s Eco-Friendly Vision with Bill Gates

PM Modi and Bill Gates Weigh in on AI’s Future: A Balance of Innovation and Ethics

PM Modi and Bill Gates Envision AI’s Future in India’s Growth

India Embraces AI Revolution: PM Modi’s Insightful Dialogue with Bill Gates

India’s Leap into the Digital Future: PM Modi and Bill Gates Discuss ‘Namo Drone Didi’ Scheme

India’s Leadership at G20 Summit Heralds New Era of Global Digital Policy

Union Minister Meenakashi Lekhi Extends Ramzan Wishes

Delhi Court Extends ED Remand of CM Arvind Kejriwal in Excise Policy Case

BJP Leader Shazia Ilmi Questions Delhi CM Arvind Kejriwal’s Stance on Bribery and Electoral Bonds

Nation’s Commitment Over Party Interests, PM Modi’s Message to Congress

Padma Awardee Dr. Damayanti Beshra and Former BJD Leader Sidhant Mohapatra Join BJP

Bhartruhari Mahtab Switches Allegiance to BJP After Resigning from BJD

Progress in India’s Bullet Train Project: A Leap Towards High-Speed Rail Travel

BJP Spokesperson Criticizes AAP’s Governance Amidst Corruption Allegations


AAP Leaders Sushil Kumar Rinku and Sheetal Angural Join BJP

Union Minister Anurag Thakur Advocates for Transparency and Accountability in Government

Transforming India’s Roadways: The Bharatmala Project

PM Modi Assures Legal Action to Return Looted Money to the Poor in West Bengal

Delhi BJP Files Police Complaint Against CM Arvind Kejriwal

Ravneet Singh Bittu Joins BJP: A Political Shift in Delhi

Lok Sabha Elections Stir Controversy Amidst Indi Alliance

Arun Govil, BJP Candidate from Meerut, Expresses Excitement About His Candidacy

Massive Protests Erupt in Delhi as BJP Demands Resignation of CM Arvind Kejriwal

BJP Demands Apology from Congress Over Comments on Kangana Ranaut

BJP Protests Demand Delhi CM Arvind Kejriwal’s Resignation
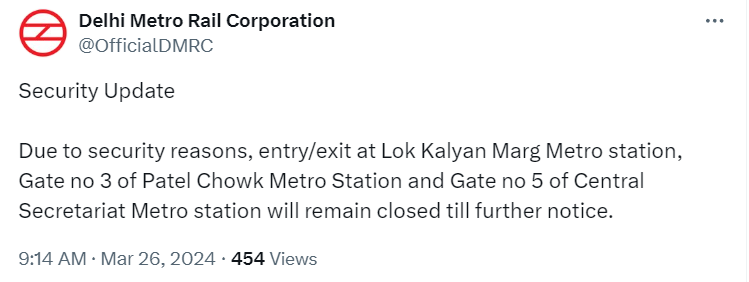
Delhi Metro Stations Partially Closed Due to Security Concerns

Karnataka Minister’s Controversial Remark Sparks Political Firestorm

BJP MP Manoj Tiwari Defends Kangana Ranaut, Criticizes Congress Leader’s Remarks

Defence Minister Rajnath Singh Attends BJP CEC Meeting in Delhi

Union Minister Bhupendra Yadav and Chhattisgarh CM Vishnu Deo Sai Attend BJP CEC Meeting in Delhi

BJP Prepares for Lok Sabha Elections: A Blueprint for ‘Viksit Bharat’ and ‘Aatmnirbhar Bharat’

Deputy CM Brijesh Pathak Attends BJP CEC Meeting in Delhi

write news article on the following news:

BJP Leader Sambit Patra Responds to Congress’ Allegations of Account Freezing

Independent MLA Hoshyar Singh Joins BJP, Cites Stagnation and Retribution in Himachal Pradesh

Delhi BJP President Accuses CM Arvind Kejriwal of Corruption

Delhi BJP Leaders Pledge to Fight Against Corruption

Delhi CM Arvind Kejriwal Sent to 6 Days ED Custody: A Shift from ‘Swaraj’ to ‘Sharaab’?

India Stands with Russia: PM Modi Condemns Moscow Terrorist Attack

Delhi CM Arvind Kejriwal Arrested: BJP MP Manoj Tiwari Responds
Delhi CM Arvind Kejriwal Arrested Amidst Liquor Scam Allegations

Prime Minister Narendra Modi Receives Warm Welcome in Thimphu, Bhutan

Prime Minister Narendra Modi Receives Guard of Honour in Bhutan

Indian Prime Minister Narendra Modi Arrives in Bhutan

PM Modi Outlines Vision for India's Future at Rising Bharat Summit

PM Modi Shares Interesting Anecdote On Bureaucrats At ‘Startup Mahakimbh’

BJP Chief JP Nadda Hits Back at Congress

BJP Spokesperson Sambit Patra Addresses Bank Account Freezing

BJP Accuses Congress of Creating Alibi in Desperation of Defeat

PM Modi Addresses Rising Bharat Summit Amidst Election Fever

PM Modi Asserts New India’s Stance Against Terrorism at News 18 Rising Bharat Summit 2024

Delhi Chief Minister Kejriwal Summoned by Court: BJP Responds

PM Modi Lauds UPI and Women Entrepreneurs at ‘Startup Mahakumbh’

PM Modi Highlights India’s Startup Ecosystem at ‘Startup Mahakumbh’

PM Modi’s Witty Remark at ‘Startup Mahakumbh’: ‘You Know What’s Going to Happen in Next 5 Years?

Prime Minister Narendra Modi Addresses ‘Startup Mahakumbh’ in Delhi

Startup Mahakumbh: A Beacon of India’s Innovative Brilliance

Prime Minister Narendra Modi Visits ‘Startup Mahakumbh’ Exhibition at Bharat Mandapam

MNS Chief Raj Thackeray Meets Union Home Minister Amit Shah in Delhi


PM Modi Sets Historic Target of 37 Crore Votes for BJP

India Assures Safety of Bulgarian Nationals, Reaffirms Commitment to Indian Ocean Security

India Reaffirms Commitment to Safeguard Indian Ocean Region

Major Political Shift in Delhi: BSP Leaders Join BJP

BJP MP Manoj Tiwari Outlines 100-Day Plan, Pledges to Follow Election Commission’s Guidelines

Major Political Shift: BSP Leaders Join BJP in Delhi

Delhi CM Arvind Kejriwal Skips ED Summons in Money Laundering Case

Delhi CM Arvind Kejriwal Skips ED Summons, BJP Leader Harish Khurana Reacts

Congress Thinks They’re the Only Smart Ones, Says BJP’s Gautam

BJP Leader RP Singh Speaks on Electoral Bonds and Political Climate

Defence Minister Lauds GAPIO Doctors for Global Humanitarian Service

Delhi: Anuradha Paudwal Meets BJP National President JP Nadda

Bansuri Swaraj Criticizes Kejriwal’s Non-Compliance with ED Summons

Delhi CM Arvind Kejriwal Granted Bail in ED Summons Case

BJP National Secretary Accuses Political Parties of Spreading Hatred

Indian Railways: A Decade of Transformation

BJP Delegation Meets Election Commission Over CAA Remarks

Central Government Slashes Diesel & Petrol Prices by Rs 2 per Litre

Bhutan’s Prime Minister Tshering Tobgay Meets President Droupadi Murmu in Delhi

BJP Leader Nalin Kohli Clarifies CAA’s Intent and Impact

Bhutan’s Prime Minister Dasho Tshering Tobgay Meets Indian Counterpart Narendra Modi in Delhi

Union Home Minister Amit Shah Unveils ‘Sankalan’, A Mobile App Compendium of New Criminal Laws

Delhi Metro Expansion: A Double Gift for the Capital City
Prime Minister Modi Empowers Street Vendors with PM SVANidhi Scheme

Prime Minister Narendra Modi Inaugurates Delhi Metro’s Phase 4 Expansion

PM Modi’s Vision: A Complete Value Chain of Semiconductors in India

Union Minister Giriraj Singh Responds to Opposition’s Allegations on CAA

Union Home Minister Amit Shah Addresses Concerns Over Citizenship Amendment Act

Union Home Minister Amit Shah Affirms Commitment to CAA

Union Home Minister Criticizes Delhi CM’s Statement on Refugees

PM Modi Unveils PM-SURAJ Portal, Extends Credit Support to One Lakh Entrepreneurs

Union Home and Cooperation Minister Amit Shah Inaugurates New Cooperative Offices

India’s Leap into Semiconductor Manufacturing: A New Era Begins

India Accelerates Semiconductor Manufacturing with Four New Units

Prime Minister Narendra Modi Kickstarts ‘India’s Techade: Chips for Viksit Bharat’

Defence Minister Approves Expansion of National Cadet Corps

Union Minister Rajeev Chandrasekhar Speaks on CAA Implementation

Union Minister Ashwini Vaishnaw Discusses Railways Challenges, Unveils ‘Rolling Block System’

Union Minister Anurag Thakur Reaffirms Commitment to CAA

DRDO Successfully Tests Indigenous Agni-5 Missile with MIRV Technology

PM Narendra Modi Attends Sashakt Nari-Viksit Bharat Programme in Delhi

Prime Minister Narendra Modi Announces Major Infrastructure Developments

BJP’s Tarun Chugh Criticizes Gandhi Family, Asserts NDA’s Unity

BJP MP K Laxman: “Populist Leaders Joining BJP to Strengthen PM Modi”

Delhi BJP President Criticizes Government Over Recent Tragedy

BJP MP Brijendra Singh Defects to Congress Ahead of Lok Sabha Elections

Telangana BRS Leaders Join BJP in a Significant Political Move

Turmoil in INDI Alliance: BJP Spokesperson Shehzad Poonawalla’s Take

Union Minister Piyush Goyal Lauds Artists at ‘Viksit Bharat Ambassador Artist Workshop’


International Drug Kingpin Jaffer Sadiq Arrested by NCB in Delhi


BJP National General Secretary Tarun Chugh Criticizes Political Dynasties in Jammu and Kashmir

Prime Minister Narendra Modi Lauds National Creators Awards

BJP MP Manoj Tiwari Criticizes Delhi Government in a Fiery Speech

Defence Minister Rajnath Singh Endorses Film ‘Article 370’

“A Significant Leap Towards Nari Shakti”: Shehzad Poonawalla Lauds PM Modi’s Initiative

Amit Shah Unveils National Cooperative Database, Aiming to Revolutionize India’s Cooperative Sector

Rescheduling of BJP Central Election Committee Meeting

Women Lead the Charge in India’s Development: Union Minister Smriti Irani


Bloomberg Challenges Trial Court’s Order on Zee Entertainment Article

Union Minister Arjun Ram Meghwal Defends Indian Democracy

Former Congress MP Rajesh Kumar Mishra Joins BJP

Delhi Inaugurates 201 New CNG Stations, Aims to Quadruple Capacity

BJP State President Criticizes AAP-Congress Alliance in Delhi

Defence Minister Rajnath Singh Leads DefConnect Seminar in Delhi


Javed Ali’s Mesmerizing Live Performance Coming to South Delhi!

Delhi Police on High Alert After Blast at Bengaluru Cafe

Amit Shah, Manik Saha to Attend Landmark Agreement Signing in Delhi

Modi Government to Induct 25 Private Sector Specialists in Key Posts

Adani Group wants to raise 21,577 crore for green hydrogen projects and airport expansion.

Today, the price of Paytm shares rises once more. What is the rally's motivation?

Pharma Varsity V-C is ordered to be removed by Delhi LG Saxena.

Reviving 1,027 vice-principal positions in Delhi is approved by the Delhi L-G.

Why drumstick water, or moringa, is a superfood

Virat Kohli will miss the opening of two test matches against England

Regarding the India merger, Sony sends a letter of termination to Zee: Report



Ankita Lokhande, winner of Bigg Boss 17, apologises to Vicky Jain and his family for striking him.

Red Alert: Delhi Experiences Its Coldest Day of the Season With A Low of...

Alert: Traffic in Delhi! Republic Day Parade Rehearsal is advised. Stay away from these paths.

Why, when constructing New Delhi, the British chose to spare the Sunehri Bagh mosque



Teachers question why Delhi University College is asking staff members to enrol in Gita courses.

Debt discussion: Finance Ministry's reaction to the IMF's most recent India consultation details

Inside Images From Salman Khan's Friends Birthday Celebration

Images From The Wedding Celebrations of Ira Khan And Nupur Shikhare


Why is apple, beetroot, and carrot juice known as ABC Juice the ultimate miracle beverage?

Expert advice on increasing feel-good chemicals in the brain

Sugar cravings: Is it fatty liver, diabetes, or obesity?

In New Delhi, Railways Minister Ashwini Vaishnaw examines the Amrit Bharat Train.

Dua Lipa travels to Humayun's tomb at the Bangla Sahib Gurudwara in Delhi after visiting Rajasthan.


Shares fall by around 3% as a result of Infosys' massive AI deal loss.



30 aircraft are delayed, 5 are diverted, and thick fog covers Delhi-NCR as the temperature drops.

Academic of medieval history at St. Stephen's College, Tasneem Suhrawardy, passes away.




Delhi Airport sends a warning when flight operations are impacted by dense fog and zero visibility.




At Umang 2023, Deepika Padukone's Sabyasachi saree was merely a royal demonstration of ethnic style.



The air quality in Delhi is'severe', with an AQI of 400. Gopal Rai has reimplemented GRAP III.

Dense fog in Delhi causes numerous domestic and international flights to be delayed.


Center requests a review of the SC ruling in the Chhawla murder case



Delhi-NCR construction is once again permitted when the air quality improves

In Delhi's Punjabi Bagh, a woman's body is discovered


What are the advantages of shutting up ladies at 9.30 p.m. in Kerala?

Rajasthan Minister's Sleazy Video Goes Viral, Calls for His Removal

Jallikattu hearing: If the court bases its decision on images, it will be harmful

cyclonic storm is expected to strike these states, and the IMD has issued a rainfall warning.



We must devise numerous equitable methods for various groups: CJI

MCD is scheduled for vote counting amidst strict security

Boy, 8, falls down borewell in MP's Betul; rescue efforts are under way

Punjab Police will examine singers Babbu Maan and Mankirt Aulakh in the Moose Wala murder case.

Mumbai residents walk instead than jog as the city's AQI rises to 309

What propelled Delhi's AAP to travel to Gujarat to battle the BJP

Journalist Khalid Gul was detained in Anantnag, Kashmir, in 2017

Day 3 of Rajasthan's Bharat Jodo Yatra for Rahul Gandhi has begun.

SC questions how long the Agusta accused can be detained with 250 witnesses still present.

PM Modi asks for a paradigm shift as India takes over the G20 chairmanship.



Winter Session has 25 Bills and little time for debate, according to the opposition


No Sign Of Sweep In Delhi; AAP vs. BJP Is Close: 10 Facts

DMK petitions the SC to review the EWS decision.

government tells 34 female Army officers to list the steps for advancement

Supreme Court rejects the request to recognise Godman as "Paramatma."
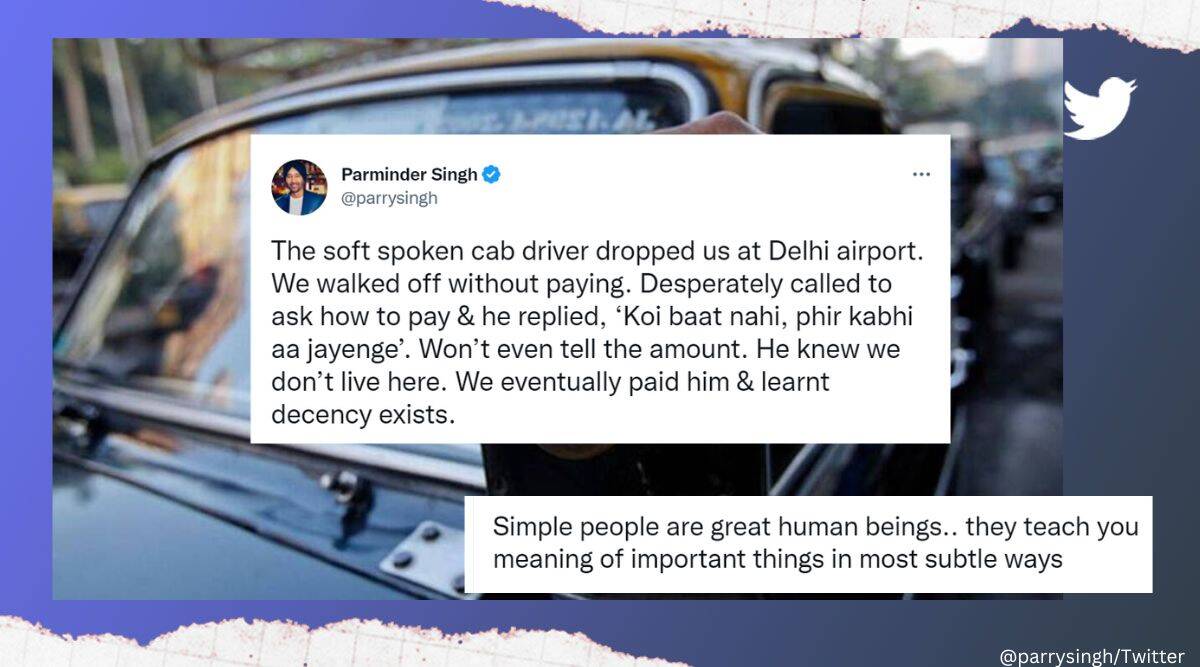

Prime Minister Narendra Modi paid homage to Babasaheb Ambedkar on his Mahaparinirvan Diwas.

Bachelor tenants must leave THIS Noida society by December 31 - Here's Why


Despite a drop in stubble burning, the smog has blanketed the capital of India.

Elections in UP: SP and BJP accuse one another of breaking the code

Over a land dispute, a man beheads his cousin, and his friends take a head-selfie

Court Refuses to Grant Man Bail in Rape Case: "Consent of Minor Is Not Consent"

Kyiv accuses Russia of attacking its air bases; retaliatory missile attacks follow | Top 5
After receiving a kidney donation, a Union minister called Lalu's daughter "proud."

Mahaparinirvan Diwas: Ambedkar's assertion that Buddhism is superior than Marxism.

Smiles, jokes, and holding hands at the PM's tea with opposition leaders at the meeting

"Serious Issue," "Against Our Constitution" conversion: Supreme Court


Voters with disabilities are enthusiastic about exercising their rights in Delhi


Pendency is growing amid standoff over judicial appointments

Right to convert others is not a part of religious freedom: Gujarat government in SC

No one is hurt as a portion of a central government-run children's hospital's ceiling collapses.


9 fire engines are on the scene at a hotel fire in Delhi's Karkardooma.


Once again, Delhi's air quality deteriorates to "severe," triggering GRAP Stage III.

Bengaluru couple loses all hope after four vehicle thefts and burglary in 50 days.

Employees of PM Package are afraid following a credential leak.


The first G20 Sherpa gathering under Indian Presidency is held at Udaipur.

After party's kidnapping charge, Gujarat Congress MLA "Ran To Save Life"

Key UP Today polls provide a significant challenge for Akhilesh Yadav's family: 10 points

Before a kidney transplant, Lalu Yadav's daughter is "Ready to Rock and Roll."

Clarify position on medical students who returned from China's petitions: Supreme Court


GM mustard will result in permanent harm, the Supreme Court said

BJP wants to change the Delhi Development Act.

Delhi High Court orders social media sites to remove online "sexually explicit videos"

Rashtrapati Bhavan is now open to the public. Check the Days, Times, And Other Information Available


200 new Vande Bharat train rakes are up for bid, according to Indian Railways.



BJP criticises Kejriwal for the police use of force in Punjab; in Gujarat, the AAP says...

2 people are arrested after a South Korean YouTuber was harassed in Mumbai while livestreaming.


KCR's Daughter Named By Probe Agency In Delhi Liquor Policy Case

Saying "Abhi bhi time hai" Bal Thackeray's vintage "Gujarat gaya" video is shared by Jadeja.


Swara Bhasker marches alongside Rahul Gandhi in Ujjain and joins Bharat Jodo.

Delhi: A woman beats a man on the dais with her slippers at a Hindu Ekta Manch gathering.


Satyendar Jain appeals a court decision denying him bail to the Delhi High Court.
7-year-old MP child saved at Delhi hospital after being harmed by plant seeds

DU to colleges: As of right away, fill all authorised assistant professor positions.

Delhi excise policy case: Amit Arora, a businessman, gets detained by ED


Harish Salve on Kiren Rijiju's "Lakshman Rekha" gaffe: Law Minister's Judiciary Remarks


Amit Shah dismisses the Gujarat challenge and says "AAP Name May Not Figure In List"

Toyota Kirloskar Motor Vice Chairman Vikram Kirloskar passes away from a heart attack.

Walking along Delhi's third-rate lanes and first-class neighbourhoods

In a polygraph test, Aaftab Poonawala claimed he had no regrets about the murder: Sources

President's Concern Over Non-Release Of Poor Prisoners Leads To Supreme Court's Recognizance

According to a US report, China warned its officials not to stifle relations with India.


Concern: The government returns 19 names that the college sent, but the supreme court reiterated 10

India opposes attempts to tie carbon reductions to agricultural practises.

Horrific Car Accident On Mumbai-Pune Highway Results In 5 Deaths And

How A Delhi Gang Committed A $20 Million Scam Inspired By The Movie "Special 26"

Delhi Murder: Police Across City Told to Look for Human Remains in Your Neighborhood

In a dispute with the AAP, Delhi's Lieutenant Governor takes action against an official.

Today's launch of India's first privately built rocket, the Vikram-S: 10 Facts


PM Modi expresses sympathy and grants ex-gratia following the Mizoram stone quarry collapse.

Punjab pavilion was the star of the New Delhi trade show.

Elections for the Delhi Municipal Corporation feature almost 2,000 candidates.

IRCTC has the option to alter the train food menu.

AAP employee caught up in ACB investigation for selling Delhi MCD ballots

Man is detained after blackmailing a student with a video of his girlfriend in bed.

Gujarat Assembly Elections 2022: Shashi Tharoor chooses NOT to run in Gujarat

Delhi High Court orders Amazon India to stop selling Rooh Afza produced in Pakistan


Supreme Court: It's time for a law regulating MPs and MLAs' offensive speech.


SGPC informs Mohan Bhagwat that it is time for the RSS and BJP to stop meddling in Sikh issues.

According to Narayana Murthy, "Even our IITs have become coaching centres."

Gurgaon Woman to Receive ₹2,00,000 in Compensation for Dog Attack

Morbi Bridge Project was awarded without a Tender, the Gujarat High Court questions.

Donald Trump declares his bid for the 2024 US presidential election, saying "I Am Ready"


According to the petroleum minister, the government would implement the GST on fuel.


CM's request, the Karnataka Milk Federation withdraws the price hike on milk and curd

UP student dies during a rape; suspect claims police he took an energy supplement


Priya, a young footballer from Chennai, passed away as a result of poor leg surgery.


Four workers are still missing following a Mizoram stone quarry collapse, leaving at least 8 dead.

Over the Satyendar Jain controversy, a Tihar official was fired.

Due to the severe rains in Tamil Nadu, these districts will close their schools TOMORROW.

Nalini encounters her husband and three others at a special camp.

"Beginning Of End Of War": Zelensky in Ukraine Following the Liberation of a Major City

According to Xi Jinping, "China Has Chinese-Style Democracy." Joe Biden

Hotelier nabbed in Andaman gang rape case while holding a Rs. 1 lakh reward

Supreme Court will convene a new bench to examine homebuyers' complaints against Amrapali Group.

Culture preservation for future generations

Delhi High Court's selection committee is authorised to appoint non-judicial personnel

Removal of 6.3 lakh posters and banners prior to the Delhi MCD elections

"Yatra will make sure the government is held responsible for people's difficulties,"

Body Disposed In Delhi Forest Over 18 Days At 2 A.M., Chopped Into 35 Pieces

Dhols, baraatis, and a haldi ceremony at a dog wedding in Gurugram


Father leaps off of the train to save his 3-year-old daughter, but they both perish.

Punjab Experiences A 4.1-Magnitude Earthquake Days After Tremors In Delhi

Gujarat elections: The Cong nominates Vadgam's Jignesh Mevani; see the fifth and sixth lists here

India is not subject to a price cap and can purchase as much Russian oil as it desires.


Shlok Mukherjee of Kolkata has won the Doodle for Google 2022 India contest.

India BLOCKS attempts at the UN Climate Summit to group it with past polluters. COP27

Gujarat elections: Isudan Gadhvi's biggest obstacle is loosening Ahir's hold in Khambhalia

Convicted in the Rajiv Gandhi Case, who seven times received execution orders


G20 summit in Bali will emphasise food and energy security.

AI and drones to examine Delhi's pollution hotspots and emissions

'Vote bandi' by the Delhi Anganwadi Union against the BJP and AAP

Missing ex-serviceman discovered; not abducted

US is planning to expand its interview waiver programme to new visa categories.

Workers for the Himachal Congress Women's Wing take pictures with

10 Guarantees From Arvind Kejriwal Before Delhi Municipal Elections

Rahul Gandhi relates a "controversial" incident, saying, "RSS chief took chopper, I walked."


What the new TV broadcasting regulations say about content of national interest

Shot in Kotkapura while on bail, a Dera Sacha Sauda adherent

One JeM terrorist was killed in a clash in the Shopian district of J&K.

Arrest of the director not at all related to operations Purdue Pharma Inc.

T.N. Rains | District-wide breaks announced for schools and universities

Rahul Gandhi's Sharp Message at Maharashtra Rally: "Daro Mat"

Delhi Police and the French Embassy start training on cybercrime

Family Alleges Threat From 3 Released Supreme Court Rape-Murder Convicts

Education not business to generate profit, tuition charge must be affordable, rules Supreme Court

Metro deploys 8-coach trains for the Red Line

Doctors and other personnel at Delhi's RML Hospital are urged not to wear casual clothing on campus.


Gurudwara Bangla Sahib: How a mansion from the 17th century become a sacred site for Sikhs

Mark Zuckerberg had a "prep talk" with staff before cutting jobs, according to a report

Meet Aruna Miller, the first Indian-American politician in Maryland history.

Ravindra Jadeja's wife is reportedly on the BJP's probable list for Gujarat.

After swearing in, Chief Justice DY Chandrachud's "Citizen First" message

Right wing leader's bow before Sudha Murthy causes a disagreement.

According to Nitin Gadkari, Manmohan Singh is responsible for India's economy's liberalisation.


Prior to the visit of the PM, the BBMP is once again busy working to renovate Majestic.

Supreme Court overturned the order to block the Congress Twitter account.

6.6-magnitude earthquake that hits Nepal leaves six people dead and causes tremors in India.

Chief Justice of Modi's polarising India is a pro-LGBTQ rights judge.

Traffic Will Be Diverted On These Routes Today In Delhi On Prakash Parv; Please Read This Advisory

Doctors at the government-run Lady Hardinge Medical College want private security to be reinstated.

Heavy vehicle entry restrictions are lifted by CAQM as the air quality improves

India Gate's food booths may be run by women's self-help organisations.


Delhi municipal elections would be contested by SAD, says Paramjit Singh Sarna



Man slain in Madhya Pradesh by his wife's family

Tripura boy kills three family members and throws their remains in a pit; he is detained

Robbers shoot the shop owner twice on camera and flee with cash and gold.

81% of Delhi-NCR families have air quality problems, according to a study.

Today's ceremonial bench hearings will be aired live by CJI U U Lalit.

Uttar Pradesh government would remove encroachment on enemy properties.

Govt to consider requests from the families of Mra and Dao

In Dhamnagar, Odisha, the BJP triumphs, defeating Naveen Patnaik's party.

Sources: Rahul Gandhi would run for office in Himachal

Delhi Pollution Drops, Diesel Vehicle Restrictions Are Lifted, and Industries

Delhi Auto driver, accused of harassing a woman, dies while escaping police

As Jaishankar departs for Moscow, concentrate on New Delhi's function


Election in Andheri is won by Team Thackeray as predicted. Finally, the Surprise


10 points from a significant Supreme Court ruling on the poor quota today

Convicted rapist's death sentence was commuted

Delhi High Court requests AAP MLA's opinion on challenging bail in Waqf Board matter

Accident in North Delhi results in one fatality and two injuries.

Reminder of PIL challenging the legality of RPA provision

Expulsion of two South Asian University (SAU) students due to protests about scholarships

Ela Bhatt's SEWA is an example of a vision for women's empowerment.


FTA between India and the UK unlikely until March as Rishi Sunak settles in

Marathi billboards: The next step won't be decided till the supreme court issues its ruling: BMC

Chandigarh maintains its second-place ranking in the school education index with a score of 927.

Mistry was murdered in an accident because of a traffic rule violation: police

Student in Andhra Pradesh beaten and burned with an iron box by classmates

After the Munugode by-election, Congress leaders concentrate on the Bharat Jodo Yatra in Telangana.


Sudhir Suri, leader of the Shiv Sena, shot dead in Amritsar.


Pension plan for employees: SC upholds modifications as valid and reads down some regulations

Elon Musk encourages staff to be prepared for mass layoffs while Twitter is down for some users.

Elon Musk removes "Days of Rest" from the calendars of Twitter employees: report

Apple switches from China to India and adds a new iPhone 14 supplier.

Delhi Primary Classes Will Be Canceled Starting Tomorrow, With "Odd-Even" Being Considered

On November 10, the Supreme Court will discuss the subject of stubble burning.

MCD polls: Delhi BJP won't re-elect departing council members in 60–70% of seats


Poor patients at Delhi's Ram Manohar Lohia Hospital suffer due to a lack of medication

Delhi NCR gasps as over 3 lakh non-BS VI diesel automobiles are outlawed.

In the Media One case, the Supreme Court has reserved judgement over a channel broadcast ban.

MEA discredits Pakistan's J&K storyline

Internet access was unavailable in 66% of schools during the academic year 21–22: Report

KCR Provides Videos To Support BJP's MLA Poaching Claims: 10 Points

Today, Arvind Kejriwal will reveal the AAP's candidate for Gujarat's chief minister.

Huge criticism and allegations following Imran Khan's attack on the Pakistani prime minister: Top 10


Supreme Court dismisses complaint against CJI-designate as "misconceived"

Appoint paramilitary security forces: Lady Hardinge's doctors

ITEC programme in Gurugram, attended by 7 nations.

Disappointed applications outnumber allottees in Bhoomiheen Camp.

In Delhi, Gautam Gambhir constructed a library. He has now received a court summons.

Report on the Performance Grading Index for States and UTs is released by the Ministry of Education.

Jagir Kaur is put on leave by SAD due to "anti-party activities"

We Love You is displayed on the Burj Khalifa as a birthday greeting for Shah Rukh Khan.

10 things to know ahead of today's market opening in the stock market




As North Korea launches missile strikes, Japan and South Korea warn the public.

Again, Delhi's air quality is "severe," causing children to have trouble breathing.

The Indian economy and M-commerce




Prices of gasoline and diesel won't drop by 40 cents as of right now.

Steel Man of India Jamshed J Irani, 86, passes away

Reserve Bank will introduce the first digital rupee pilot programme today.

Saudi Prince, Jack Dorsey, and Qatar Are Among Elon Musk's Twitter Investors

Delhi experiences "severe" air pollution


Lt. General Batoo Tshering of Bhutan is presented with the Guard of Honor at South Block.

Supreme Court issues a warning to the states over the 2-finger test to prove rape

Fire breaks out at a footwear factory in Narela; 2 die

Package specifications for the IRCTC Mahabaahu-Assam River Cruise

How the government, businesses, and residents collaborated to restore a Tughlaq-era garden

All India Muslim Personal Law Board is opposed to the Uniform Civil Code.

70-person business group led by a Taiwan minister will visit India this week

Delhi Confidential: A persistent petitioner is trying the CJI's patience.

Task force member: Cheetahs will relocate to a larger enclosure at Kuno Park in November.

Police in Uttar Pradesh arrest a man for allegedly "digitally raping" a 6-year-old girl.

IIT-Kharagpur student's family wants CID investigation into death



According to the government, the CAA may not be subject to judicial review.

National Unity Day: HM oversees Sardar Patel's birth anniversary celebrations in Delhi

More than 3 lakh PUC certificates are issued by the Delhi transport department in 12 days.

SC will hear 232 petitions on the CAA issue today.


"Mere engagement does not permit a man to sexually abuse his fiancé," said the Delhi High Court

According to a poll, 70% of respondents or their families were impacted by low air quality.

In order to stop inmates from smuggling, nets are being placed in municipal prisons.


Overview of the Russia-Ukraine conflict as of day 250 of the invasion

Jair Bolsonaro is the first president in office in Brazil to lose a reelection campaign.

Bridge collapse in India: 141 dead, many unaccounted for

Medical director of RML Hospital issues a directive: "Start OPD on time."

An inning is divided into three sections by Rohit, Kohli, and Suryakumar.

In a car crash in the US, three Indian students were killed


Top 10 things to know before of today's market opening for the stock market


`Rajnath Singh: To get to Gilgit, Baltistan, we have to walk north.

Putin's Big Praise: "PM Modi A Patriot... Future Belongs To India"

Prime Minister Research Fellowships offered to 12 Jamia students

SC denies MoS Mishra's request to transfer the appeal in the murder case.

Aspirants for DU find there is no "downgrade" option.

President Murmu will award the Silver Trumpet to the President's Bodyguard.

Ram Rahim was paroled, according to Manohar Lal Khattar, the chief minister of Haryana.

list of the most popular courses at DU with open seats

In Telangana, "Op Lotus" Cops said they were caught trying to bribe KCR's MLAs.


Elon Musk informs Twitter staff that he does not intend to eliminate 75% of jobs.

In Bihar, individuals flee as a speeding cargo train's engine drags a waggon.

Channel and broadcaster fired for turning a discussion into a "community issue"

CBDT extends the deadline for submitting the IT return for AY 2022–23 to November 7.

AAP Criticizes Lt. Governor Over Chhath Puja Scandal as "Hungry For Cheap Publicity"



Akal Takht Jathedar criticises "so-called pastors" who preach "pakhandwad" on Bandi Chhor Diwas.

Cricketers from India dissatisfied with post-practice lunch due to lack of hot food


Governor serves notice to two more after 9 Kerala VCs refuse to resign.

Biden's invitation to three Indian-Americans for a Diwali party conveys this message.

Chinese envoy: India and China shouldn't meddle in one another's internal affairs.

Cyclone Sitrang strengthened winds and helped spread air pollution.




Biocon Chief Kiran Mazumdar Shaw Mourns Husband, "Devastated"
SGX Nifty down 30 points; here are the market changes that occurred while you slept

During a Diwali clash in Gujarat's Vadodara, 19 people were detained by police.

Lowest Covid daily tally in India in six months | 5 points

Sale of tainted alcohol: Delhi police conduct multiple raids, arrest four people

Delhi experiences a minimum temperature of 14 °C.

Sanjay Arora, the commissioner of the Delhi Police, met with the officers who were on duty.

More than 54,000 students are accepted into DU colleges.





As Cyclone Sitrang hits Bangladesh, 7 people are killed and many are evacuated.






On October 26, Rahul Gandhi will be in Delhi for Kharge's inauguration.

Trains' top speeds can be raised to reduce travel times on the busiest routes: Railways

UN cracks down on terrorists: India criticises China's blocking tactics

Row over Udit Raj's statement about the "evils of Hindu" as PM Modi travels to Kedarnath

UP hospital is sealed as a Dengue patient dies because "juice was transfused, not platelets."

WHO Chief Scientist: Omicron Variant May Cause Another Covid Wave
Text of Congress's response to Shashi Tharoor's allegation of "poll irregularities"


P Chidambaram's "Advice" to the PM Regarding Rupee Concern Mentions Raghuram Rajan

Police in Ghaziabad Say the Woman Made Up the "Gang-Rape Story"


"Biggest Proof Is" That Nitish Kumar Has Contact With the BJP, claims Prashant Kishor

As residents and guards of Noida apartments fight, hair is pulled and slapped.


80,000 candidates made the cut on the initial list of seats at DU that was released.


To experience Delhi's holiday fervour, there are three Diwali melas you simply must attend.

Delhi: Anurag Thakur announces a national cleaning campaign
Make the decision to include transgender people as a third gender in bus representation.

Haryana gives the go-ahead for the Palam Vihar metro

Karnataka: You must use a seatbelt or you'll be fined ₹1,000

Kerala Man Arrested For Using a Baton on His Wife Who Refused To Resign From Her Job

US Reaction To Pulitzer Prize-Winning Journalist Stopped At Delhi Airport

Rajasthan MLA's remark about how things have changed as Gehlot supporters meet Kharge


Doctors criticise the "special services" provided to MPs for medical care at AIIMS.

Ghaziabad gym instructor passes away from a heart attack while seated on a chair, as seen on camera

In Amritsar, a Punjab cop "accidentally" fires at a worker from a mobile store.

Despite UN pressure over Ukraine drones, Russia and Iran remain stubborn.

Sleepless Nights For Some, Eknath Shinde Declares on Stage with Sharad Pawar

Suella Braverman, the UK's home secretary, resigns after saying, "I've Made A Mistake."

Mallikarjun Kharge desired to see Sonia Gandhi following his victory. She had another idea.

Ali Fazal wants Sajid Khan, a #MeToo accuser, ejected from Bigg Boss 16



Bobby sent Sunny Deol, who turned 65 today, the best birthday greeting.

Varun Dhawan's werewolf in the Bhediya trailer is both humorous and dangerous.

PWD officials expect the Delhi Ashram Flyover project to be finished on schedule.


Name the policeman who apologised to the public or made the "offer": BJP

By year's end, Delhi Municipal Corporation plans to install 90 more EV charging stations.

The Central Organization for Modernization of Workshops will close, according to Indian Railways.

Bangladeshi government cancelled Nora Fatehi's dance performance for THIS reason.

In J-Shopian, K's a hybrid terrorist was killed in a counterterrorism operation.

China again opposes an Indian-American UN resolution to blacklist terrorists based in Pakistan.


Himachal Pradesh Assembly Election 2022: The BJP's list of 62 candidates is uncontested
Security benefits of new airfield close to the Pakistan-India border: PM Modi

Shashi Tharoor Claims "Damning" Evidence Of Election-Rigging In The Congress


2 Collegium members reject formal authorization

Residents in Air India Colony in Vasant Vihar, Delhi, claim there was an attempt to evict them.


DCC works to ensure the success of Rahul Gandhi's Bharat Jodo Yatra

Andhra Doctor Requests the Rescue of His Pet Jaguar and Panther from Ukraine


Following a bus accident that killed 25 of the wedding party, Uttarakhand Gorge

KCR's National Party Launch in Telangana at 1:19 PM, Eye on 2024: 10 Facts

Horrific Car Crash On Mumbai's Bandra-Worli Sea Link, 5 Dead


According to the Supreme Court, accepting a job on compassionate grounds is not a right.
Delhi now has 937 confirmed cases of dengue, over 400 more than before.


4 members of the outlawed Popular Front of India are arrested by the Delhi Police




Burn injuries and throat slitting in J&K jails DGP's remains, police looking for domestic help




Family Alleges Murder, Rape After UP Teen's Naked Body Is Found In Field

Everything You Need To Know Before Trading On October 4 in the Stock Market Today




Agreement on a Line of Credit for the Maldives of USD 100 million

Three men chase and stab a 19-year-old in Delhi's public space.
Today, MoS Muraleedharan will travel to Oman for two days.

After using marijuana, two boys kill a child, claiming that Lord Shiva demanded a sacrifice.

21-year-old Gujarati student is seen on tape collapsing and dying while doing a garba.

Rahul's visit connects two communities that were split apart after the 1993 conflict


Tri-services As the era of stand-off weaponry dawns, Missile Command is on the verge.

UP Puja Pandal Fire: Over 60 Injured, 5 Dead, 3 Children

Day After M. Kharge's "He Wanted A Fight" Remark, Shashi Tharoor's Post

Today, the Air Force receives the first light combat helicopters built in India.


This penny stock, trading at one cents, announces the record date for 2:1 bonus shares.


Nitin Gadkari to Mercedes-Benz: "Even I Can't Afford Your Car"

Why Sensex is up 1,000 points despite the RBI raising interest rates today

Sebi tightens IPO disclosure standards

Price reductions for commercial cooking gas cylinders. Rates For Your City, Check


Concern concerning Punjab's management of air quality is expressed by the Center.

On October 2, Delhi Metro services will be reduced.

Delhi: If you scrap an old vehicle, you can receive a road tax credit.


Can't let inexperienced drivers operate in public, HC informs DTC

Rain is predicted to start falling in Odisha today.

In October, banks were closed for 21 days, including state holidays. Details

UGC approves "experts" without a PhD for the position of professor of practise.

Shashi Tharoor responds after holding out a false map of India, "Not done on purpose,"

Justice Magrey will take over as the new Chief Justice of J&K and Ladakh.

'Rattled' BJP places historical fiction on the top page of a Kannada newspaper: Iram Jairam

Finally, Shashi Tharoor and Gandhi supporters' Mallikarjun Kharge are facing off: 10 Facts

India abstains from voting at the UN in protest of Russia's alleged annexations of Ukraine.


Moscow cautions New Delhi against restricting the price of Russian energy

Coming soon: Delhi's days with poor air quality

G-23 leaders gather in Delhi as the contest for Congress presidency intensifies.

In Delhi, Sachin Pilot speaks with Sonia Gandhi about the recent events in Rajasthan.

After a brawl over a woman, a bullet was fired at South Delhi Hospital.

Top 10 Reasons Why Mallikarjun Kharge, A Gandhi Family Favorite, Will Be President

model, 30, discovered dead in a hotel in Mumbai with a suicide note!




"Time To Close Ranks," G-23 Member Manish Tewari Declares He Won't Contest

New Vande Bharat Express to Launch Today From Gandhinagar Under PM Modi In Gujarat

Priyanka Gandhi Vadra Participates Actively In Congress's Internal Voting, According to Sources

While walking, Rahul Gandhi claims to have knee problems, although


After a fight at the university, a Jamia student was shot inside a Delhi holi family hospital.

Delhi High Court requests government position on using veterinarians to treat lumpy skin disorder.


Gurdwaras praise HSGMC's Bhupinder Singh Hooda

"862 more dengue cases are being investigated, and the outbreak will persist"

When an official encounters the mother of an accident victim child, they both cry.

In Jammu and Kashmir, Another Bus Explodes Hours After the First Blast: Police

India Shuts Down PFI Leaders' Twitter Accounts After Being Banned Group

Who will win the Congress presidential nomination—Shashi Tharoor, Digvijaya Singh, or Ashok Gehlot?

An ancient Temples and Caves Found in the Madhya Pradesh Tiger Reserve

Lt. General Anil Chauhan is the new chief of the defence staff (CDS) (Retired)

Girl student who requests a sanitary pad receives a warning from a senior Bihar official.

Muslim teens seeking to enter Ahmedabad "garba" venues are attacked by Bajrang Dal members.

Karnataka becomes the official home of HAL's rocket engine production facility.

Punjab reports 30% more rain in September, but the paddy crop is unharmed.

Complaint against Assam CM, Sadhguru for night safari inside Kaziranga: "Law is same for all"

An Unusual Law Ministry "Circular" to Explain Mukul Rohatgi Refusing the Position of AG

Regarding the firing of Charlie Dean, Deepti Sharma said, "We had warned her."

Significant victory for Delhi L-G as the high court orders AAP to remove "defamatory posts"


Raids On India's Popular Front Round 2: 247 Arrested, 10 Points



Iranian anti-hijab protests used the phrase "Death to the tyrant," which resulted in 75 deaths.

Government bans 45 videos from 10 YouTube channels for providing false information

Unauthorized cremation of a teen in Uttarakhand, according to the mother



After revolt, Ashok Gehlot Is Disqualified As Congress President: 10 Facts



Harsha Engineers Listing Today on BSE and NSE: Listing Time, Gains, Share Price, Key Information

Vaishali Paresh of Prabhudas Lilladher suggests two stocks to buy today, September 26.

Dollar reaches a fresh two-decade top as rupee falls to a record low of 81.55.

Sensex Drops 1,000 Points, and the Nifty Crash Bloodbath Below 17,000

This week, 5G services in India will go live. 5 important things to remember


vehicle falls into the Himachal Gorge, 3 IIT students are among the 7 tourists killed.

In Rajasthan, the "Congress chhodo" mission is raging as Rahul's yatra begins Day 19.

Woman Cuts Hair From Brother's Grave After He Killed in Anti-Hijab Protest

When Will Cheetahs From Namibia Be Available For Public Viewing? PM Gives a Response

Sadhguru and the Assam chief minister were charged during a jeep tour in the park after dark.

Pakistani politician on London heckling: "Toxic impact of Imran Khan's politics."

S. Jaishankar Criticizes the "Biased" Coverage of India in the US Media for "Politics at Work"

Sikh refugees evacuated from Afghanistan to India recall Taliban atrocities.

Italy is ready to get its first female president. PM, far-right leader Meloni polls well.


Byju's pays $234 million to Blackstone in connection with the Aakash transaction: Report

Nestle would invest 5,000 crore in India and look into acquisitions.



After farmers' demands are met, a 21-hour highway blockade in Haryana comes to an end.


If Patna Sahib and Delhi can have gurdwara bodies, why can't Haryana?

Farmers in the Northwest are on alert due to heavy rains.

Delhi closes 11 Covid Care Centres after cases are under control.

Commuters in Delhi should avoid these locations owing to waterlogging and potholes.


Islamist PFI is in disarray as a result of a meticulous NIA raid execution

Manmohan Singh was remarkable, but India stagnated under the UPA era: Murthy Narayana


Manmohan Singh was exceptional, yet India's economy stalled: Murthy, Narayana
"Prime Minister Should...": Ex-Vice President Venkaiah Naidu Offers PM Modi Advice

Uddhav Thackeray's Court Victory Over Team Shinde

Uddhav Thackeray Signs $20 Billion Deal With "Eknath Shinde's Delhi Mujra"

Over 100 people arrested in anti-terror raids against the Popular Front of India.

Here are the top 10 developments in this big story.

Comedian Raju Srivastava died at the age 58, leaving fans in tears.

BJP leaders responded to Kamal Nath Congress' comment that he "should buy a lot more automobiles"

Weekly Numerology Predictions for September 19th to September 25th, 2022

Gauri Khan's Dating Advice for Daughter Suhana on Koffee with Karan 7


Buy or sell: Prabhudas Lilladher's Vaishali Parekh advises two stocks to buy today

Supreme Court directed the Centre to clear OROP arrears within three months.

For three years, the NDMC hold the tree ambulance project hostage.

'Chor Parivar' of Delhi arrested for stealing Rs 1 crore in jewellery.

Cops go undercover to solve a murder case that has been open for 25 years in Delhi.

Arvind Kejriwal Forms AAP With "God's Intervention" to Save the Constitution



In Rajasthan, a dog was attached to a car and hauled throughout the city; the driver was arrested.

Leaked Chandigarh Hostel Video: University Closed Until Saturday, 3 Arrests


PM Modi has named one cheetah Asha, and other huge cats have also been named.

After Assam, Arunachal Pradesh has been affected by the 'leak' of recruitment exam papers.

Punjab ex-CM Amarinder Singh will join the BJP today, bringing his party into the saffron fold.

Auto driver-turned-chef is overjoyed after winning Rs 25 crore.

Before the AG, present all cases: Ministry of Law

Jamiat opposes the High Court's appeal of the Waqf Act.

shut down law schools that don't have the bare minimum of amenities; Delhi HC to BCI

How Mahatma Gandhi "fired the first salvo" in establishing Jamia Millia Islamia

Over 35,000 tuberculosis patients will be adopted by CII as part of the effort to end TB.


Pakistan PM Refuses to Respond to Masood Azhar's Question Regarding Action


1,200 souvenirs that were presented to PM Modi are now up for e-auction; see how to place a bid

Will AIIMS Delhi Get a New Name? Faculty Write To Health Minister With Concerns

Jignesh Mevani, a Gujarat Congress MLA, was imprisoned for six months in 2016

Loan recovery agent in Jharkhand crushes pregnant woman under tractor

China uses the Pakistan issue at the UNSC and denies Sajid Mir the terrorist label.

Amit Shah and KCR Lead Different Celebrations For The Same Event In Hyderabad


Asiatic cheetahs once lived in India, but the species was deemed extinct there by 1952.

Delhi: MCD introduces new tax-relief programme for refugees from Kashmir

Drugs worth Rs 200 crore found in Gujarat; intended for Punjab and Delhi

Delhi Excise Department investigates disposal alternatives for 70 lakh bottles of unopened alcohol.


UP Dalit Sisters Were Found Hanging From Tree; They Were Raped, Killed, and 6 Arrested

Jaishankar on the India-China disengagement at Patrolling Point 15: "One border issue less."

Regional SCO Summit Starts Today, Talks With China in Suspense: 10 Facts

Taliban claim that the leader of Jaish-e-Mohammed in Pakistan is Maulana Masood Azhar.


BJP's BS Yediyurappa and Others to Face Investigation in Corruption Case, Per Court


Is Kartavya Path dirty? 16 teams are sent out by New Delhi Municipal Council.

Traffic, water shortages, and now flooding: Is India's Tech Hub Dying Slowly?

Mukul Rohatgi: An attorney with extraordinary talents and a lengthy list of clients

8 of the 11 Goa Congress MLAs switch to the BJP, mocking Rahul Gandhi's "Bharat Jodo Yatra"


Complete disengagement between China and India in the Ladakh Hot Springs region: sources


Why the Delhi High Court ordered Amazon to stop selling Rooh Afza produced in Pakistan

According to a Mercedes study, Cyrus Mistry's brakes were applied five seconds before the incident.

India's slavery symbol is gone, according to Prime Minister Modi on the newly renamed Rajpath.

Prior to September 16th, a special court requests that ED respond to Sanjay Raut's bail request.

10% EWS quota: SC says it will look at whether the law violates its fundamental principles.

Soldier's family returns with a gallantry award after it was deemed "protocol violated"

Prashant Kishor's point-by-point response to Nitish Kumar's "Let 12 Months Pass"
11-Year-Old Pit Bull Attack Leaves 200 Injuries On His Face

Regarding the adornment of Yakub Memon's grave, Eknath Shinde said, "Strict action would be taken."

Beginning with Rahul Gandhi, the Bharat Jodo Yatra, full with symbolism and vigour

Anushka Sharma's wishes for Virat Kohli on his 71st century are listed below.


Government influencer standards to promote openness and accountability

4 stocks to buy or sell today, September 8 according to Rajesh Palviya's stock recommendations

Today's 24 carat and 22 carat gold prices in India are down by Rs 210.

Unit of Fitch Group modifies tone on debt held by Adani Group and corrects dat

Delhi's 18 metro stations will feature platform screens for safety, according to an official

Delhi Police conclude their arguments in the HC opposing Umar Khalid's release.

Launch of a three-day workshop on "Science in Kashmiri Language"


MCD announces a directive prohibiting outsiders from entering schools without authorization.

As Central Vista is opened today, Delhi residents should avoid travelling along these routes.

"Heart-Touching Note" from an Indian Oxford Graduate Goes Viral

CreditSights discovers mistakes in Adani Group companies' debt reports.

In 2 Days, 2 Suicide Deaths Have Been Reported From IITs in Hyderabad and Kanpur


Nitish Kumar receives 4 photo-tweets in response from Prashant Kishor. then removes it



Akhilesh offered the position of CM, and Yogi's deputy says "their 100 MLAs are ready to join."

PM Modi will today present Central Vista: Uneven path to the megaproject

NEET 2022 Results: Why is Tanishka the winner while four other students received the same scores?

Security Vulnerability During Amit Shah's Visit to Mumbai; Arrest of Andhra Official

After the Center increases the windfall tax, fuel exporters are concerned about their supplies.

Day trading advice for Friday, September 1st: 4 stocks to buy or sell

CBI deputy legal advisor's body was discovered hanging in his south Delhi home.

SC requests information about FIRs brought against mobster Lawrence Bishnoi

SpiceJet returns with a jet that had a problem.
All you need to know about India's first HPV vaccine for preventing cervical cancer

An environmentally friendly Ganesh idol created in Hyderabad with 17,000 coconuts

Punjab AAP MLA's husband slaps her in front of the camera; the video goes viral

A BJP official close to the Haryana chief minister was killed inside a store in Gurugram.

Write to Congress with the following request: Make electoral rolls public.


Strong Rebuttal from Lt. Governor to Arvind Kejriwal's Accusation of Corruption

Hooda explains why three G-23 leaders met GNA after he resigned, saying, "Told Ghulam Nabi Azad...

Minister for Nagaland posts a picture of the dinner served on the Rajdhani Express.

Bhupinder Hooda responds to criticism for meeting Ghulam Nabi Azad by asking, "Why not"

Supertech declares days after demolition that the Twin Towers' land will be used for.

We are grateful to India for helping us with the IMF deal, the Sri Lankan diplomat said.


First Home-Built Aircraft Carrier in India, INS Vikrant, Commissioned: 10 Points

Delhi's AAP and BJP legislators argue about "Operation Lotus"

Resident doctor groups vow additional protests in response to the NEET PG 2022 Counselling delay

Police Find a Mutilated Body in Indore and a Piece at the Residence of the Accused
India's first cervical cancer vaccine will be unveiled tomorrow: Report

Supreme Court: "Your Cheque, Your Responsibility, Even If Someone Else Fills Details"

Sonia Gandhi's life and future were shaped by Paola Maino.

UN: Xinjiang "crimes against humanity" may have been committed by China.


Your son's master demands "18-hour work" CEO apologises and promises "you won"

Military representatives from India and China meet in eastern Ladakh.

Delhi's old alcohol regulations take effect today: What will they mean for drinkers?

Sidhu Moosewala murder: Gunmen hid in fields, police missed them

Indian Americans in California are subjected to racist abuse

Pawan Khera's jab at Azad over the Gandhis' international travels is "such a trivial..."

IAF Chinooks continue to fly normally despite US fleet grounding

Portuguese minister resigns after pregnant woman dies during hospital transfer.

Aap iske chakkar mein: BJP leaders claim Nitish Kumar and KCR's popular video...

Reliance Capital lenders are presented with 14 resolution ideas

Will domestic flight tickets become more expensive from this day forward?

Campa and Sosyo soft drink brands are purchased by Reliance from Pure Drink

Delhi drug user detained for killing a man for more than Rs 100

India L-G Vinai Kumar Saxena notes a "delay" in the audit at government universities in Delhi


Chinese influence in Sri Lanka is a concern for New Delhi.

Delhi: Within a week, the DU admissions application online will be live.


Delhi BJP says AAP MLAs who claim the BJP offered them money should submit to a lie detector test.

Karnataka court permits the Ganesh festival. Muslim Group Had Fought Back


As Delhi returns to the previous excise policy, new stores and an application for alcohol

umbai developer has warned that you could share Supertech Twin Towers' fate.

UP BJP Leader Expelled From Party for "Buying" 7-Month-Old Stolen Baby


Missing Near China Border: First Arunachal Climber To Scale Mount Everest

91-year-old Mikhail Gorbachev, the Soviet leader who ended the Cold War

BSE and NSE are closed today in observance of Ganesh Chaturthi.



GN Azad explains to the leaders of the "G-23" in Congress why he resigned "without telling"

Food from India could be imported, according to Pak FM. PM Shehbaz Sharif is having an issue.

Hot Stocks | MMTC, SJVN, PCBL can provide high, short-term double-digit returns. Reasons why


Returns on these securities are anticipated to range from 11% to 26%.

During the Reliance AGM, Mukesh Ambani clarifies succession to the shareholders.

The third-richest person in the world and the first Asian to make the top three is Gautam Adani.

DSGMC raises the abduction of a Sikh woman with the Pakistani Embassy.

Pandit Nehru is revived when the museum and library are renovated.



According to forest department data, many trees that were transplanted for Central Vista perished.

Overnight Protests, Songs, and Slogans in Delhi Assembly in AAP vs. BJP

Raj Thackeray Meets with Dy CM Fadnavis in Secret, Fueling Rumors of Alliance for BMC Elections

Security guard in Gurugram was smacked after assisting a guy in exiting a malfunctioning lift.

Rafale issue cannot be brought up repeatedly: Supreme Court

Floods in Pakistan: Modi talks of extending aid


According to reports, Shashi Tharoor is thinking about running for president of the Congress.

According to Moin Khan, Pakistan "Have The Ability To Win" the 2022 Asia Cup.

Rahul Gandhi congratulates team India on victory over Pakistan, saying, "What a thrilling match!"




During the India-Pakistan Asia Cup, Hardik Pandya gives Mohammad Rizwan a hug.

Bank in Delhi was tricked into granting a loan of Rs. 1.9 crore in the name of a deceased woman.

Delhi students discover a grassroots solution to plastic straw pollution

Delhi's AAP and BJP exchange jabs on public schools and the educational system


Firefighting and breaking barriers: Delhi's female firefighters

Regarding the Karnataka Seer Charged with Sexual Abuse, the Chief Minister Said




Arvind Kejriwal will test his majoritarian credentials today in front of all AAP legislators.

Supertech claims that the demolition of the Noida Twin Towers cost it 500 crores.

PM Modi is targeted by Derek O'Brien for having "failed to deliver in 2022... "




GN Azad resigns from the Congress, saying that "Rahul Gandhi, or his guards, took the decisions."

Supreme Court panel indicts the Punjab Police for violating PM Modi's security

WhatsApp and Facebook's petitions against the CCI probe are denied by the Delhi High Court.

By 2025, 80% of Delhi's bus fleet will be electric, according to Arvind Kejriwal.

Supreme Court delays creation of an expert panel on giveaways amid controversy

Factionalism is harming us, but I won't stop campaigning for Congress, says Anand Sharma

Government of Punjab prepares a red carpet for Prime Minister Narendra Modi

Woman from Noida who assaulted and abused security personnel is granted bail


She felt in danger and terrified: Sonali Phogat's brother alleges that she was murdered.

Emergency landing by an Air India flight in Kolkata

Did Sonia Gandhi offer Ashok Gehlot the position of Congress' chief? His Retort

Government didn't comply with investigation, panel finds in Pegasus Supreme Court hearing


Will the SC re-incarcerate 11 convicts in the Bilkis Bano case? today's requests to be heard


Additional Protests in Hyderabad Following BJP MLA's Prophet Remarks Suspended

Supreme Court claims that Praful Patel is attempting to sabotage the competition.



Delhi weather today: Expect heavy humidity, cloud cover, and strong winds.

Conflict between ABVP students and JNU security personnel over fellowship funding

Woman researcher informs Minister that CSIR institutes lack a sanitary napkin disposal system.

After the death of a first-year student, paramedical students had gone on strike.


Delhi: MCD doubles its goal for collecting property taxes and tightens the noose against defaulters

Large gatherings are prohibited in Patna due to teacher protests, Section 144 is in effect.


After widespread protests, a BJP MLA was detained in Hyderabad for making a pro-prophet comment.

Mamata Banerjee's Big Announcement For Durga Puja Celebrations

24-Year-Old Mumbai Resident Dies After Falling From Dahi Handi Pyramid

Man Pushes Woman in Front of Moving Train and Runs With Kids in Front of Camera

Explained: Imran's terrorist accusation and Pakistan's newest political flashpoint


Police: Butcher raped and killed a Delhi girl after learning she had an affair with her mother.

BJP leader on Arvind Kejriwal's statement about Bharat Ratna-Sisodia: "Next Nobel Prize..."

No god is a Brahmin, claims the vice chancellor of JNU, calling out Manusmriti's "gender bias"

Actress and BJP leader Sonali Phogat dies in Goa from a heart attack.

According to the Delhi High Court, citizens must take care not to incite hatred.


SGPC employees abuse an elderly man; they are suspended

Professor in Kolkata "Forced To Quit Over Instagram Photos" after being "Slut-Shamed At Witch Trial"

Alcohol Stunt on Dehradun Road to Land Social Media Influencer Bobby Kataria in Jail

Will respond appropriately when the time is right: On allegations of "jungle raj," CM Nitish Kumar



Those who were unable to vote previously owing to a particular circumstance may now do so.

Terrorism panic when boat carrying weapons washes up on the shore of Maharashtra

India pursues strategic independence, concerned by Russia's "no limit" China tack

Demand To Reverse Bilkis Bano Convicts' Release Has Over 6,000 Signatories

Political parties cannot be stopped from making poll promises: High Court

Delhi experiences 106 deaths per lakh due to PM2.5

Rising swine flu cases in Delhi, with symptoms resembling Covid, according to physicians

Incredible Video of Delhi's Mughal-Era Lake Being Rejuvenated

Terrorist slain after stealing police officer's firearm close to Jammu: J&K Police

Minister's Rohingya Tweet Sets Off Delhi vs. Center Refugee Fight

Farmers in Lakhimpur Kheri, Uttar Pradesh, start a three-day protest against the government.

Reverse this damage and restore my right to life: Bilkis Bano following the release of rape convicts

Ramdev to Delhi High Court: "Don't mislead the public on allopathy."

Bihar Class 9 Student Shot In Neck By Man She Rejected, Caught On Camera

Author Salman Rushdie survived his assailant's surprise.


BJP Top Body: No Yogi Adityanath, Nitin Gadkari, and BS Yediyurappa

Delhi is the most polluted city in the world, and Kolkata comes close behind.

Jacqueline Fernandez, an actress, has been charged in a 200-crore extortion case.


NTPC, Cipla, SAIL, Tata Chemicals, and more stocks are today's stocks to watch.

Man beheads a villager for more than 500 and brings his severed head to the police station.

7 ITBP soldiers are murdered when a bus plunges into a river in Pahalgam, J&K.

6-Year-Old Haryana Girl Murdered, Raped, and Accused Arrested: Police

Truck Falls On Their Car, Killing 4 Noida Executives In Horrific Accident

Bikers injure a woman after stealing her bag from a posh market in Delhi.

Yamuna again reaches danger level in Delhi, and is expected to rise even more

Flight AI from Bangkok to Delhi is delayed due to an alert about lats.

MCD hosts a photo exhibition on Partition in Delhi.

6 family members were discovered deceased at their Jammu home.

Jaishankar on India purchasing Russian oil: "They may not always appreciate it but"

Hours after being appointed, GN Azad resigns from a key position in the J&K Congress.

PM Modi calls Macron and talks about the wildfires in France and global food security.

In response to the death of a Dalit youngster, Sachin Pilot demands action from his own government.

Killings in Kashmir fuel a new surge of dread among minorities.


9 short-term trading suggestions from experts for today, August 16, for stocks to purchase




Why Taliban wants India in Kabul and New Delhi is expanding mission

Vladimir Putin's Independence Day message: "India by right enjoys" India News

after 38 years, a soldier's remains are discovered in Siachen


On the anniversary of the Partition, PM Modi promises to make India a developed nation in 25 years.

After his death, Rakesh Jhunjhunwala's stock holdings worth about $4 billion come under scrutiny.

Chinese ship arrives in Sri Lanka amid worries in India.

4 people were detained in Shivamogga, Karnataka, after a fight over a Savarkar poster.

Both Modi and Manmohan discuss science, youth, and unity on I-Day. however in a distinct tone

Nitish Kumar's Cabinet to Add 31 Ministers, the majority of Who Are From Tejashwi Yadav's Party



The BJP advised Tejashwi Yadav to wait four days, according to a new claim made by Team Nitish.

While Nifty maintains in an uptrend, buy these two stocks for gains; resistance is at 17800.

Delhi: To reduce aircraft turnaround times, Terminal 1 now has 14 new parking stands.

IMD predicts moderate rainfall for 3 days in Delhi.

17 opposition parties want the Supreme Court to reconsider its ruling on the ED's authority.

Bulldozers cannot be used to forcibly evict residents without prior notice: HC Delhi



After five years of effort, the government withdraws the data protection measure.

Rahul Gandhi receives "deeksha" at a Lingayat mutt before the elections in Karnataka.


During her visit to Delhi, Mamata Banerjee may meet the prime minister.

Despite not joining India, the Nagas cannot coexist alone. NSCN (I-M)

On the list of the 11 most violent gangsters in Canada, 9 of the men are Punjabi.

Even the BJP, according to Babul Supriyo, cannot accuse him of corruption.

Cops Outside Sonia Gandhi House, Young Indian Office Sealed: 10 Points

2,073 new cases and 5 covid deaths in Delhi in 24 hours

During Pelosi's visit to Taiwan, US military power puts haughty China in its place.

ONGC, Reliance, Bank of India, Adani Green Energy, and ITC are stocks to keep an eye on.

Top 10 things to know ahead of today's market opening for the stock market

Today's RBI MPC meeting begins: Repo rate increase anticipated following aggressive Fed push

Zomato shares drop following massive block deals, with Uber presumably the seller

Delhi: The Airport Express Line will extend in a month.

Punjab: Competition between SGPC & DSGMC enters next level

CBI books, Delhi 2 DMRC officials for bribery in contracting for fibre optics

Restaurants and bars fight against the turmoil

President Murmu is called by the President of the Maldives at Rashtrapati Bhavan.

24 workers on the Central Vista project were hurt in the Delhi bus disaster.

DSGMC's "Dharam parchar" drive will start today.

Delhi records 1,506 new cases of COVID, the most since June.

Mumbai: A jobless man extorts 12 neighbours from a housing community

After a gas leak at an Andhra garment manufacturing, 100 people became ill.

'Unaccounted' GST number for Arpita Mukherjee's nail salons: 10 points

Indian agencies are on high alert as they concentrate on the next Al Qaeda leader India News


No GST on Cash Withdrawals From Banks, According to Finance Minister: 5 Points

Delhi: An NGO was fined Rs. 5 lakh for filing a twin tower petition.



Authorities at the jail claim that a wristwatch was found in the cell of JNU student Sharjeel Imam.



West Bengal will add seven new districts, bringing the total to 30.


Delhi: AAP government's cabinet note details lessons learned about alcohol policy

Prior to the elections, Arvind Kejriwal made a significant new promise for Gujarat.

Verification Of Income Tax Returns: Significant Rule Change. Find Out More.

After a 10-day hunger strike, Yasin Malik, a prisoner in Delhi's Tihar, ends it.

Killed Pakistani army general; Baloch rebels suspected

A Kabul Safe House: How the CIA Tracked Down and Assaulted Al Qaeda Chief Zawahiri

Comments Made About Sonia Gandhi By Nirmala Sitharaman Are Discredited

BJP begins planning for the Lok Sabha election in Delhi after identifying polling places

Incorrect ITR filing for AY 2022–23: Deadline, Penalty, and Other Information
A Kerala teenager who passed away on Saturday tested positive for monkeypox.

Suspension of 4 Congress MPs lifted; Lok Sabha resumes business: 10 Points

Devendra Fadnavis claims that Sanjay Raut's prosecution by the ED is based on evidence.


Judges cannot be targeted beyond the Supreme Court.

Mahua Moitra yells, "Silly souls!" during the tandoori chicken dispute at the Parliament complex.


Final 11th Marksheet on August 3, 2022 HSCAP Kerala Plus One Trial Allotment Result

After two ED raids, three TMC leaders petition the party to fire Partha Chatterjee.

young person was hacked to death in another gruesome crime in Mangaluru.

Two pilots perished in the crash of a Mig-21 near Barmer, Rajasthan.



SSC Scam: India Under Modi Hates Corruption, TMC Must Clean House to Compete with BJP



Sonia Gandhi Issues an Apology for Her "Rashtrapatni" Remark, Listing 10 Points

Yasin Malik, a jailed Kashmiri separatist leader, was treated in a Delhi hospital

Fashion with a hint of tactile geometry

At SBI New Delhi Circle, Kalpesh K. Avasia takes over as Chief General Manager.

Goa government will conduct a "bhoomi poojan" and send soil to the new Delhi Parliament building.


Sonia Gandhi was prompt in her response to the question, which may end today: Sources

60 Residents of the Home District of the New President Eat Lunch At Rashtrapati Bhavan

During a flood-like condition in Rajasthan, 4 children drown and trains are cancelled.


Fourth death in two weeks involving a schoolgirl in Tamil Nadu


Delhi monkeypox case: What is known about his health and symptoms

One day after Aide's home was raided for $20 Crore in cash, a Bengali minister was arrested.

I-T return deadline of July 31 won't be extended: Govt

CBI now oversees the investigation into two instances involving BJP officials, including Fadnavis.

Shinde supporters' latest assertion: "Got a call from Uddhav when Eknath received a Naxal threat."

Killers were in Punjab despite extensive searches following the murder of Sidhu Moosewala.

Live CBSE Class 10th Results: Latest Updates on Date and Time



Shashi Tharoor digs in as the Paneer Butter Masala trend on Twitter.

Days after the Shiv Sena coup, Sharad Pawar dissolves all party units.


Delhi court releases Ansals in the case of evidence tampering


"My heart will always belong to Delhi"

PWD will renovate three crucial flyovers for easy travel between Central and Outer Delhi.

Delhi's Blue Line metro services experience a technical issue.

Prophet statements controversy: Supreme Court rules that Nupur Sharma won't be detained at this time

Justin Beiber will perform in New Delhi on October 18 as part of his "Justice" world tour.

Gang of 4 nabs MLA cabinet seat in Maharashtra for Rs. 100 crore.
Kerala problems SOP for treating and isolating cases of monkey pox


Center cancels popular science test due to cost KVPY

Kerala Won't Apply GST To Basic Goods Sold By Small Shops, Says Minister

SC prevents arrest of Nupur Sharma in connection with blasphemy FIRs

India-China likely to see fall-back of military from Patrolling Point 15: Govt sources

Commerce Ministry Announces Rules for Working from Home

Girls Asked to Remove Bra for NEET Exam, 5 Women Arrested: 10 Points



Leader of Sri Lanka's opposition appeals to PM Modi before of the vote


Today's gold price: Prior to the ECB meeting, yellow metal increases as the dollar declines.

Companies with international obligations scramble to mitigate currency risk

In Delhi, about 1,400 bus shelters will receive a computerised makeover.

HC strikes down AIIMS admission standards, calling them "unreasonable"

Teachers from DU join AAP because they "want a good relationship so we can work together"

Nehru Place in Delhi is becoming worse due to potholes and waterlogging.

Delhi: Ex-NSDC officials and a Varanasi-based organisation are arrested for embezzling money

Delhi Confidential: Being cautious


Droupadi Murmu hails the NDA lawmakers and claims the tribal community is "excited."

CM reminds Modi of the time the US refused him a visa while his Singapore trip remains in limbo

Yashwant Sinha criticises Murmu and implores BJP lawmakers

After running out of money, a US woman fakes a kidnapping in India.


Army Captain & Junior Officer Died in Jammu Grenade Explosion


121 tribe members arrested in 2017 Red assault that killed 26 CRPF personnel was cleared

Sanjay Raut, the vice presidential candidate of the opposition, will receive support from Shiv Sena

Telangana CM KCR After Flood Review: "Foreign Hand" Behind Cloudburst, Need to Protect People

Acting President of Sri Lanka announces a state of emergency

Yashwant Sinha trails Droupadi Murmu by a wide margin in the presidential polls.

Rashmika Mandanna blushes when Anupam Kher requests to take their photo with his phone.

Today's Aquarius horoscope for July 16, 2022: More money will come in!

Why Katrina Kaif chose Vicky Kaushal as her husband for five reasons.

Multibagger stock rises to a new record as the board shortly considers a bonus issue.

Yes Bank chooses JC Flowers to buy $6 billion in bad loans.


Today's 24 carat and 22 carat gold price in India is down by Rs 160.


Before the G-20 conference in 2019 there will be a renovation on 41 stretches of Lutyens' Delhi.

After 4 years of dating, a man accused of rape is granted bail by the SC

In 2022, the ATMC Education Group will hold the Industry 4.0 India Conference in New Delhi.

RBI action in the forex market prevents the rupee from falling below 80.

Pakistan "journo-spy": BJP intensifies assault, Hamid Ansari reiterates denial

2 people are placed on remand after the Moose Wala killing.

Ranil assumes leadership for the time being and prepares the way for the next President.


Sri Lankan cricket player claims to have waited in line for petrol for two days.

Ex-diplomat: US sanction waiver is a crucial step in India's acquisition of Russian weapons

IITs dominate rankings, IISc leads universities, and Delhi colleges shine in the NIRF Ranking 2022


10 things to know about the 296-kilometer Bundelkhand Expressway before PM Modi opens it

Review of Shabaash Mithu: Taapsee Pannu in Top Form Explains The Mithali Raj Story

Prathap Pothen is no longer acting

Chiranjeevi's home hosts a screening of Laal Singh Chaddha




Government checks aside, Chinese phone brands in India are conducting business as usual

Top 10 things to know ahead of today's market opening in the stock market

SBI increased lending rates beginning today. Loan EMIs will increase

Stocks to Watch: ACC, Adani Ports, Dabur, L&T Infotech, Tata Elxsi, and Adani Ports

Delhi's five-year plan to fight a farm fire with fire
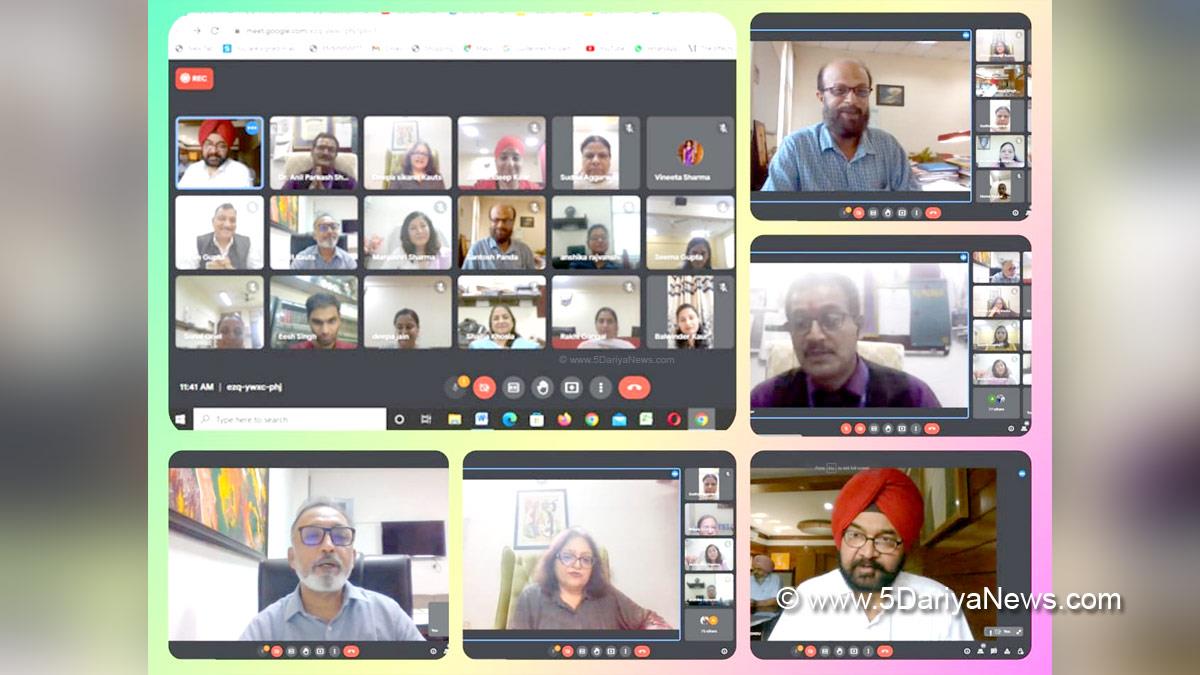

Delhi court asks how many individuals were insulted by Mohammad Zubair's tweet.

Associate Professor Suspended by Delhi University for "sexually harassing" Female Teachers


Sanjay Raut: Shinde-Fadnavis government is unlawful and won't serve out its full term.

Tihar went to court after Sharjeel Imam fabricated up an assault to get him released on bond.

MHA issued a warning for Kanwar Yatra and suggests that extremist forces may pose a threat.

Parliamentary vocabulary from Mahua Moitra: "Amritkaal" rather than "eyewash"

No terms are forbidden: Speaker Om Birla is in a dispute about "unparliamentary" language


BJP supports new allegations made by Hamid Ansari's attorney: "He was upset when"

I2U2 has established a constructive agenda and a useful example of practical cooperation. P.M. Modi

Aviation Organization Requests Transgender Trainee Pilot Reapply for Medical Examination

New "Chief Economic Astrologer" to be appointed: Chidambaram's jab at Sitharaman

Road from Dhaula Kuan to IGI airport to be upgraded aesthetically

As temperatures drop, more rain is predicted for Delhi city.

Due to an increase in dhaba and restaurant licence fees, eating out may become more expensive.

Actor Rhea Chakraborty Has Been Arrested In A Sushant Rajput Drug Case

Lal Bazar Attack: A police officer died and two other people were hurt


"Shortcuts Lead To Short Circuits," says the PM in reference to the Jharkhand opposition

Busiest airline route in India is from Delhi to Mumbai. Which is the second-busiest?

Uddhav Thackeray supports Droupadi Murmu amid rumours of a possible Shiv Sena split.


Anupam Kher on the controversy over the national anthem: "Lion will, of course, show its teeth."

Congressman: "Droupadi Murmu Represents Very Evil Indian Philosophy"

President Rajapaksa of Sri Lanka Takes a Military Plane to the Maldives: 10 Points



Ranveer Singh Gets Rid of For Kissing Man vs. Wild Host in "Bear Grylls' Scariest Moment"

BEYOND THE STAR & more BTS MONUMENTS titles are revealed as HYBE gets a Disney agreement.



8 stocks to purchase today, Tuesday, July 12th, according to the day trading guide

PM Modi will inaugurate GIFT city's SGX Nifty futures trading.

Today's price rise for 24 karat and 22 karat gold in India is Rs 30.

HCL Tech, Delta Corp, 5paisa Capital, and Other Stocks to Keep an Eye on Today

2 top JEE students from Delhi main miss the 100 percentile by a little margin

'Lenient' dowry-killing order: Sacked judge receives no reprieve from the SC


Supreme Court rules that gangster Abu Salem must be released after serving his 25-year sentence.

Delhi Transportation Department intends to install high-definition CCTV cameras at each bus stop.

Will it rain today in the capital city of India, Delhi? the most recent IMD forecast

Reverse the increase in electricity rates, claims Delhi Congress

Experts agree that Canada must strengthen its strategic connections with India in the Indo-Pacific

Ashok Gehlot: "BJP Must Specify On Alleged Links With Udaipur Murder Accused"



To redo ward lines, the delimitation panel will use digitalized maps and recruit MCD workers.

IndiGo's technicians take sick leave in Hyderabad and Delhi after the cabin crew.

Visit this page for more information about the Kashmir trip package that IRCTC has announced.

Supreme Court demands that Abu Salem be released.

TMC and Mahua Moitra's argument over the Kaali poster has escalated.


After a two-day hiatus caused by the cloudburst disaster, the Amarnath Yatra has resumed.

PM Modi unveils the national anthem atop the new Capitol structure.

To prevent mutiny, the Goa Congress wants the Speaker to disqualify two of its MLAs.

Supreme Court sentences Vijay Mallya to 4 months in prison for contempt

Rajapaksa, the absent president of Sri Lanka, returns to duty and directs the distribution of gas

SC requests that Sena MLAs' requests for disqualification be tabled by the Maharashtra Speaker.

Government to receive $12 billion from tax on crude in remaining fiscal year: Moody's


Delhi: Azadi Park construction will begin in August and contain new attractions.



Kanwar Yatra registration has been put up the Delhi Police.

Delhi Municipal Corporation will work with NGOs on educational projects.

Judges and ex-bureaucrats condemn the Supreme Court's comments about Nupur Sharma

RJD leader Lalu Prasad Yadav will be flown to the AIIMS in New Delhi.

Delhi: Nigerian national gets 10 years in prison but is cleared in narcotics case

Artwork and Indian ethos will be featured in the new parliament.


Delhi weather update: From Wednesday, there will be heavy rain in Delhi, according to IMD.

Eknath Shinde responds to Uddhav's jab at the "auto driver" by claiming Mercedes fell behind.


Twitter sues the Center in court for some material removal orders



Explained: Alluri Sitharama Raju's fame and the political import of his legacy

LeT militants were captured by J&K locals after a night of vigilance and morning of capture.

Chief Minister Eknath Shinde Cries Out "Two Of My Children Died"


Heavy rains cause alerts in 6 Maharashtra districts; traffic in Mumbai is interrupted
NTPC, D-Mart, JSW Steel, HDFC twins, and auto stocks are among the stocks in the headlines.

JPMorgan criticises ONGC as its shares decline further following a windfall tax.


Stocks to Watch: NMDC, Indigo, Hero Moto, Glenmark, and the HDFC twins

RIL shares: What brokerages are saying following the revelation of the windfall tax

DGCA requests a report after over 50% of IndiGo flights are delayed because of ill crew members.


MCD and DDA have been instructed by Delhi L-G to plant 10,000 chandan trees.

Residents in medicine in Delhi continue to wait a month for their salaries.


Delhi-NCR housing sales decreased 19% from April to June: Anarock

Scramble and other indoor activities will be introduced in Delhi's primary school classrooms.

What the election of the Speaker of the Maharashtra assembly reveals about trust votes

According to Union Home Minister Amit Shah, the BJP will rule for the next 30 to 40 years.

Explanation: The GST Council decides on modifications to tax rates.

Murmu will travel to Ranchi today to solicit support for her presidential campaign.

Today, Punjab CM Bhagwant Mann will increase his cabinet.

Lashkar Terrorist Caught, Was Jammu BJP's Minority Morcha IT Cell Chief

Shooting in Denmark mall leaves 3 people dead and 3 badly injured.

French Safran offers to participate with the AMCA project and establish an engine MRO in India.

As a bus falls down into a gorge in Himachal, 9 people, including schoolchildren, are killed.

PM Modi's advice to the party: Speak up for the underprivileged in all communities

"Never Demanded Chief Minister's Post," Says Eknath Shinde

Sensex starts July off poorly, losing 800 points, and Nifty50 is below 15,600.

Bookings for the Toyota Hyryder SUV are now available.



30 people were saved in Delhi after a building in Paharganj partially collapsed.

PM Modi kept his anger over the accusations to himself, said MoS Muraleedharan at a book launch.



Amartya Sen, a Nobel laureate, said on the state of India: "There is reason to be frightened."

Salary workers should be aware of the following essential new labour laws:

Modified Three foreign satellites are launched into orbit by PSLV

Supreme Court: Suspended BJP leader Nupur Sharma should "apologise to country"

Bal Thackeray's Legacy Is Revealed In Eknath Shinde's Latest Twitter Picture

Unknown complainants; SC informed that their complaint resulted in the demolition of the UP

Chief Minister Eknath Shinde's initial action satisfies a key BJP demand: 10 points

Mumbai has experienced waterlogging as a result of the heavy rain; an Orange alert has been issued.


Delhi Jal Board: Reduced water in Yamuna and canals may impact Delhi's supplies.

PM Modi will take part in today's "Udyami Bharat" programme.

GST rates updated: What's cheaper, what's more expensive? Here is the complete list.


Raj Thackeray tweets the day after Uddhav Thackeray is removed, "Therein begins deterioration."




2022 Mahindra Scorpio vs. Tata Safari vs. XUV700 vs. MG Hector

Top 10 things to know before the market opens today for stocks

After son Akash takes over Jio, Mukesh Ambani's daughter Isha will play a significant role.

By looking beyond India and building, Pallonji Mistry's "Phantom of Bombay House"

MHA holds an MCD review meeting


At Delhi Police Headquarters, a cultural event was arranged to honour the artist Raja Ravi Varma.


Three students in Hyderabad commit suicide after failing their Inter exams.
Here is the direct link to the Punjab Board PSEB 12th Result 2022.

After being suspended, a Mohali cop who shot at a man during a fight was arrested.

What Simranjeet Singh Mann's return, a fervent supporter of Khalistan, means for Punjab politics

Pre-packaged food is subject to GST, while hotels have a 12 percent lodging tax up to Rs 1,000.



Experts say there is less of a risk of massive, destructive Covid waves in India.

Chief Sena Rebel: Will Return To Mumbai Tomorrow "To Complete Formalities"


In Udaipur, two people kill a guy on camera, disrespect the Prophet, and threaten PM Modi.


Delhi has not yet experienced monsoon; IMD will confirm when it will.

Emergency Landing by ONGC Helicopter in Arabian Sea; 6 So Far Rescued


My tribe vs your tribe: Chhattisgarh Congress seeks a solution to the Murmu problem


Delhi court files a rape complaint against Sonia Gandhi's personal assistant

Over 22 lakh people are affected by the Assam floods, and the situation in Silchar is dire.


FIR Filed Against Zubair of Alt News for 'Highly Provocative, Deliberate' Post

Mumbai: Building collapse in Kurla leads in 2 dead and 11 injuries

PM Modi meets counterparts from the UK, Japan, and Italy during the G7 Summit in Germany.

After Supreme Court relief, Sena Rebels Examine No-Trust Vote: 10 Points


Check out the most recent rates in Cuttack and Bhubaneswar here for gold and silver.

Indicators show that the economy picked up steam in May thanks to pent-up demand.

IKEA has drawn large crowds to Bengaluru's Nagasandra metro station.

Firefighters in Delhi put out a fire in a three-story building using robots.

VK Saxena, the lieutenant governor of Delhi, requests that the baoli in Sanjay Van be restored.

Today's SC session will concern rebel MLAs from Maharashtra's appeal of disqualification letters.

JNU administration implores canteen proprietors to pay debts

Rewari: Panchayat grants Haryana government two weeks to purchase land for the AIIMS.

HC publishes alerts about the lack of facilities in the capital for rainwater collection


In Uttarakhand, 201 pilgrims perished during the Char Dham Yatra in fewer than 60 days.



CM Naveen Patnaik chats with Murmu and reiterates his efforts to get all of Odisha's votes for her.

In Haridwar's Roorkee, a woman and her 6-year-old kid were groped in a moving automobile.

Eknath Shinde makes a call. Despite a worsening political crisis, Raj Thackeray

Man-made flooding in Silchar; embankment breached, says Assam's chief minister


Regarding China's new aircraft carrier Fujian, the Indian Navy has concerns.





Zomato will purchase Blinkit for $4,447 crore in an all-stock transaction.

Delhi's morning is pleasant, with a low of 24.3 degrees C.


New Delhi ambassadors are welcomed back by the Taliban.


Most Covid booster doses are administered in West Delhi, with greater coverage in New Delhi.


Is Lawrence Bishnoi the mastermind or a pawn in the Sidhu Moosewala murder plot?



8 SFI employees were detained and Rahul Gandhi's Wayanad office was vandalised.

According to estimations, rebel Shiv Sena MLAs live affluent lifestyles in Guwahati.


Handler of the 26/11 Mumbai terror assault is imprisoned in Pakistan for 15 years.

After 50 years, the US Supreme Court ruled against Roe v. Wade. Here's Why

Sena MLA Kailas Patil said, "I Flew Away, Some (MLAs) Are Signing Under Pressure."

New discovery announced by ONGC Videsh in Colombia


Indian rupee rises 12 paise to open at 78.20 against the US dollar after hitting a record low.

Trouble for Indian stock markets is unlikely to end soon, according to three charts.

In a second round of layoffs, Netflix fires hundreds more workers.

Nitin Paranjpe, chairman of HUL, warns that the economy is in its worst possible state.

Delhi: Three more months of navigating the maze of ashrams

In a bank fraud case involving a Delhi company, the CBI is searching five premises.

Couple granted protection by Delhi High Court in criminal breach of trust case

A request in the Delhi High Court to change the call sign "VT" on Indian aircraft

10-year-old Delhi boy perishes while doing a stunt from a video while skipping rope.

Rain is expected in Delhi from June 27 to 30.

PWD to fix important roads in East and Central Delhi; bids have been floated

Agnipath is a dark plot, says Khaps

After 2024, India would have 50 states, according to a minister, with Karnataka splitting into two.


Karnataka High Court: If the abuse is not in public, there is no case under the SC/ST Act.


Modi is being told by Xi to give up the Cold War attitude and bloc hostility.
India's daily count had 17,336 cases, which is a 4-month high: Updates for Covid today

For the third day in a running, the Jammu-Srinagar national highway is closed.


Lawrence Bishnoi gang assassinated singer Sidhu Moosewala on their third try.

JMM leans to put weight behind Droupadi Murmu, which causes Yashwant Sinha to halt in his tracks.

Agnipath scheme: Air Force agniveer registration is now open. Check specifics

At a crucial meeting, OPS encounters taunts and bottles while AIADMK is in disarray.

Droupadi Murmu is supported for president by Andhra Chief Minister Jagan Reddy

NDMC proposal to declare Kejriwal's seat vacant

Delhi: A man perishes as his motorcycle rams a metro station glass panel.

50 "illegal constructions" in the ITO region are removed by MCD

Delhi requests that only BS VI-compliant buses be sent beginning in October.

Why Railways are a focal point of demonstrations, from Farm Bills to Job Protests to Agnipath

Man Punished for Kissing Wife While Bathing in Ayodhya River on Camera

Flooding in Assam: Slightly better, but Silchar is still in serious need

Due to corruption, Delhi's Lieutenant Governor has suspended Arvind Kejriwal's deputy secretary.

Legal demolitions unrelated to demonstrations: the Supreme Court

2 educational institutes in Bengaluru are the target of I-T raids for alleged tax evasion.
38 fatalities from COVID have been reported in India in the past day.

Eknath Shinde's uprising is due to Uddhav Thackeray's actions | OPINION

Tamil Nadu's EPS suffers a late-night setback as the court halts the party takeover bid

Devastating Afghanistan Earthquake Leaves At Least 1,000 Dead

Have respect for Murmu, but the conflict is one of ideologies: Yoshwant Sinha

According to the US, India and China may be purchasing more Russian oil than we think.

Droupadi Murmu was well positioned thanks to JD(U) & BJD.


Today will be rainy in parts of Delhi and Noida; temperatures will be three degrees below normal.

According to Prime Minister Modi, yoga brings serenity to our universe.

Uddhav and Aaditya pushed me to bring Waze: Param Bir back to CBI.

Yashwant Sinha Says It's 'Time to Step Aside' From TMC Amid Presidential Candidate Buzz

Experts advise people to follow SOPs in the face of an increase in COVID.


Gopal Gandhi also declines to be the opposition's presidential candidate at the last minute.



PM Meets With 3 Service Chiefs Today Amid 'Agnipath' Row: 10 Facts




'Shamshera,' starring Ranbir Kapoor, will be released on July 22. Check out the first look poster.


Taapsee Pannu's Mithali Raj Redefines "The Gentleman's Game" in Shabaash Mithu Trailer



Six new Chief Justices have been appointed to the High Courts.

JPMorgan becomes positive on India's 'cheapest, biggest' stock.

Vedanta is selling the Tuticorin copper facility.

Nifty50 may be preparing for a pullback, according to the trade setup for June 20.

Report: Air India is preparing one of the world's largest aircraft deals.

Load off the Delhi highways for a faster travel to Noida and Ghaziabad.


India's past, present, and future are all documented.

After a traffic incident escalates, a JNU professor is 'abducted and attacked.'

In Delhi, Prime Minister Narendra Modi opens the Pragati Maidan tunnel and five underpasses.

Haryana MC poll: Elderly and female voters come to Naraingarh booths

ONGC official dies in a pile-up near Panvel in Navi Mumbai.


In Aligarh, Uttar Pradesh, a cop and his wife fell down a drain on their scooter due to flooding.

'Bypoll an opportunity to halt Azamgarh from becoming aatankwad ka garh,' says Yogi Adityanath.

Team Uddhav Thackeray's New Contest With The BJP Is Stung In Rajya Sabha Polls

1 238 people have been arrested in India as a result of the Agnipath demonstrations.

Congress's 'glow and nice scheme' dig before Rahul Gandhi's ED questioning

PM Modi Responds to 'Agnipath' Blowback: "Misfortune That Many Good Things..."
Making Sense of the Covid Spike (explained)

SpiceJet praises its Patna-Delhi pilots, saying, "We're proud of them."

Live Updates From The 'Agnipath' Protests: States On High Alert Amid Bharat Bandh Call

In case you missed it, Kim Sharma wished "Soulmate" Leander Paes a happy birthday.


Shilpa Shetty's reappearance makes you fear for Bollywood's future in Nikamma.

Kiara Advani on Personal Life Rumours: "I'm Not Completely Immune To It"

Netizens savagely criticise filmmaker Kartik Aaryan as he ignores Karan Johar during an award show.


Rakesh Jhunjhunwala loses approximately 560 crore in a single day in this Tata stock.

Several staff at SpaceX have been fired after criticising CEO Elon Musk, according to a report.

Digital payment transactions are expected to triple by 2025, according to the RBI's Vision Document.

On June 18, check out the new gold and silver rates in Bhubaneswar and Cuttack.
As inflation rises and hawks howl, the Sensex and Nifty have had their worst week since May 2020.



The Indian Journalists Union has elected Vinod Kohli as its president.



The Delhi Transport and Metro Corporation are planning to build "world-class" bus terminals.

'There is no shortcut for remedies in criminal proceedings,' says the Delhi High Court.

In New Delhi, a logo commemorating India-Vietnam diplomatic ties was unveiled.

After Friday's rain, Delhi's maximum temperature drops to its lowest level in 20 days.

Warring cries vendetta politics when a former Congress MLA is arrested for illicit mining in Punjab.

PM Narendra Modi will raise the flag at the newly refurbished Pavagadh temple in Gujarat.

In Assam, 31 people have died in floods, while 3,000 villages in Meghalaya have been flooded.

MEA is watching the situation after IS links were alleged in the Kabul Gurdwara bombings.


The Agnipath scheme: Why may age relaxation be a problem?

'Agnipath' Protests: 12 Trains Burned, Stations Vandalized: 10 Facts

The Centre announces more concessions as the 'Agnipath' protests spread.

Terrorists assassinate an off-duty cop in Pampore, Jammu and Kashmir.

ISIS terrorists opened fire inside a Kabul gurdwara, causing Sikh casualties.


'It's great to see the BJP publicly condemning insulting remarks about the Prophet,' said the US.

Renuka Chowdhury grabs a cop's collar during a violent Congress protest in Hyderabad.

2 Passenger Train Coaches Set On Fire In Bihar During 'Agnipath' Protest






Asian Paints, PNB Housing, Engineers India, and other stocks to keep an eye on today


Vinay Rajani believes that once the Nifty hits 15,858, it would turn positive.

Paytm's stock is up 471 percent after a jump in loan disbursements in April and May.

Cryptos and NFTs are dismissed by Bill Gates as being based on the "Greater-Fool Theory."

In early trade, the Indian rupee rose 5 paise to 77.99 against the US dollar.

Nifty50 manages to defend 15,700 on June 15, but may not bounce back quickly.

In Delhi, the weather forecast calls for mild rain for the next six days.



Ex-judges write to the CJI about "state repression" in Uttar Pradesh.

People Spend Sleepless Nights in Sweltering Heat Due to Power Outages in Several Parts of Delhi

BJP Leader: "Solve Dispute in My Lifetime" Approach Won't Work With China

Air India gets fined Rs 10 lakh by the DGCA for violating compensation rules.

A portrait of the man who murdered Beant Singh and 16 others has been displayed in a Sikh museum.


In the midst of a custodial torture charge, UP cops deny knowledge of the "Return Gift"

Chief Minister KCR's Party Skips Opposition Meeting: "Congress Ganging Up..."

I beg your pardon for this: Kiran Bedi apologises after receiving flak for her 'Sikh' remark.

In J&K, Jamat-affiliated Trust schools will close.

'I spent 104 hours with a snake. 'A brave boy,' says Chhattisgarh's chief minister. 11-year-old


Left-wing politicians meet with NCP chief Sharad Pawar before of the presidential election.

Kashmir Police say a terrorist involved in the murder of a bank manager was shot and killed.



Day 2 of Rahul Gandhi's ED grilling, after much drama and detentions: 10 points


Heinrich Klaasen explains how he controlled a thorny Cuttack pitch.




What Shilpa Shetty's Mother Told Big Brother Producers About Her Son's Appearance


Ranbir Kapoor speaks candidly about his marriage to Alia Bhatt: 'It's still a mystery to us...'

5 Things To Know About Shakti Kapoor's Son Siddhanth Kapoor, Who Was Arrested For Drugs


As anchor investors' lock-in period expires today, the LIC share price falls to a new low.

The Indian rupee has hit a new low, with four factors acting against it.

Amazon's Anti-Future Petition Is Rejected; A $200 Million Penalty Is Imposed

Delhi High Court has rejected a plea accusing the AFP of racial discrimination.



In Delhi, a hand grenade was discovered in a canal.

The Supreme Court will rule on a petition for a hate speech FIR against BJP leaders today.

Residents in parts of Delhi are irritated by power outages.

Here's why it's good news for Delhi that Jaipur and Chandigarh are getting closer.

Minimum temperature in Delhi has reached a four-year high of 32.8 degrees Celsius.

BJP has written to Delhi's Lieutenant Governor, VK Saxena, requesting more fire stations.

A vision for a future that is more accepting of LGBTQ people

Until Covid halted the AI effort, around 400 SC rulings were translated.

A massive fire breaks out at Gaffar Market in central Delhi, injuring one firefighter.


In connection with the protests at the Jama Masjid in Delhi, two people were arrested.

The Prophet speaks about India's situation... Why should we start a discussion: Bangladesh

The significance of the Sant Tukaram shrine is explained.

Asaduddin Owaisi Slams Demolition: "UP Chief Minister Has Become..."
Kuwait is planning to deport expats who protested against the Prophet's remark. This is why:

Demolition, arrests, and periodic violence keep governments on their toes.


The father-daughter duo at the centre of the devastation of Prayagraj


India's life expectancy has increased by two years to 69.7 years.

VHP claims there was a well-planned conspiracy behind the violence in Kanpur.

Pune Rural Police arrest a Gujarati suspect in the Sidhu Moosewala case.


Mobs attack trains in Bengal, arms seized from Prayagraj conflict accused's razed home | Top Points


Rahul Gandhi Is Summoned: Congress Protests, Sister Priyanka To Join Him




'It will go back to normal,' Justin Bieber said of his partial facial paralysis.

Sanjay Dutt's cancer tale inspired Mahima Chaudhry, who describes her experience as "tough."

At Nayanthara and Vignesh Shivan's wedding, Shah Rukh Khan was photographed with Jayam-Aarti Ravi.

For banks and fintechs, the RBI's UPI-credit card connecting plan isn't adding up.


According to the government, the drop in the stock price of LIC is a one-time blip.

Asola's Neeli Jheel will be developed as an ecotourism hub.

Punjab's chief minister has announced the launch of super-luxury buses to the New Delhi airport.

The Supreme Court has rejected a petition for special counselling for 1,456 NEET-PG seats.

Under the supervision of IRP, NCLAT allows Supertech arm to complete all tasks.

Days with high heat in April are up 122 percent from the previous year in Delhi.

Haryana should release Delhi's share of water, according to the Delhi Jal Board.

The Delhi High Court has ordered that the Yamuna be cleaned for 45 days.

Delhi is at 43.5 degrees Fahrenheit, four degrees above normal, with no sign of relief in sight.

A man she met on a dating app raped a Delhi woman in a hotel.

Police will conduct potency tests on all accused in the Hyderabad gang rape case.


In Bengaluru, a BJP MLA's daughter'misbehaves' with traffic cops in order to impose a fine.

The Delhi Police will issue notifications to social media companies for making hateful comments.


In Karnataka, masks are required in all enclosed spaces.


PM Modi's Former Teacher Was Successful In Getting A Meeting With Him

AIMIM's restrained response to a party MP's demand for Nupur Sharma's execution.


Kumaraswamy, bruised, rants at the Cong and vows to never work with it again.

Here's why Congressman Ajay Maken lost: 1 abstention, 1 invalidation, and 1 cross vote

The BJP Scores a Big Win in Maharashtra, but Loses in Haryana: 10 Facts


Gangsters in Tihar call the shots in the Sidhu Moosewala murder.

Experts estimate that by 2031, Delhi would require an additional 530 MGD of water.


In the last 48 hours, there have been six fires in Delhi.


The ABVP is hosting free classes taught by scholars for DU applicants.

Staff will be trained by the MCD to respond quickly to floods.








Bengaluru VIP arrogance: BMW halted, MLA Aravind Limbavali's daughter assaults cops

Cops on link to mobster Lawrence Bishnoi in Salman Khan threat letter case


IIT-Delhi rises 11 places to 174th in the World University Rankings, while DU and JNU fall.



On July 18, the next President will be elected, and the opposition will face off against the NDA.

With a win in the Nations League, the Netherlands put an end to Wales' World Cup celebrations.


India vs. South Africa T20I: KL Rahul, Kuldeep Yadav ruled out; Rishabh Pant named captain


Coal use as a fuel in Delhi-NCR will be phased out by the end of the year.

Zafar Mahal, the last Mughal palace, is in need of repair and restoration.

A fire breaks out near Batla House in Delhi, rescuing over 20 people.

Water supply will be disrupted in several areas of Delhi today.

Today, Prime Minister Modi will kick off the Biotech Startup Expo 2022.

Coal will be phased out as a fuel source in Delhi-NCR by the end of the year.

Wary of expansionist China, India and Vietnam sign pact to strengthen defence ties.

Uddhav Thackeray on the Prophet row: India brought to its knees, PM's photos stuck in garbage cans



Delhi cops say Lawrence Bishnoi was the mastermind behind Sidhu Moosewala's death.

Police in Hyderabad want 5 minors tried as adults "for maximum punishment."

India reports 7,240 new Covid cases, a nearly 40% increase for the second day in a row.

One person was killed and 16 others were injured when a three-story building in Bandra collapsed.

China says it is willing to collaborate with India to assist Sri Lanka.

Don't give up your freedom for economic reasons, says a Dutch MP who supported Nupur Sharma.

Case Against Suspended BJP Leader and Journalist 2 Weeks After Prophet Row

BSF returns a suspected Pakistani drone in Jammu's Arnia to search for explosives.


Sonakshi Sinha responds to marriage rumours by quoting Shah Rukh Khan. View this amusing video



In a statement to police, Salman Khan denies receiving threats from anyone.

The RBI intervenes ahead of a policy decision to prevent the rupee from falling to a new low.

RBI Repo Rate Increase! Borrowers will pay more for loan EMIs as lending rates rise.

The RBI proposes to link credit cards to UPI, beginning with Rupay cards.
Delhi: An UPSC dream is dashed, and a family loses a devoted daughter
After a wedding, a man overdoses on Viagra and ends up in the hospital twice.

National Herald Case: Rahul Gandhi Returns to Delhi From Abroad Following ED Summons

Rain in the next few days will bring relief to Delhi, according to the weather forecast.

The Delhi BJP defends L-G in the face of AAP allegations.

The Places of Worship Act is being challenged once more, with the plea citing barbaric invaders.

Kerala Chief Minister named in gold smuggling case


A 16-year-old Lucknow boy shoots and kills his mother for preventing him from playing PUBG.

Roddur Roy, a YouTuber, was arrested in Goa for making 'derogatory' remarks about Mamata Banerjee.

India will not accept unilateral changes to the status quo: Jaishankar


Qaida threatens suicide bombings in Indian cities in the wake of the Prophet Row

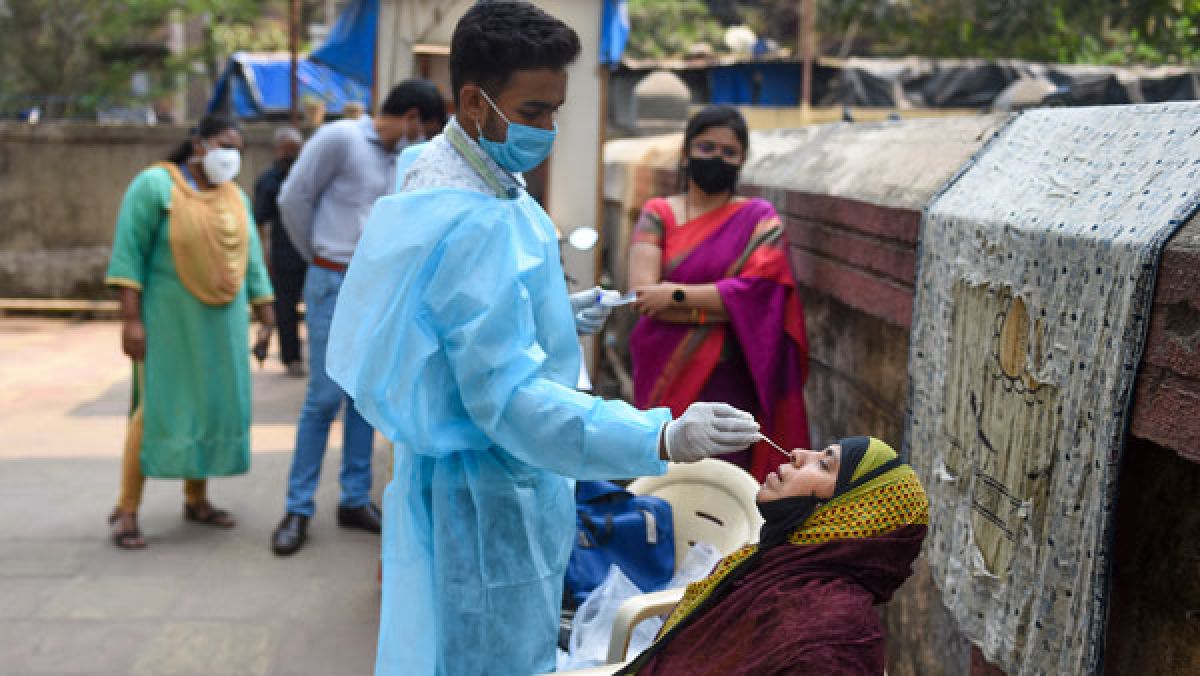
With 5,233 new infections, there has been a nearly 41 percent increase in daily Covid cases.

AIMIM MLA's Son Named As Accused In Hyderabad Gang Rape

A UP BJP functionary was arrested for tweeting about the Prophet.

Today's gold price in India falls by Rs 350 for 24 carat and Rs 350 for 22 carat.



Adani and Apollo are considering bids for a majority stake in Metropolis.

LIC investors lose over ₹1.08 lakh cr since debut against the IPO issue price

Following the FCRA bribery case, the MHA overhauled its Foreigners Division.

In many parts of Delhi, the temperature remains above 45 degrees Celsius.
The punishment of Nupur Sharma and Naveen Jindal has riled a section of the Delhi BJP.

Max Estates pays Rs 306 crore for a 10-acre mixed-use residential land parcel in Noida.

Jan Sunwai of the Delhi Municipal Corporation received 43 complaints on Day 1.

Maharashtra: A panel recommends changing the design of the elevated Thane-Ghodbunder Road.

An old-money billionaire takes on Gautam Adani.


Op Blue Star anniversary: Akal Takht Jathedar discusses churches and youth weapon training



British tourist brutally raped in front of partner near Goa beach: police


Despite the fact that the car was washed, cops in Hyderabad claim "ample evidence."

The killer of Sidhu Moosewala has been apprehend

Sadhu Singh Dharamsot, a former Punjab minister, has been arrested in a graft case.

Corvettes and Su-30 engines: Indian industry receives a Rs 76,390-crore boost in procurement.




The government intends to renovate the New Delhi Railway Station on its own.


AAP accuses BJP candidate in Delhi of running 'illegal' motels.


Recovering a Hyderabad car in which a teen was gang-raped took a week.

Trains will start running starting tonight at Bengaluru's first airport-style railway station.

In Mohali, police commemorate Operation Bluestar with a flag march.

"Modi Ji, What You Permit...": KTR's Swipe in Telangana Amid Arab Backlash

Talib Hussain, a Hizbul Mujahideen militant, was apprehended in Kishtwar, according to J&K police.


Whistleblower Documents Name BJP MP Vinod Sonkar in 'Fake Account' Scandal on Facebook


After 100 years, the 'lipstick' plant has been rediscovered in Arunachal.

The number of weekly Covid cases has surpassed 25,000 for the first time in over three months.

In a gang rape in Hyderabad, the fourth culprit is thought to be the son of a Telangana leader.

Uttarakhand: A bus carrying Madhya Pradesh tourists crashes into a canyon, killing 25 people.

Tathagata Roy, the former governor of Tripura, expressed his sadness over Nupur Sharma's suspension.

Punjab Police Makes Another Arrest in the Sidhu Moose Wala Murder Case


7 people drowned in the Tamil Nadu River after becoming stuck in quicksand.

The first flight from Delhi city to Haj is scheduled for June 5th, following a two-year wait.

The Delhi Municipal Corporation draws up a plan to help flatten landfills and submit a report.


In front of the camera, a Delhi man is assaulted, his head is smashed, and his throat is slit.



In Anantnag, Jammu and Kashmir, a Hizbul terrorist was killed in an encounter.

Sidhu Moosewala's parents will meet Amit Shah this evening in Chandigarh.



During Johnny Depp's defamation trial against Amber Heard, demand for his Dior fragrance surged.


Aashram... Ek Badnaam 3 Bobby Deol's Performance In The Series Is Impressively Solid

Rhea Chakraborty will miss the IIFA 2022 in Abu Dhabi, citing a lookout notice.

As Yogi Adityanath watches 'Samrat Prithviraj,' the opposition sneers: 'Janta ne...'


UltraTech Cement, Castrol, CIL, Aether Ind, and Deepak Nitrite are stocks to keep an eye on.




This weekend in Delhi, expect light rain and thunderstorms.






NEET PG 2022 topper from Delhi, 23 years old, never stopped being the best in her class.

Kerala's Chief Minister has stated that the Citizenship Act will not be implemented in the state.


In Vadodara, Gujarat, a fire breaks out at the Deepak Nitrite GIDC facility. Watch

After KK: A singer's death and a discordant political note


Uttarakhand Chief Minister Pushkar Singh Dhami Wins Critical Bypoll

Rahul Gandhi's meeting with the Enforcement Directorate has been rescheduled for June 13th.

The Gandhi family is the "most corrupt" in the world, according to the BJP.


A Day After Two Murders, the Centre Meets On Kashmir, and Pandits Issue an SOS

'Dangerous than the 1990s,' say government personnel fleeing Kashmir as the death toll rises.




Stocks hold stable as oil prices fall amid talk of increased output.

Splendor, Passion, Destini, Xpulse - Hero MotoCorp Sales May 2022



Delhi's air quality has deteriorated to'very poor,' with dusty winds to blame.

In Delhi, a man was slain when he refused to give up his beer bottles.

Based on a Supreme Court order, a detailed hearing on Sharjeel Imam's bail plea is required: HC


The Delhi High Court rejected an appeal against Akshay Kumar's film due to the king's caste.

Before he became famous, KK was a student at Delhi University.

To facilitate liver movement, the Delhi Traffic Police creates a green corridor.

Haryana MLAs are being summoned to Delhi by the Congress, and they may be sent to the resort.

Vistara has been fined ten lakh rupees for the way an Indore flight landed.

In "Sologamy," a Gujarat woman will marry herself, according to reports.

ExpressVPN rejects CERT-In directives and suspends operations in India.

In May, India's coal shortfall was eased thanks to record renewable energy output.

Arvind Kejriwal, citing "Reliable Sources," claims that Manish Sisodia would be arrested next.

In Kulgam, Jammu and Kashmir, a bank manager from Rajasthan was shot and killed.



Today's Stock Market: Top Ten Things to Know Before the Market Opens

All Parties Support Nitish Kumar After He Clears Caste-Based Census In Bihar

Hardik Patel, dubbed "A Small Soldier," is preparing to join the BJP with a tweet and a puja.

GST receipts for April sales are up 44 percent year over year, which is extremely promising.

The ED summons to the Gandhis, according to Chidambaram, is 'absurd':'money-laundering without...'


A Civil Services Aspirant's Viral Tweet: "I Missed By 11 Marks"

How much oil does the European Union import from Russia, explained?




For India, trade and terror with Pakistan cannot go together

Following PSU opposition, India has put its plan to monetize oil pipelines on hold.

Teachers from the Jammu region who are serving in Kashmir have been stunned by the killing.

In Uttar Pradesh, a farmer leader and witness in the Lakhimpur case was shot.

"Result in 2 Days": A Revenge Threat After Sidhu Moose Wala's Murder


The principal of Kalindi College denies assault claims.

'College of Art should not be allowed to join Ambedkar varsity.'


Teachers at DU have won their pension dispute after a 16-year court fight.


In a major bureaucratic reshuffle, the Lieutenant Governor of Delhi transfers 40 officers.

A storm hit Delhi on Monday, killing two people and injuring a dozen others.


If wheat exporters present back-dated LCs, the DGFT will refer the case to the CBI.

After a family feud, a Maharashtra woman kills her six children by throwing them down a well.

After the Rajya Sabha List, the BJP may not have a Muslim face in Parliament.


PM Modi appreciates pilgrims for keeping religious sites clean during the Char Dham Yatra.

The temperature in Delhi has dropped by 16 degrees due to heavy rain and high winds.


Nepal's civil aviation authority reports that 21 bodies have been recovered from the disaster scene.

"I Relied On My Own Notes, Not Coaching Centers," says Civil Services Exam Winner.

Never buying fuel from OMCs: Petrol pumps

What's next for Satyendar Jain now that he's been arrested by the ED for money laundering?

Sameer Wankhede was relocated to Chennai a few days after NCB gave Aryan a clear bill of health.

Over 4,000 children have received individual letters from PM Modi as part of the PM CARES scheme.


IPL 2022 RCB Review: Royal Challengers Bangalore's Hot-and-Cold Season Continues



Edava Basheer, a Malayalam singer, died after fainting on stage during a performance.



The price of 24 carat and 22 carat gold in India has risen by Rs 260 today.



Nykaa, ONGC, Glenmark Pharma, JSW Steel, and M&M are stocks to keep an eye on.

Ford India's Sanand facility will be acquired by Tata Motors.
Delhi: Many people are having sleepless nights a year after defeating Covid.

In Delhi, the IDRC hosts the 'Arbitrate in India Conclave.'

The Delhi High Court has dismissed Harsha Buildcom's appeal against a one-year sentence.

Hotel and bar retail licences to serve alcohol on premises have been renewed until July 31 in Delhi.

Develop a CP to Airport stretch that follows the Aerocity model: LG Saxena, Vinai Kumar

Officials, please provide a plan for razing waste heaps.

Today's forecast for Delhi is for partly overcast sky with the risk of thunder and lightning.

The development of SARS-CoV-2 has been decoded by an IIT Delhi study.

Get ready, get set, go: the Delhi By Cycle programme

Mission of the Delhi government Buniyaad is creating a firmer educational basis.


Punjab FM Harpal Cheema to Capt Amarinder Singh: Provide proof of corruption.


After a flurry, the government has withdrawn the UIDAI's Aadhaar caution note.

Nirmala and Piyush are on the BJP's Rajya Sabha list; Naqvi and the RCP's destiny is unknown.




India will purchase 26 fighter jets for the INS Vikrant on a G-2-G basis.

Army finds the wreckage of a Tara Air plane in Nepal.





The 'Queer Palm' prize goes to a Pakistani trans drama.



On day 8, Kangana Ranaut's film Dhaakad sells 20 tickets and earns Rs 4420 at the box office.



GAIL is interested in purchasing Russian oil and gas assets.

The circulation of 2000 currency notes continues to decline. What the RBI has said

For Rs 820 crore, United Spirits will sell 32 popular brands to Inbrew.




In the Axis mutual fund front running case, the Sebi searches 16 organisations.

Elon Musk sets a stipulation for Tesla car production in India.

In the mud: Delhi's rainwater harvesting system is broken.



In Delhi, a gang scans and steals cars in under 5 minutes.

iKargos receives new money to help it grow and expand its international footprint.

Applications are being accepted for 36 skill-based certificate courses at the DU Learning Center.

The CUET battle between DU and St Stephen's is still going on.

Sharjeel petitions lower court for bail in sedition case after HC judgement.


The 'inverted rifle and helmet' from India Gate has been relocated to the National War Memorial.

The NDMC has a new chairman, according to the Centre.

Ex-Uttarakhand minister commits suicide just days after his daughter-in-law is charged with murder.

Om Prakash Chautala, who is now 87 years old, is Tihar's oldest prisoner.

The security of 424 VIPs in Punjab has been withdrawn.

PM Ranil Wickremesinghe thanked India for its assistance during a "tough moment" in Sri Lanka.


At the Chandigarh airport, over 4 kilogramme of gold was seized from two travellers.

Vedic Sangh chairman demands a prohibition on disclosing survey results in the Gyanvapi row.

The SC maintains the onerous HC conditions for Azam Khan's bail in the land-grab case.

AH Vishwanath, Karnataka, thinks the textbook revision commission should be scrapped.

In the matter of Rana's arrest, a court panel summons Maharashtra's DGP, CS, and Mumbai's CP.

Cops stood outside a school in Texas while students begged for assistance.

When a vehicle crashes into the Ladakh river, seven troops are killed and 19 are injured.

Over alleged misuse of a sports stadium, the Centre transfers an IAS officer to Arunachal Pradesh.

India slams the Islamic Nations Group's remark on Yasin Malik as "unacceptable."





Hindalco's fourth-quarter profit doubles to Rs 3,851 crore, with sales up 38 percent year on year.

Salil Parekh, the CEO of Infosys, has had his compensation increased by 88 percent to Rs 80 crore.


Shareholders of Twitter have filed a lawsuit against Elon Musk for stock price manipulation.

BPCL, Hindalco, Piramal Enterprises, Tata Power, and Nykaa are stocks to keep an eye on.

In a major shake-up, EY is considering spinning off audit operations, according to a report.

Experts predict a 'par' start for Paradeep Phosphates shares.

St Stephen's says it will continue with interviews and has written to DU.



The Delhi Municipal Corporation (MCD) tries to assuage anxieties about the demolition campaign.

Harsh Vardhan, a BJP MP, quits the swearing-in ceremony of the Delhi L-G after being forced to wait.

The Punjab Congress chief wants the wheat export prohibition lifted.

The petition filed by Hindu plaintiffs is not maintainable, according to a mosque panel.

Navjot Singh Sidhu is being treated with chamomile tea and chia seeds while incarcerated.

There is a strong buzz. Yogi Mantri may be sent to the Rajya Sabha by the BJP.

Karti Chidambaram's Move Against CBI Over Raids Is "Grossly Illegal"

According to a Supreme Court panel, voluntary sex work "is not illegal."

Exhibit A: Chief Minister KCR's Team's Response To The Prime Minister





Has the Delhi Police made a decision on the opening of restrobars until 3 a.m.?
Haryana outperforms Delhi in every subject in Class III, V, VIII, and X.

Justice Chandrachud describes himself as "intriguing." Articles about the GST Council's Decision






Indigo Q4 Results | Fuel costs rise, resulting in a net loss of Rs 1,681.80 crore.

The government has approved the sale of Hindustan Zinc's remaining shares.


In early trade, the Indian rupee is gaining ground against the UAE dirham.

Bookings for the Kia EV6 Electric Crossover have begun in India.


Coal India, Indigo, Vodafone Idea, and Infosys are stocks to keep an eye on.

Buying a new car or a motorcycle? Prepare to spend more money starting in June; here's why.


In Delhi, the DSIIDC will establish e-vehicle charging stations.


The weather in Delhi is expected to be clear with a high temperature of 37 degrees Celsius.

After two days, the Covid positive rate in Delhi falls below 2% once more.



The BJP has filed a breach of privilege complaint against Atishi Marlena in Delhi.


In Delhi, scorching heat and limited supply have resulted in a shortage.

At Umar Khalid's bail hearing, the'veracity of witnesses' statement cannot be challenged.



PM Modi is expected to arrive in Hyderabad, while KCR is expected to depart.

A dead lizard is found in a McDonald's drink.

Yogi Adityanath Steps In After Akhilesh Yadav Uses "Indecent Words" In UP Assembly

Quad pledges $50 billion in Indo-Pacific investment to fight China's clout



In a money laundering investigation, a Maharashtra minister's home was searched.

Three Lashkar terrorists were killed in Kupwara, J&K, when an infiltration attempt was disrupted.

The pyrotechnics of Harmanpreet and Shafali help to improve the image of India.

After successfully defending 127 against Zimbabwe, Namibia wins a historic T20I series.

Virender Sehwag Reveals How Anil Kumble's Message Revived His Career

Tejasswi Prakash and I haven't discussed shaadi yet, Karan Kundrra.



Salman Khan signs Jagapathi Babu for Kabhi Eid Kabhi Diwali, following Venkatesh and Pooja Hegde.


Birlasoft has announced a $500 per share share repurchase. There are five things you should know.

After Grasim doubles its capex plan, paint stocks are down.

For FY22, Adani Group shares recommends a 250 percent dividend.

Holcim insures Adani deal receivables for $5 billion in currency.

Flyer's Video vs. SpiceJet Delays Flights Due to "Attempted Ransomware Attack"

With a rise of 71 points on the SGX Nifty, the wider index in India is off to a bullish start.

Delhi's Jama Masjid is undergoing renovations, with work due to begin soon.

The Covid positive rate in Delhi has returned to over 2% after a brief decrease.


Dyal Singh College history student documents a variety of historical stories.

The Supreme Court has put on hold the NGT's decision to fine Coke's bottler.

New Delhi will join the Bahrain-based naval force of 34 nations.


Nagaon village retrieves papers after bulldozers leave, surprised at the 'terrorist' label.

What occurred when the Haryanvi singer left her house in Delhi?

Dawood is in Pakistan and sends Rs 10 lakh each month to his siblings, according to witnesses.


Sadhu begging for alms is assaulted and has his hair cut off in Khandwa, MP


"Does this belong to Andhra Pradesh or Pakistan?" The BJP wants "Jinnah Tower" renamed.

From June 1, India will put export limits on sugar.

Day 2 of the Quad Summit: PM Modi covers the Indo-Pacific, China, the Ukraine war, and Covid-19.

Thank you for your commitment to making democracies work, says Biden to Modi.
A Texas school shooting claimed the lives of 19 students and the teen gunman.

Today's Bharat Bandh: The nationwide strike's demands and other topics. Recent changes

On October 18, Justin Bieber will perform in New Delhi.

The New Lt Governor of Delhi Is the First Non-Bureaucrat To Be Appointed: 5 Points


PM Modi emphasises India's Covid immunisation initiatives and "people-led governance" in Tokyo.

The government claims that worshipping at 'non-living areas' is against the rules.



Opinion: The Constitution had nothing to do with Indian nationalism. It has a long history.
Tamil Nadu is on high alert for possible instances of monkeypox.


Restaurants are being warned about service charges, and a meeting has been called to discuss it.

The Gyanvapi mosque case will be decided by a Varanasi court today.

The government reshuffles the inter-state council, with Prime Minister Modi at the helm.

New late-night rain showers hit Delhi-NCR, causing outages and airline delays: 10 points

ITC and Cargill cancel wheat procurement contracts, putting suppliers in a tight place.

PM Modi: Quad A Force For Good Makes Indo-Pacific Better: 10 Points

Shastri: Washington Sundar is India's "future"

Sardar's coaching debut is a key talking topic before of India's Asia Cup match against Pakistan.




In Cannes, Hina Khan gives a black pantsuit a bling-tastic makeover. Wow photos

Today's Leo horoscope is: Avoid credit transactions!

Deepika Padukone Shows Off Her Million-Dollar Smile In A Green Polka-Dot Outfit At Cannes 2022

'I'm well settled,' Kiara Advani reveals, breaking her silence on her marital plans.


Stocks expected to trade on May 23, 2022

Should You Buy On Day 2? eMudhra IPO GMP, Subscription Status, Financials, and Reviews

General Atlantic is to invest $2 billion in India and Southeast Asia.

Close to securing Rs 20,000 crore to combat competitors: Ravinder Takkar, CEO of Vodafone Idea

Broadcom is in talks to acquire VMware.

The stocks of Tata Steel, SAIL, and JSW Steel have all fallen. Here's the lowdown.

Today is an orange alert for rain and squall in Delhi.

Love For Japanese In Delhi Confidential

Rain cools Delhi's scorching heat by 11 degrees in 1.5 hours.

At Delhi University, the entire globe is a stage.

It's time to revisit the scope of the sedition law.


The bulldozer gathers speed in Delhi.

UP has withdrawn from assisting Delhi in its water issue, as has Haryana.

A 22-year-old Chennai technician died during a dance party after an apparent overdose.

Gyanvapi's assertion is backed up by a 1936 British statement.

Himanta said at an RSS event that closing madrassas and establishing UCC is for the good of Muslims.

In the previous 24 hours, India has recorded 2,022 new cases of Covid-19, with 46 deaths.

In Srinagar, two 'hybrid' LeT terrorists were apprehended.

In a Unity Message, a Karnataka politician makes a gesture towards a Dalit priest.


Gurgaon Traffic Cops After Rain: "We Don't Have Option (WFH), But..."

Yogi Adityanath lists highlights at an RSS event: Loudspeakers, no Eid namaz on the road


Banks will no longer provide brokers with collateral-free intra-day funding.

Excavations at Qutub Minar have not been decided: Culture Minister


"Ohayo, Tokyo," says PM Modi as he arrives in Japan for the Quad Summit.


At a dinner gathering in Cannes, Deepika Padukone turns attention in a little jacket dress.

Today's Cancer horoscope is: Handle problems with patience!


Helly Shah's "Fan Girl Moment" With Aishwarya Rai Bachchan at Cannes 2022


For the second time in 2022, Indian teenager Praggnanandhaa stuns Magnus Carlsen.



Wimbledon loses ranking points due to the ban on Russia and Belarus.


This IPL, Ashwin is the all-rounder.

Axis Mutual Fund has fired a second fund manager who is being investigated for violations.


The CEO of a Singapore fashion start-up explains why she was fired.

In 2021-22, FDI inflows will be at a record $83.57 billion, according to the Centre.

In Delhi-NCR, CNG has increased in price by Rs 2 per kilogramme. Get the Latest Rates

Ola and Uber have received over 3,200 complaints from consumer watchdogs in the last month.

Paytm Q4 results: Loss rises to Rs.761 crore; revenue rises by 89 percent

The Supreme Court erred in admitting writ pleas:

The EDMC blames government employees for illegal structures.


Delhi's peak power demand has reached a new high of 7,070 MW.


For postgraduate admissions, JNU chooses CUET.

Fearing a CBI investigation, the state moves to the Supreme Court.

On Perarivalan's release, Tamil Nadu's chief minister hugs him, angering the Congress.

Wheat trade has halted, with prices returning to pre-ban levels of Rs 2,250-2,300 per quintal.

Rabri Devi, Lalu Yadav's wife, smacks a supporter who is heckling CBI officers.

Why did the Delhi government's doorstep ration delivery scheme get invalidated by the High Court?

The conviction of Yasin Malik could spell the end of separatist politics.

When a Zepto delivery lad is killed in a hit-and-run, the company pays a compensation of Rs 10 lakh.

The British did not teach India democracy: JNU V-C

Assam Floods Affect 8 Lakh People, Over 500 Families Live On Train Tracks.
Dominica Drops Charges Against Mehul Choksi for "Illegal Entry"

A new complaint has been filed against Lalu Prasad and his relatives in the land-for-jobs scandal.


TSRTC will give pupils with a complimentary bus ride to the SSC exam centre.

The Supreme Court has remanded the Gyanvapi mosque worship case to a Varanasi district judge.


25,000 Dollars a Month? Report If You Are In India's Top 10%
China's Pangong Lake bridges are illegal, according to India.

Navjot Singh Sidhu could serve less than eight months in prison.

Professor from Delhi Hindu College was arrested over a post on Varanasi's Gyanvapi.

The 'truth is out' for the accused's family, and the victim's relatives say they support the police.


Nikhat Zareen's education continues as she approaches the international championships.


Nazila, Munawar Faruqui's GF, Discusses Anjali Arora and Says, "I am Not Open to This."

Ravi Shastri, Aamir Khan has a question for you: "Toh Pakka Selection?"


Dhaakad Movie Review: Kangana Ranaut's Actioner Is Kill Bill and Atomic Blonde's Aborted Child!

Kangana Ranaut's Actioner Dhaakad Is The Aborted Child Of Kill Bill And Atomic Blonde!

Review: Kartik Aaryan's Bhool Bhulaiyaa 2 holds its own against the original; Tabu steals the show

Deepika Padukone Is An Object Lesson In How To Slay The Red Carpet At Cannes 2022

Prudent Corporate Advisory Services makes its debut with a 4% premium.

In the grey market, Delhivery falls below the IPO price ahead of its Tuesday offering.

Next Month, Mahindra Scorpio prices will be revealed.


Metro plans to exit the Indian market for $1.5-1.75 billion after 19 years.

Elon Musk Says "Tesla Is On My Mind 24/7" Amid Twitter Distraction Concerns



Hyderabad has the most new Covid cases in Telangana, with 36 new cases.

Couple booked after domestic help is beaten up

Ex-DU students have a 'centenary chance' to finish their degrees: In 19 days, 1,560 people applied.

Ghazipur slaughterhouse to be decommissioned

Supreme Court rules that the GST Council's ideas are not binding on states.

Yasin Malik was found guilty of supporting terrorism by a Delhi court.


The battle between Delhi and Haryana over the Yamuna's falling level has not abated.

What will the temperature be in Delhi today? In the weather prediction, IMD reveals

The BJP Yuva Morcha vandalised an Aurangzeb Lane signboard in Delhi.


What explains Sharad Pawar's NCP's hostile turn? Egg, ink, and slaps.

Azam Khan, the leader of the Samajwadi Party, has been released from prison after 27 months.

Domestic help found in pool of urine after being thrashed by a Delhi couple: Report

Navjot Singh Sidhu is expected to surrender to police today in a road rage case.

With immediate effect, AIIMS eliminates all laboratory and testing prices up to $300.

At least ten people are trapped after a tunnel under construction in Ramban, J&K, collapses.

Survey findings show temple debris and Hindu features in the Gyanvapi complex.

The CBI is investigating a new corruption case against Lalu Yadav and his family members.


Today's Leo horoscope for May 19, 2022: Your career will grow!

Should You Buy on Day 2 of the Paradeep Phosphates Initial Public Offering?


Check out the latest petrol and diesel prices in Mumbai, Delhi, and other cities on May 19.

Gold and silver prices are down today on the MCX | Get the latest rates here.


After the latest price hike, a domestic cooking gas cylinder will cost more than $1,000.

ITC stock is at a multi-year high after Q4 results beat expectations. Should you purchase?


On May 22, the municipal corporations of Delhi will be formally merged.



The Delhi BJP chief writes to Amit Shah about bus fires and requests a CBI investigation.

Delhi: A 13-year-old girl was abducted and gang-raped, and four boys were apprehended.

Kashmiri Pandits will not be stationed in high-risk areas: DC Kashmir

Several landmines explode along the Line of Control in Poonch, J&K.


In a vicious brawl, Bengaluru schoolgirls rip each other's hair out.

Floods in the northeast have forced half a million Indians to escape.

At Cannes, why was Deepika Padukone dressed in sofa upholstery?

Gyanvapi pond area is now a fortress, with increased security.

Punjab: Farmers in Abohar and Fazilka remain in for the night and celebrate with potato curry.

Under the Senior Citizens Act, a daughter-in-law cannot be asked to pay mother-in-law maintenance.

Aurangzeb's plaques should be removed, and roads should be renamed: Mayor of Agra

India's Covid Vaccine Swipe At The West At The United Nations As It Defends Wheat Export Ban



Congress condemns the verdict, but the DMK applauds it.


Indrani Mukerjea was granted bail by the Supreme Court after she "spent 6.5 years in prison."

Hardik Patel Leaves Congress, Swipes At Rahul Gandhi With "Chicken Sandwich"

Yogi Adityanath's tweet and statue have sparked talk of renaming Lucknow.
Amid a controversy about "locked doors," photos of Taj Mahal rooms have surfaced.

Bengaluru rains: Heavy rains cause flooding, and more showers are expected.

Ukraine's 'final bastion of resistance', Mariupol, is on the verge of surrendering to Russia.

Farmers' Protests Are "Unwarranted," Says Punjab Chief Minister

Supreme Court orders DM to conserve the Gyanvapi Pond Area

CBI raids are being questioned by Chidambaram and Congress.

Amber Heard Responds to Accusations She "Pooped" in Johnny Depp's Bed with "That's Disgusting"


The Archies and the perplexing case of "relatable cinema"




The Tata Nexon EV Max gets a bigger battery, a longer range, and more features.

Check out today's gold and silver prices in Bhubaneswar and Cuttack.



NCLT order on Pawan Hans sale to be examined by the Centre

LIC Stock Makes a Sluggish Market Debut, Listing At An 8.62 Percent Discount: Live Updates

With a gain of 42 points on SGX Nifty, the wider index in India is off to a positive start.



SDMC conducts special drive to curb mosquito breeding

Temporary move to online programmes in some DU colleges

Samiti advises Haryana government to purchase property for AIIMS before May 31 or face a backlash.


Five renowned Delhi city marketplaces will be chosen for rehabilitation.


The Delhi municipal corporations are planning the next anti-encroachment drive.

The Yamuna has run dry, and the capital's water supply will be affected starting today.

South MCD is leading a campaign to prevent vector-borne diseases in Delhi.


KPs distraught with mainstream opposition's sloppy, vague response to targeted killings in Kashmir

Hardik Patel says he would decide his future after meeting Rahul.

During a police operation in Uttar Pradesh, a man was detained for shooting a woman.

Shopian community unable to come to terms with civilian killings amid dread and darkness


Rajnath Singh will deploy two Indian Navy boats in Mumbai today.

'The Kashmir Files,' says Mehbooba Mufti, are to blame for the violence in the Valley.

Hindus allege a shivling in the masjid reservoir, while the mosque claims a fountain

Wheat prices are up 6% as a result of the export ban.

Elon Musk answers with a faeces emoji after Parag Agrawal defends Twitter's spam account count.

Regional parties, according to Rahul Gandhi, cannot defeat the BJP. RJD retaliates

PM Modi believes that stronger India-Nepal connections are required to address rising issues.

Today is a crucial Supreme Court hearing in the Gyanvapi Mosque case in Varanasi.

Today's LIC BSE, NSE Listing: Time, Price, Strategy, LIC Share Price, and Details

Residents in Delhi are requested to stay indoors due to the high temperatures.

The Vatican has declared an 18th century Indian who fought casteism a saint.

Devendra Fadnavis Issues Warning to Maharashtra Government on "Babri Structure"


The expulsion of Biplab Deb before of the 2023 elections exposes a schism inside the Tripura BJP.

IPL: What does the future hold for the Jadeja-CSK partnership?










The LIC has set the IPO issue price at Rs 949 per share.

Today's gold price: Silver was sold at Rs 60,800 per kilo for 10 grammes of 24-carat gold.


"Not at all what I expected...": Twitter Fires Two Executives Ahead Of Elon Musk's Takeover

Should Investors Book Profit or Hold Tata Motors Shares Following Q4 Results?

The Sensex has dropped 6,000 points in the last month, costing investors Rs 34 lakh crore.

The Delhi government's transport department has issued a licence to chaos for e-rickshaw drivers...

Haryana's water supply would be significantly impacted, Delhi writes again.


'We are losing our homes; why is everyone quiet?' says SDMC's anti-encroachment campaign.

Supreme Court will investigate Tablighi visa petitions

The Waqf Act has been challenged, and the Delhi High Court has requested a response from the Centre.

Cattle grazing in green notified areas must be monitored, according to the Delhi High Court.

The journey of a 28-year-old from a Mumbai slum to a university in the United States, via JNU

Hate speech is ruining the atmosphere, according to the Supreme Court.


The Crime Branch of the Delhi Police has arrested an IAF employee for espionage.

Delhi: A businessman claims that six people posing as CBI agents threatened him.


From a Sukhoi fighter jet, India tests the BrahMos long-range missile.


An investigation agency goes after the Dawood Gang and arrests gangster Chhota Shakeel's aides.

Chopper crashes near Raipur, Chhattisgarh, killing two pilots.

After the killing of a Kashmiri Pandit, J&K saw overnight protests.


Rahul Gandhi is greeted at 5 a.m. ahead of the Congress' three-day brainstorming session.



Asaduddin Owaisi on Gyanvapi Verdict: "I Don't Want To Lose Another Masjid"

Rizwan: Pujara is second only to Younis Khan in terms of attention and focus.

Ravichandran Ashwin's strange stance towards Kuldeep Yadav has sparked a meme-fest on Twitter.


CSK Unfollow Ravindra Jadeja On Instagram Following Rift Rumours: Source

"The Second Alia Bhatt": A Woman's Video Goes Viral On The Internet

Actor Harsh Varrdhan Kapoor was mocked for his remark on the "sad reality of my life."

Kangana Ranaut Says She'll Never Marry: 'There are rumours that I beat up boys.'

As filming for Rajkumar Hirani's Dunki begins, Shah Rukh Khan looks stylish in black. BTS picture


Latest Crypto Crash News: What's going on in the crypto market? Important developments

According to a report, Saudi Aramco has dethroned Apple as the world's most valuable company.

As expectations of a Fed rate hike grow, the rupee falls to a new low, sending stocks plunging.

Allotment of LIC IPO shares is expected today. Direct connections to online status checks

The Supreme Court upholds a school's objection to financial statements being uploaded.

Two new campuses will be built at Ambedkar University.

SDMC clears encroachments in Najafgarh zone's Lodhi Colony.

DU sees 97.5 percent turnout in its first totally offline exam in two years.

South Asian University is rushing to fulfil the deadline for shifting to its new campus in Delhi.

Journey of a 28-year-old from a Mumbai slum to a US institution via JNU

The Delhi High Court issues a split decision on the criminalization of marital rape.

Noida CEO's stay has been extended by the Supreme Court.

Rahul Gandhi is reportedly considering a run for Congress president.

Will follow the court's decision in Yasin Malik's case: Union Minister of State


Bengal civil society issues a statement condemning Mamata's award.

Aman Chopra, a TV journalist, has been charged with sedition by the Rajasthan High Court.


Mukul Goel, the DGP of Uttar Pradesh, was fired for 'neglect of duty' and 'indolence.'


In the Datia district of Madhya Pradesh, a cop strangles a 6-year-old boy to death.

Thursday's day trading guide: 6 stocks to buy or sell today — May 12th

A Shiv Sena MLA from Mumbai has died while on vacation in Dubai with his family.

A railway cop is caught on camera dragging a woman from a gap in the train platform.

Pooja Singhal, the Jharkhand mining secretary, was arrested in a money laundering case by the ED.

Trinamool concedes, but fumes, as Deputy Speaker Babul Supriyo is sworn in.

A sex worker, not a married lady, has the legal right to refuse: HC judge in Delhi


Wednesday's day trading guide: 6 stocks to buy or sell today — May 11th

Vodafone Idea, Wipro, Cipla, Adani Ports, PNB, and other stocks to watch today

Himanta Sarma says, "PM Amit Shah." Congress sneers, "Not A Slip Of The Tongue."

To protect the currency, the RBI may exercise caution in expending foreign exchange reserves.


DGCA investigative team to investigate into operational and legal norms on IndiGo flight row


According to the high commission, no Sri Lankan politician has fled to India.

In the epidemic year, India's population increased, but death records in Uttar Pradesh decreased.

Mahua Moitra on the Centre's response to the Sedition Law, "What Pandit Nehru could not accomplish."

As the storm scrapes the coast of Andhra Pradesh today, widespread rainfall is expected.

The ACB wants Aadhaar data, but the UIDAI argues cardholders should be heard first.

Jharkhand CM Soren's disqualification due to a mining lease is insufficient: Ex-CJI




As Bitcoin approaches $30,000, buyers of 2021 are hurt.

ACC expects JSW Group to make a $7 billion deal for Ambuja.

The Supreme Court has put a hold on applications challenging the Information Technology Rules.

The Delhi High Court is furious with the movie's depiction of a sex determination test.

The High Court has ordered that the child's mortal remains be released to the family.




According to the Delhi High Court, the elephant will spend the rest of its life in Yamuna Nagar.

Tajinder Bagga of the BJP has filed a court case in Delhi, demanding his phone and security.

A Pakistani drone carrying ten kg of heroin is shot down by border security forces.

ASI expresses worry the day after LG Manoj Sinha prays in a protected J&K temple.

Due to a power outage in Madhya Pradesh, the bride marries her sister's fiancé.

On Monday, the state awoke to the 'Azaan vs Hanuman Chalisa' competition.

Anth-heen lectures, Chintan Shivirs, Antony panels

No arrogance, receptive CM Mann is praised by Sidhu.

How Uttar Pradesh's 'Tejo Mahalaya' notion gained traction

A Union minister discusses why Prime Minister Narendra Modi decided to revisit the sedition statute.


Parts of Mumbai's suburbs are without power.

Who is Salim Fruit, the NIA's detainee following raids?

The bulldozer reaches Shaheen Bagh, but is forced to retreat due to objections.


Bhagwant Mann's Reaction To The Punjab Police Intelligence HQ Bombing In Mohali


Amit Rohidas, an Indian hockey player, feels that playing in Rourkela will be fantastic.

Sunil Gavaskar Jokes About Virat Kohli's Form: "All These Runs Are Being Kept For..."


MS Dhoni says it won't be the end of the world if CSK doesn't make the playoffs in IPL 2022.

Today's Sagittarius horoscope is 9 May 2022: Practice patience!
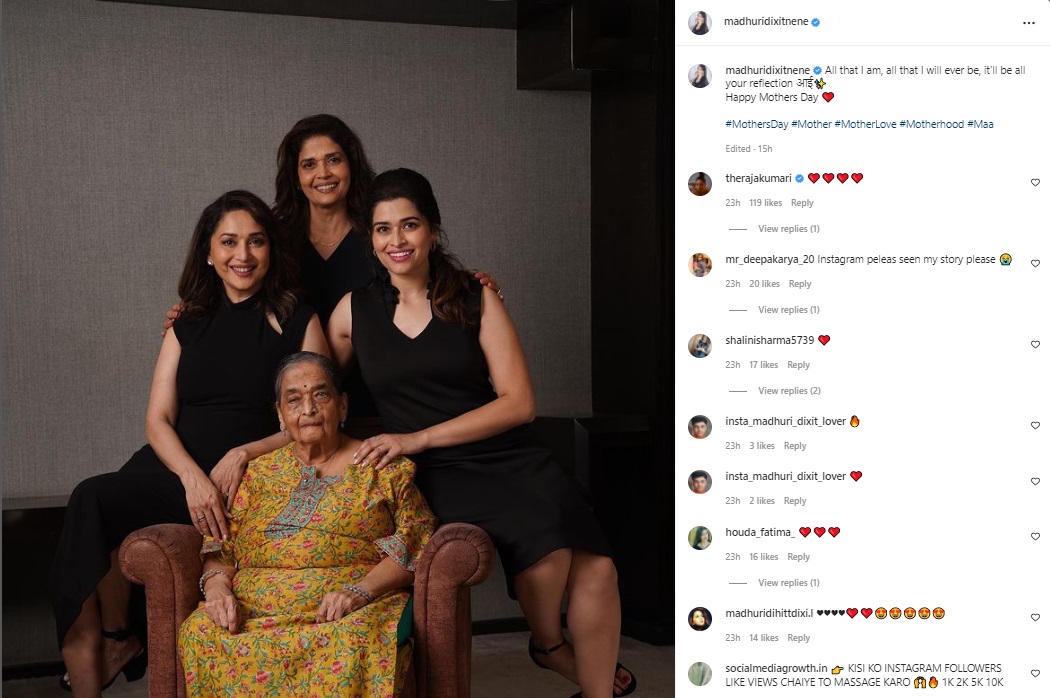

KGF Chapter 2 Box Office (Worldwide): Surpasses RRR To Become India's 3rd Most Successful Film!


Why Have Foreign Investors Mostly Avoided The Mega LIC IPO?

LIC IPO GMP, Subscription, and Review; Is This Your Last Chance to Invest Today?

RIL, Campus Activewear, Tata Power, UPL, and other stocks in the news today

Rupee Drops To New Lows, Trading Above 77.40 Per Dollar

The rupee hits an all-time low, as the Sensex plummets 700 points.
On a down market, RIL shares plummet. What should investors do after the fourth-quarter results?

Members of the Delhi BJP's booth committee will be replaced.

The new OPD block for Lady Hardinge will open today.


Supreme Court judges Justice Dhulia and Justice Pardiwala take their oaths.

Tajinder Bagga of the Delhi BJP said he had no intention of physically injuring Kejriwal.


In Punjab, terror suspects from Karnal supplied arms and drugs.

Manoj Sinha, J&K LG, performs puja at the ruins of an ASI-protected temple.

The strange situation of the Abdullahs' silence on the delimitation panel order

Passengers strain as services are delayed due to an overhead wire failure in Mumbai.


"Open 22 Rooms In Taj Mahal": A Court Petition Requests an Investigation Into Its History


Domestic Help Kills US-Returned Chennai Couple, Steals $5 Million


Justice Jamshed Pardiwala, a fourth-generation lawyer, has been promoted to the Supreme Court.


Elon Musk's latest tweet perplexes netizens: 'If I die under unexplained circumstances...'


Shah will assess vigil upgrade while on border reconnaissance.

In Mumbai, Western Railways trains have been affected.

IndiGo 'denies' boarding to a special needs youngster.
Is the Nifty50 going for 16,200 on May 9?


Putin will lead the commemoration of WWII triumph, and Zelensky reminds us: ten marks

Himachal seals its borders and intensifies checks in the wake of the 'Khalistan' referendum call.

Rain and high winds are expected in three states as Cyclone Asani intensifies.




As Mumbai Indians beat Gujarat Titans in IPL 2022, actor Ranveer Singh celebrates in style.

Athiya Shetty Addresses KL Rahul's Wedding Rumors, Says She's Moving In But Not With Him

Manoj Pahwa recalls his and'samdhan' Supriya Pathak's families sharing summer vacations.



The price of gold in India has dropped by Rs 100 for 24 carat and Rs 200 for 22 carat today.

Should Investors Book Profit Or Hold Adani Power Shares After A Year of Gains?

Inside Elon Musk's Big Twitter Plans

Should You Buy on Day 4? LIC IPO GMP Today, Subscription Status, Price, and Other Details

The price of cooking gas cylinders has been raised by $50 for homes.

Debashis Chatterjee will lead the united Mindtree-L&T Infotech company.

Summer farm fires foreshadow the winter disaster.

Punjab takes 'detention' to the High Court, and Haryana and Delhi are given time to respond.

Justice UU Lalit has offered to recuse himself from hearing Prashant Bhushan's appeal.

Experts undertake a campaign to educate Haryana farmers on how to make the most use of pesticides.

What We Know About the AAP Government's Proposal: Delhi MLAs Will Soon Be Paid Rs 90,000 Per Month



MLAs in Delhi will receive a 66 percent pay raise; here's how much they'll make.

Tahir Hussain, a former AAP councillor, has been charged as a "active rioter" by a Delhi court.

Lalitpur teen tells counsellor he is raped every night under a bridge.

Medical students are urging Prime Minister Modi to postpone the NEET PG 2022 exam.

The footage of Imran Khan making the 'donkey' remark has gone viral.

In Srinagar, a cop in civilian clothes was shot and seriously injured.

In a custodial death case in Chennai, cops are being investigated for murder.

ED raids locations linked to IAS Pooja Singhal; CA mansion valued at Rs 18 crore

Rahul Gandhi on WHO Covid Deaths Report: "Science Doesn't Lie, Modi Does"

An Inspection Survey Takes Place At A Mosque Near Varanasi's Kashi Vishwanath Temple

In Bengal, Shah rules out Prez rule and advocates for a political war.

Sri Lanka's emergency, Nepal's currency depreciation, and South Asia's crisis

Asaduddin Owaisi on the Hyderabad Killing: "We Don't Stand With Murderers"

Tsahi Halevi of Fauda sings Tere Jaisa Yaar Kahan and says India has a lot of creativity.





Inside Photos From AR Rahman's Daughter Khatija's Wedding Have Gone Viral

Today, the Department of Pensions and Pensioners' Welfare hosts an All-India Pension Adalat.

The SC collegium has recommended Justice Sudhanshu Dhulia.

Manish Sisodia: SCERT is the backbone of Delhi's educational model.


Punjab Police have arrested Delhi BJP spokesperson Tajinder Bagga, according to party leaders.

On May 9, the Congress Working Committee will convene to examine the Nav Sankalp Shivir's agenda.


In the High Court, Delhi corporations argue that a House panel cannot inquire for information.

SC upholds Rs 4,600 crore arbitral judgement in Delhi airport line

'You're a Hero,' a Shark Tank India contestant writes Ashneer Grover a thank-you letter.

Delhi Police publishes suspect sketch in MCD school sexual assault case

According to WHO, India has the highest rate of excess Covid-19 fatalities in the world.

The current heat wave in India emphasises the importance of switching from coal to solar energy.



In Mumbai, Reliance instals the world's largest elevator.

Lokesh requests a postponement of the NEET PG exam in Vijayawada.


Sena's Raut criticises Raj Thackeray: 'Because his brother is the Chief Minister...'

After learning that his fiancée is a conman, an Assam cop arrests him.



"Don't patronise," says India's UN Ambassador in a terse response to the Dutch envoy on Ukraine.

WHO estimates 47.4 lakh Covid fatalities in India, over ten times the official figure.

Punjab Police detain BJP leader Tajinder Bagga at his Delhi home for allegedly threatening Kejriwal.

In Hyderabad, a man was stabbed to death in an alleged case of honour killing.


The Governor of Tamil Nadu has sent an anti-NEET bill to the President for his approval.

The Telangana High Court has denied Rahul Gandhi's request to hold a rally at Osmania University.

Devendra Fadnavis Speaks Out On Wife-Maharashtra Chief Minister Feud

Outside the Calcutta High Court, P Chidambaram was heckled by party workers.

Patna ke Prashant Kishor: Why is his second innings in four years such a challenge?

Lalu Prasad Yadav on the Hanuman Chalisa-loudspeaker row in 'Bahut Galat'

In a photo with Rahul Gandhi, a Nepali singer writes, "such humble, simple person."

A teen rape survivor was raped again in a police station in Uttar Pradesh, and the SHO was arrested.

In Delhi, two girls were 'forcibly undressed in MCD school,' but no arrests have been made so far.

De-escalation on the Ladakh LAC has yet to take place two years after Galwan.

PM Modi and President Macron have called for a "immediate end to the suffering" of Ukrainians.

Jodhpur's calm was shattered by flags, loudspeakers, and a rope on a freedom fighter's bust.

Poonam Pandey is ousted, and Munawar Faruqui believes Munawar Faruqui should be the winner.





With a rise of 98 points on the SGX Nifty, the wider index in India is off to a bullish start.


"I'm Not An Android," Elon Musk Admits, "I'm Not An Android."
Tata Steel, Titan, Future Retail, SpiceJet, and Kotak Mahindra Bank are stocks to keep an eye on.

HUL loses its position as India's largest FMCG company to Adani Wilmar.

A DU panel will look at the drop in SC/ST admissions.

Signals will reappear at Rajiv Chowk after three years to help alleviate traffic congestion.


The Foreign Correspondents' Club has cancelled Vivek Agnihotri's news conference, according to him.

The SGPC's 'intervention' is a source of contention for the Delhi Sikh community.


Putin will undergo cancer treatment and relinquish power, according to a report in the US media.

Keep your umbrellas handy since rain is expected to persist in Karnataka for the next two days.


Tejashwi Yadav: I don't watch or listen to news on Prashant Kishor.


Eid celebrations in old Delhi are back after a two-year hiatus due to the removal of regulations.

'They tried to kill my brother,' says victim's family after a road rage incident in Noida.


In the RSF 2022 World Press Freedom Index, India drops eight places to 150th place.

NEET PG 2022 should be postponed, according to 86 percent of students.



PM Modi is told in Copenhagen that India should try to persuade Russia to cease the war.

Raj Thackeray is facing a police investigation, yet he is committed to his loudspeaker plan.

This Akshaya Tritiya, there are three clever strategies to buy gold.


Elon Musk's Twitter ambitions are being tested in India and China.

On greater volumes, Tata Steel expects a 33-38 percent increase in net profit in the fourth quarter.

In Minutes, a Citi Trader's Error Wiped Out $315 Billion From Europe Stocks

Adani Wilmar buys Kohinoor to strengthen its position in the Basmati rice market.
The Supreme Court has extended the stay on the destruction of Delhi.

In May, 6,194MW of heat upsets the balance of power in Delhi.

IIT Delhi and ITC have signed an agreement to collaborate on STEM research.

In Delhi, the heatwave exacerbates the symptoms of COVID-19 patients.

The Delhi Commission for Women assists retired teachers in receiving around Rs 20 lakh in back pay.

The homeless at Haj Manzil have been requested to leave, and the government has given a notice.



Today's Delhi weather forecast calls for a high of 39 degrees and partly cloudy skies.


According to stats, Delhi airport was the world's second busiest in March.


The US Supreme Court is expected to overturn a landmark abortion decision, according to reports.

The doctors' association has requested that NEET-PG 2022 be postponed by the Ministry of Health.

Why are Punjabi farmers burning wheat stubble while the rate of dry fodder is so high?

Maharashtra Minister Nawab Malik is in a serious condition in hospital, according to a lawyer.

Before you subscribe to the LIC IPO, here are some things you should know.
In the wake of rumours of his departure, Hardik Patel deletes Congress from his Twitter bio.


Due to the heat wave, Haryana has changed school hours from Wednesday to 7 a.m. to 12 p.m.


DGCA grounds SpiceJet crew due to air turbulence: 2 in ICU


Before Modi meets Macron, the French have decided to withdraw from a submarine deal with India.


On Rajiv Gandhi's "15 Paise in One Rupee" remark, PM Modi takes a shot at the Congress.

As Army Chief, Bombay Sapper General Pande will be put to the test.
Passengers on a SpiceJet nightmare aircraft recall a near escape as terrifying as Runway 34.

The Rajasthani railway station 'Miyan Ka Bada' has been renamed 'Mahesh Nagar Halt.'


PM Modi danced with an Indian boy as he sang a patriotic song.

GST collection reaches an all-time high of Rs 1.68 lakh crore in April.

PM begins a three-nation European tour; Germany's agenda includes the post-Covid economy.

Actor Vijay Babu offers to remain away from AMMA, but he declines.

Satyajit Ray's Pikoo, a Little-Known Film That Is Mostly Ignored by His Admirers

Because of Acharya's failure, Jr NTR and Koratala Siva's NTR30 has been postponed.


Mohsin Khan, the trump card of the Lucknow Super Giants, outfoxed Rishabh Pant.







Dharmendra, a veteran actor, has been admitted to the hospital.

In 2022, when is Akshaya Tritiya? The Shubh Muhurat and Time are important to know.



Should you buy actual gold or a Gold ETF on May 3rd, Akshaya Tritiya?

The Delhi BJP has compiled a list of 'failures' by the AAP government during the last seven years.

Dust storms are expected in Delhi, with a minimum temperature of 27.4° Celsius.

Employees of the Ram Manohar Lohia Hospital in Delhi demonstrate against their dismissal.

The government wants to restore the east Delhi Cultural Club to its former splendour.

Centre tries to mislead the public and instil fear over the electricity situation in Delhi.

The 'State Emblem of India' will soon be atop the New Parliament building in Delhi.
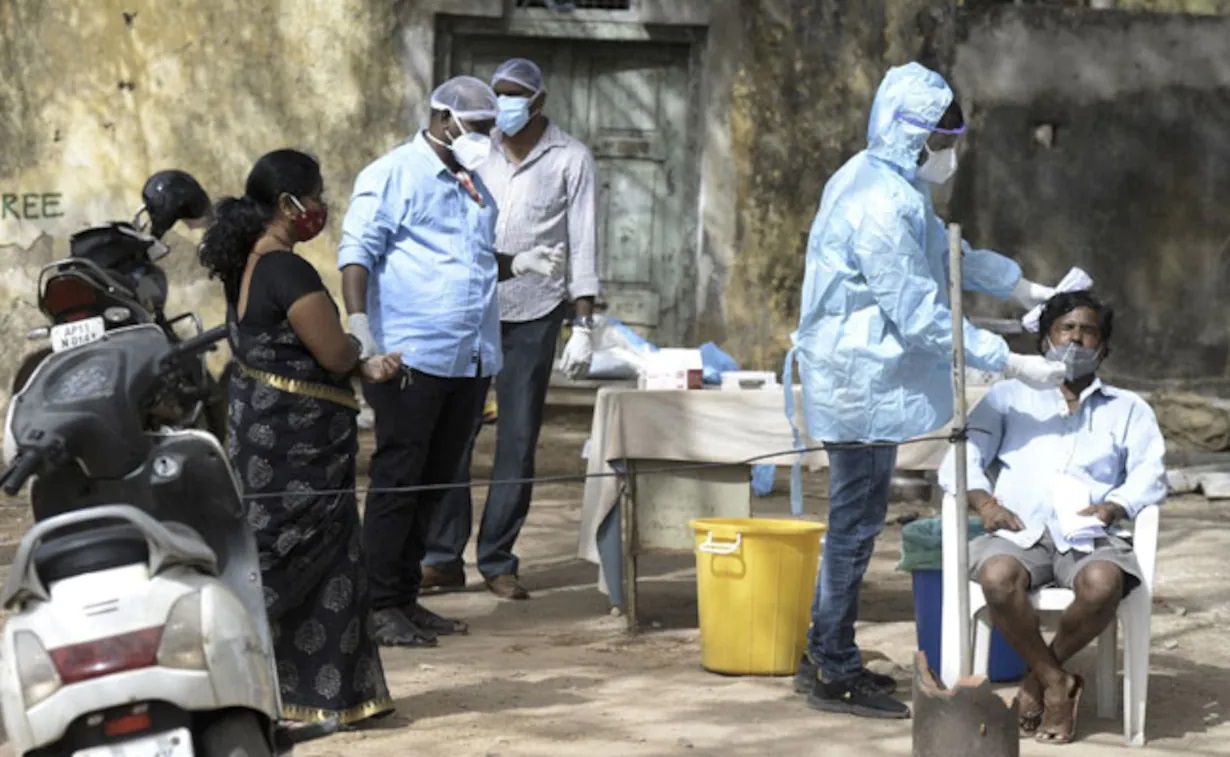
After two months, India's Covid positive rate surpassed 1% once more.


At a Delhi Metro Station, a man commits suicide.


In Delhi, an ex-boxer has been arrested for a series of offences.

HC takes a firm stance on Sainik Farm.


UP cops are accused of assaulting a woman during a raid, according to her family.

Pregnant woman kidnapped and gang-raped in front of her family at a train station


After eating bad shawarma at a snack bar in Kerala, a student died.

After an argument about road rage in Noida, a car hits a man on camera.


Why did Bengaluru get bombarded with hailstones in the middle of a heat wave?


Osmania University'refuses to allow' Rahul Gandhi's visit, causing a ruckus on campus.

After Tamil Nadu medical students took the Sanskrit Charak oath, the Dean was fired.


During turbulence, a video reveals terror onboard a SpiceJet plane; 14 people were injured.

Prashant Kishor's New Tweet: "Time To Go To The Real Masters"

Yuvraj Singh is the "Right Guy To Lead Indian Test Team," according to this "Future Legend."



P Chidambaram claims that the "Umran Malik Hurricane" has blown him away and that the BCCI should...


Vijay Sethupathi, Samantha, and Nayanthara excel in Kaathuvaakula Rendu Kaadhal, despite its flaws.



Kannada leaders unite in response to Ajay Devgn-'Hindi Sudeep's is national language' controversy.

Singapore defends itself, saying 'US...' in response to the execution of an Indian-Malaysian.

Elon Musk 'jokes' that he will purchase Coca-Cola and 'bring cocaine back' on the market.

Modi is considering introducing the Uniform Civil Code ahead of the 2024 elections.


Uddhav Thackeray's suggestion as Maharashtra faces an increase in Covid

Kerala wants to study Gujarat's dashboard, which has sparked controversy.

The Uttar Pradesh government has removed 10,900 loudspeakers, despite a four-year-old directive.

5 key takeaways from PM Narendra Modi's meeting with state chief ministers on the Covid-19 rise

Mamata blasts Modi for spending Rs 15k crore on petrol subsidies in three years.

Wife of guy killed in Noida bar brawl: 'What did he do to deserve this?'

LIC IPO: Here's how to get the most out of the mega-bidding. issue's

Thursday's day trading guide: 6 stocks to buy or sell today — April 28th

Because...: the United States, India and Russia became allies of choice out of need.

Russia received ruble payments from four European gas buyers, according to a report.

The "Laxman Rekha" response of the Delhi High Court to Umar Khalid's criticism of the Prime Minister

Evicted Padma Shri Awardee, 90; Government Sets Deadline for 8 More Artists


'Bible, Quran, and Bhagavad Gita cannot be compared,' says Karnataka's education minister.

The German minister is not in India to preach or instruct about the Ukraine issue.


From May 2-4, Prime Minister Modi will go to Germany, Denmark, and France.

Telangana Minister to Centre: Scrap Cess So We Can Give Petrol At 70

The heat wave in north India is expected to continue. Relief is most likely due to...

India's likely 18-man roster for the T20I series against South Africa


'In India, there is always jealousy and individuals who want you to fail,' says Shastri.

During Match 39, Riyan Parag and Harshal Patel got into a heated argument.

Ravi Shastri's Advice To Virat Kohli: "Pull Out Of The IPL, For All You Care"


'We Started Seeing Each Other Without Even...' says Ajay Devgn on his marriage to Kajol.

KGF 2 has been praisingly received from all quarters.

Anees Bazmee Reveals Why Akshay Kumar Wasn't Cast In Bhool Bhulaiyaa 2



LIC Policyholders: Interested in a Discounted LIC IPO? What You Should Know


The Supreme Court has ordered that no hate speech be allowed at Dharam Sansad.


Ahead of the elections, the Congress appoints the ex-chief minister's wife as the Himachal chief.


Cops refuse to attend a religious conclave in Roorkee after a hate speech in Haridwar.


A massive fire has erupted at a landfill in Delhi.

Where were you when Asia's order was threatened: EAM to Europe?

In northeast Delhi, a man has been arrested for inciting hatred.

The fire at a junkyard near a metro station in South Delhi has been extinguished.

It's for this reason that Mukesh Ambani's $3.2 billion Future Retail acquisition fell through.



CBSE Term 2 2022 Live: Class 10 English test begins; paper analysis will be available soon

Today, PM Modi will convene a Covid review meeting with CMs.

At Karachi University, a female suicide bomber detonates her explosives.

Prashant Kishor requested four points from Congress.

Two children were among 11 people electrocuted during a temple chariot procession in Tamil Nadu.

IPL 2022: Why was Rishi Dhawan, the guy who fired MS Dhoni, wearing a face shield?

"They have stuff that maybe we still don't have," Guardiola says of Real Madrid.

The "Jealous Gang" in India wanted me to fail, but I was tough: former coach Ravi Shastri



What Happened On Day 1 Of Promotions For Kartik Aaryan In Bhool Bhulaiyaa 2




Nawazuddin Siddiqui reveals he was not paid his fee for a brief role in Shool, which was $2500.


Kiara Advani posts a photo with her rumoured boyfriend Sidharth Malhotra amid split rumours.

Today's hottest stocks include Bajaj Finance, Tech Mahindra, Steel Exchange India, and others.

Indonesia has lifted its restriction on the export of crude palm oil.


What Twitter CEO Jack Dorsey Said About A Possible Hiring Freeze After Elon Musk's Deal

According to the 2011 census, Delhi has 4,200 transgender people, yet only 23 of them have IDs.

'Police booths are a must,' says the people.




Today, the nurses' union at AIIMS will go on an indefinite strike.


DU's Left student groups want the notice on required protest permit revoked.


KCR's Son On Talks With Congress: Prashant Kishor "Latching Onto Sick Unit"

The BJP has now joined the party, and Azam's guest list has grown.


Two men offer a ride to a woman walking home and then murder her after rape, according to police.


Assam Police re-arrested Gujarat MLA Jignesh Mevani after he was released on bail.

'Russia's final hope....': Ukraine after WWII-3 remark- 10 points

After Uddhav Thackeray, PM Modi was offered a tit-for-tat Hanuman Chalisa.







Kiara Advani Burns Up Instagram Amid Breakup Rumours; Sidharth Malhotra Turns 'Foodie' in New Selfie

Aww! This post-wedding family photo of Ranbir Kapoor holding Alia Bhatt is all kinds of lovely.



Kangana Ranaut has revealed that she was sexually molested as a child by a boy from her hometown.

Day 11 of the KGF Chapter 2 box office collection: Yash's film collects Rs 880 crore globally.


Q4 Review of ICICI Bank: Closing the Gap With HDFC Bank

Elon Musk's takeover offer is being re-examined by Twitter, according to a report.

Six variables are driving the sell-off in the Nifty and Sensex for the second day.


From May 3, Air India will restart flights between Gannavaram and New Delhi.

A Delhi court has denied bail to a Jamia lecturer who was arrested for allegedly demanding a bribe.


With demand rising, the Centre's procurement of Punjab wheat has dropped to a 15-year low.

In Kolkata, a massive factory fire has erupted.

Born a guy but mistaken for a girl: After 16 years, a teen reclaims her gender identity.


Due to a dramatic drop in generation, there is currently a power crisis.


Professor Fired by University After Allegedly Making Offensive Remarks About Lord Ram

According to IMD, the mercury in Delhi could reach 44-46 degrees by April 28.

The Masterstroke of Delhi Confidential

Ukraine wants heavy weapons from the US at discussions in Kyiv on April 24: Zelensky


As retaliation, India has suspended tourist visas for Chinese citizens, according to reports.

On Wednesday, the Prime Minister will meet with officials to discuss the Covid situation:


Prashant Kishor Talks in Congress, and KCR's Party Joins I-PAC

PM Modi will meet with President Emmanuel Macron to strengthen connections between India and France.

'Everything should return to normal,' says a chorus during the Jahangirpuri tiranga yatra.

Cops use tear gas during protests in Paris following Emmanuel Macron's election victory.



Amrita Rao and RJ Anmol talk about their surrogacy experience and how they lost a baby.





As the 'Hanuman Chalisa' row knocks on Uddhav Thackeray's door, Sena growls.

Sharekhan recommends purchasing Tata Elxsi with a target price of Rs 9000.

Today's Petrol Price: Fuel Prices in Delhi and Mumbai Remain High; See City-Wise Prices

Creditors reject the transfer of FRL assets to Reliance Retail, but shareholders approve.

Four people were injured in a shooting in a Washington neighbourhood, and the gunman was found dead.

A blast rips through an Afghan mosque during Friday prayers, killing 33 people.




Tina Dabi, an IAS officer, married a fellow officer | Video

"I've made it clear to India that I don't want them to rely on Russia," the Pentagon said.


Rajiv Kumar will stand down as NITI vice chairman, and Suman K Bery will take his place.

Rakesh Asthana, the Commissioner of Police in Delhi, interrogates the main accused, Ansar.

'Can select a friend, not a neighbour...' says Nirmala Sitharaman of India-US relations.


Mike Tyson, a former boxing champion, hits a passenger on a plane.



Alia Bhatt's "Mami" Reaction To Samara Sahni's "Welcome To The Family" Instagram Post



Day 8 box office collection for KGF Chapter 2: Yash's blockbuster earns Rs 268 crore.

The price of gold in India has dropped in the previous 24 hours; check current rates.

Elon Musk receives a $46.5 billion loan for Twitter and considers a tender offer.

The HCL Tech report card has been released: Should you buy, sell, or keep the stock?

The Adani Group has purchased a 100 percent share in Ocean Sparkle, a marine services company.

In Delhi, a 24-year-old woman was stabbed to death.
Now, the Delhi BJP is launching an anti-encroachment campaign in these areas.

Sachin Pilot arrives in New Delhi for a meeting with Sonia Gandhi.

Delhi: Ambedkar schools hold parent-teacher meetings, and children are assessed.

PM Modi meets with UK Prime Minister Boris Johnson at Rashtrapati Bhavan.


Russia declares Mariupol "liberated," while the United States rushes further aid to Ukraine.

At a power show, Imran Khan praises India once more and demands for new elections in Pakistan.

In the Abu Salem case, the Supreme Court urges the Home Secretary not to lecture us.


India condemns US Representative Ilhan Omar's visit to Pakistan-controlled Kashmir.

The new Arunachal CS is well-known for following the rules.

"In India, no one will go to bed hungry if...": Gautam Adani

A Kishor PowerPoint from the past surfaced, calling for a "non-Gandhi party chief."

'I don't know who he is,' Assam Chief Minister Himanta Biswa Sarma said of Jignesh Mevani's arrest.


Three militants have been killed in the ongoing gunfire in Baramulla, according to authorities.


PM Modi said Guru Tegh Bahadur was a rock in the face of Aurangzeb's oppression.

'Pace isn't that crucial,' says Kapil Dev of SRH fast Umran Malik's rapid rise in the IPL 2022.

Rohit Sharma, Jasprit Bumrah Among Wisden's 5 Cricketers Of The Year Rohit

Wisden's 5 Cricketers Of The Year include Rohit Sharma and Jasprit Bumrah.


The glimpse of Kiara Advani in Bhool Bhulaiyya 2 would send shivers down your spine. Watch


Malti Marie Chopra Jonas is the daughter of Priyanka Chopra and Nick Jonas, according to reports.


Because attrition is significant, Infosys uses a job clause to keep employees.


Carlyle has been hired by JSW to support its bid for Holcim's Indian cement operations.

During the fourth quarter, Rakesh Jhunjhunwala sells his position in this PSU metal stock.

A BJP leader got brutally murdered outside his residence in Delhi.

As the stock achieves a new high, Reliance Industries (RIL) is 'burning on all cylinders.'

Imran Khan accuses Pakistan Army Chief for his dismissal in a veiled attack.

Russia fires a nuclear-capable missile that Putin claims is the best in the world | WATCH

The Supreme Court has commuted a rape convict's death sentence to allow for his rehabilitation.

Babita Phogat ignites a storm by saying, "Everyone knows whose civilization instigates riots."

Check out these last-minute revision ideas to help you ace the CBSE Class 10 examinations in 2022.


With the AAP on its tail, the Haryana Congress is getting its house in order.

Karnataka To Check Tenders Over $50 Million After Contractor's Suicide

The chief cop in Nashik has changed his mind over the azaan controversy.

Six TRS leaders have been detained after a mother-son duo committed suicide.

India is wooing travellers for traditional therapy with the 'Ayush visa.'

A shootout breaks out in Baramulla, north Kashmir, injuring three soldiers and a civilian.

Jitu Chaudhary, a BJP activist, was shot and killed outside his Mayur Vihar home.

In Mumbai, India receives its sixth Scorpene-class submarine, the INS Vagsheer.


At Delhi Airport, gold worth 30 lakh was discovered hidden beneath a wig.


The sinking of the Russian ship Moskva is being investigated by the Indian Navy.


Boris Johnson's visit to India signals a push for easier visas as part of a trade deal: 10 Facts

Suryakumar Yadav On IPL 2020 Face-Off: "Virat Kohli's Sledging Was On Another Level"

Ravi Shastri and Kevin Pietersen Explain Why Former RCB Captain Virat Kohli Needs A Break

Ronaldo will miss Man United's match against Liverpool due to the death of a newborn twin.

Maharashtra Sikh Group Slams "The Delhi Files" as Filmmaker Announces It


Vicky Kaushal is flexing his abs casually, and his father Sham is "proud of his puttar."




Abu Jani is now primarily owned by Reliance Brands. Sandeep Khosla is an Indian businessman.

With a rise of 62 points on the SGX Nifty, the wider index in India is off to a solid start.

Elon Musk says the "Woke Mind Virus" is making Netflix unwatchable.

Webinars on CUET and UG admissions will be held by Delhi University.



Between Kalindi Kunj and Maharani Bagh, a four-lane service road will be built in Delhi.



India's most sustainable corporation is Grasim Industries.

The heat wave is expected to subside as the IMD forecasts rain and thunderstorms in numerous states.



'Kemcho...majamma,' says the WHO chief as he and Prime Minister Modi launch a medical centre.

After clashes in states, Yogi Adityanath issued an order banning religious processions.

Six leaders from Chief Minister KCR's party have been arrested for mother-son suicide.


According to a member of the WHO expert panel, India's claim of an excess of COVID deaths is false.

Prashant Kishor to Join Congress: How the Party Finally Made the Decision


Today in Delhi, there are 632 new Covid cases, and the positivity rate has dropped to 4.42 percent.




According to DU V-C, Stephen is adamant about all candidates having an interview.


Haryana Health Minister Anil Vij has made masks essential in four Haryana districts bordering Delhi.

"The Karnataka government is slashing grants for mutts by 30%," says a source.



Sunil Gavaskar's Prediction For Young SRH Pacer: "He'll Play For India"


'I am in love,' says Vivian Dsena, who is about to marry Egyptian journalist Nouran Aly.



Jayeshbhai AKA Ranveer Singh Sent Anushka Sharma A "Special Letter" And Soap

Can the Nifty50 overcome the HDFC Bank and Infosys shockers on April 19?

On April 19, the price of 24 carat and 22 carat gold in India increased.

Elon Musk takes aim at the Twitter board of directors, saying, "Their salary will be $0 if..."



The Sinking of Russia's Flagship: Putin's Biggest Setback? - Nidhi Razdan on 'Hot Mic'

In the midst of the crisis, Sri Lanka's Rajapaksa admits to making mistakes.


"Pyramid Fraud": Consumer Goods Seller Amway India Frozen 750 Crore

Shehbaz Sharif, the Pakistani Prime Minister, has charted his own path with India.




Today, there are 1,247 new Covid cases in India, a significant decrease from yesterday's total.

Vishwa Deenadayalan, a table tennis player, died in a car accident on her route to a tournament.



On weak global cues, the Sensex drops over 1,000 points, while the Nifty trades below 17,200.

During the day, Delhi is likely to experience scorching conditions.

Initiatives like Hunar Haat are helping to strengthen Aatmanirbhar Bharat. Thakur

Millind Gaba, a singer, marries Pria Beniwal, his fiancée, in Delhi.

Thana is attacked by a mob in Karnataka because of a WhatsApp post; 40 people are detained.



'Speaks against terror, yet releases terrorists,' says Nadda of Congress.

Day 4 of KGF Chapter 2's box office collection: Yash's film surpasses Rs 500 crore.

Market LIVE: Sensex slides 1200 points, Nifty falls below 17,200; Infosys falls 7%.


After a sting operation, the BJP expels four Delhi councillors on corruption charges.

In Hubballi, Karnataka, there was a riot over a WhatsApp status, and 40 individuals were arrested.




With 2,183 new cases, India's daily Covid count has increased by 90%.



The first day of box office collection for Vijay's Beast is Rs 38 crore.


In India, gold prices have risen for the second day in a row; check rates in your city.

Elon Musk Calls US Regulators "B*****ds" Over Tesla Fraud Settlement

This summer, India faces extensive outages.


Students are boycotting e-classes because Jamia has yet to reopen.

There aren't many takers for paid booster, so don't be complacent: AIIMS

Stephen's College was not informed of the 85:15 admittance policy: DU V-C

Former AAP councillor receives court relief in Delhi.

Ex-wife Reham's jibe: Imran Khan might replace Navjot Sidhu in Kapil Sharma's comic show.

What Education Minister Manish Sisodia Said About The Fee Increases at Delhi's Private Schools


Number three: "KGF: Chapter 3" is teased by the makers.


Next year, the CBSE will reintroduce a single board test.

Bengaluru is hit by heavy rains for the second day in a row.


In a clash in Shopian, four militants were killed.



Real Madrid vs. Chelsea, 2 - 3; 2022 Champions League Quarter Final 2nd Leg

IPL 2022 Points Table Update: CSK vs RCB Match 22: Latest Orange Cap, Purple Cap Lists


With the romantic Brahmastra video, Ayan Mukerji confirms Ranbir Kapoor and Alia Bhatt's wedding.

Based on her airport appearance, the internet believes Katrina Kaif is pregnant.

Fans of Thalapathy Vijay call the action-thriller 'Mindblowing' and 'Blockbuster' on Twitter.




Hariom Pipe Industries begins trading at a premium of 43% to the issue price.

Infosys, HDFC Bank, Tinplate, Brightcom, Vedanta, and Thermax are stocks to keep an eye on.

As March inflation rises to 6.95 percent, the RBI may be forced to raise rates sooner than expected.




The Congress will soon depart Lutyens' Delhi bungalow, which houses the Sewa Dal headquarters.

Delhi BJP MPs evaluated social media performance during a meeting with Prime Minister Modi.

Drivers' unions in Delhi are planning a two-day chakka congestion.


Ukraine's Zelensky mocks Putin for claiming that the war will be "planned."


'No violence on Ram Navami,' said Yogi Adityanath of 'progressive' Uttar Pradesh.

S Jaishankar, India's Foreign Minister, explains what drew him to diplomacy.


TS decides to buy paddy at MSP, and GO 111 is revoked.


Sri Lanka declares a $51 billion debt default to deal with the situation.


Suicide Message Said "He's Responsible" in Case Against Karnataka Minister

SpiceJet has barred 90 of its pilots from flying Boeing 737 Max aircraft. This is why:


Due to a hand injury, Washington is expected to miss at least two IPL games.


Kane Williamson shines as SunRisers Hyderabad defeat Gujarat Titans by eight wickets in IPL 2022.


Gold price in India remains constant today

The TRAI proposes a significant reduction in the base price of the 5G spectrum band.

The Sensex and Nifty are down 1%, and there are four causes dragging the market down.

Elon Musk's "Chilling Effect" on Twitter Employees


The Supreme Court has postponed hearings on Bikram Majithia's appeal.


The House of Tamil Nadu has passed a motion asking the Centre to abolish the CUET.






KGF 2: The Tsunami is out in theatres now, and Jr NTR and Ram Charan are new sensations.

Uneasy quiet in Gujarat towns a day after Ram Navami clashes


India's ties with Russia developed when…: Top US diplomat at 2+2

Good Relationships with India Not Possible Until Kashmir Is Resolved: Pakistan's New Prime Minister

After getting off another train, five people were run over by a train in Andhra Pradesh.

According to CBI sources, fugitive Nirav Modi's close aide was brought to India from Cairo.



The Kuldeep Yadav Renaissance is still going strong.







Shiv Subrahmanyam, Alia Bhatt and Arjun Kapoor's co-star in the film "2 States," has died.

Alia Bhatt reacted to a YouTuber's ROFL "Alia Weds Ranbir" post with this comment.

Elon Musk has "decided not to join our board of directors," according to Twitter CEO Parag Agrawal.

Top ten things to know before the market opens today

The stocks that will be traded on April 11th are listed below.
TCS, Tata Motors, Tata Steel, Axis Bank, and Vodafone Idea are stocks to keep an eye on.




The Swiss will put India's anti-money laundering legislation to the test.
After Imran Khan's fall, the phrase "Chowkidar chor hai" was raised against the Pakistan Army. Watch


Shah Rukh Khan makes an appearance in Mumbai with his children Suhana and AbRam.

After the new PCC leader was appointed by Congress, there was resentment in the Sidhu camp.


For the fourth day in a row, Delhi has seen extreme heat.

COVID-19 cases have increased by 141 in Delhi.

How India's much-touted participation in global bond indexes fizzled out

Terrorist Funding Case | SIA searches in Delhi and Faridabad in its first operation outside of J&K.

Telangana chief minister to lead TRS protest in Delhi; wants 100 percent 'Punjab-style' procurement

With Ukraine and 2+2 in the background, Modi and Biden will meet virtually today.

Shehbaz Sharif has been nominated for the position of Prime Minister of Pakistan.




Yuzvendra Chahal's Shocking Revelation: "Drunk Player Dangled Me From Balcony"






Here's Why Shahid Kapoor Won't Talk About Alia Bhatt And Ranbir Kapoor's Wedding

Day 14 box office receipts for RRR: SS Rajamouli's flick The Rs 1000 crore mark has been reached.

Priyanka Chopra soaking in the sun in a yellow kurta in Los Angeles. View Stunning Photographs





Despite Moscow's warning, India abstains from voting at the UNGA.


In the United States, a bill has been filed to provide H-1B spouses the automatic right to work.


During Imran Khan's reign, Bushra Bibi's companion Farah Khan's wealth skyrocketed: Report

The accused in the Gorakhnath temple attack says he was harassed in a video.


LIVE Coronavirus Updates: India has 1,109 new Coronavirus cases, up 7.3 percent from yesterday.

The conversation between Shashi Tharoor and NCP MP Supriya Sule in the Lok Sabha has sparked memes.


The court instructs the CBI head to remove the lookout and apologise to Amnesty India's Aakar Patel.

People from various states should communicate in Hindi rather than English: Shah, Amit


"Until the Last Ball," says a defiant Imran Khan, who faces a no-confidence vote tomorrow.



Bayern Munich's Champions League defeat to Villarreal earned them match awards.


Aamir Khan, Ram Charan, Jr NTR, and Palak Tiwari attend the RRR 1000 crore success event.


Zee, Bandhan Bank, Titan, Uma Exports, HDFC Bank, and Other Stocks to Watch Today



Punjab's Power Minister meets with the Union Coal Minister and the Union Power Minister in Delhi.


'If the mayor of South Delhi wants to close meat shops, he should also close the slaughterhouse.'

Congress must abandon'soft Hindutva,' according to Sitaram Yechury.


Modi warns BJP members against dynastic politics on the party's founding day.

The HDFC Bank-HDFC Merger Signals The End Of The Golden Age

CUET 2022 Application Process Begins At Cuet.samarth.ac.in; Learn More About Registration


'What Was the Need?' Sharad Pawar Slams ED Action Against Sanjay Raut in One-on-One With PM Modi

What are WMDs, and what is the existing law that India seeks to change?



'Food subsidies kept severe poverty low during the pandemic,' according to an IMF working paper.

State-sponsored hackers targeted a power grid near Ladakh, according to a report.

What is the XE form of coronavirus, and why should you be concerned about it - at least for now?

Shoaib Akhtar names the player who should have played more games for India in the IPL 2022.


Chaitanya and Venkat Prabhu's Film Has Been Announced


RRR Day 12 Box Office (All Languages): Ram Charan and Jr NTR Starrer Crosses 650 Crores!

Hrithik Roshan and Saba Azad partied in Goa with Sussanne Khan, his ex-wife, and Arslan Goni.

TCS, Adani Group, Zomato, Marico, and Other Stocks to Watch Today


Petrol and Diesel are nearly a rupee more expensive, and this is the 14th hike in 16 days.



IMI is giving away souvenirs to commemorate 30 years of achievement and the Azadi Ka Amrit Mahotsav.

After ten years, Parliament passes a bill to reunify three Delhi corporations.


In parts of Delhi's north corporation, e-food carts are permitted.

Admission to all DU UG courses using CUET: Yogesh, Yogesh, Yogesh, Yogesh, Yogesh

Ramzan circulars for employees are issued by the Jal Board and the NDMC, then withdrawn.
The Congress has drawn up a religious itinerary ahead of the Madhya Pradesh elections.



The 13 new districts of Andhra Pradesh are explained, as well as why they were created.


The CUET admission policy for 2022-23 has been revealed by Delhi University.




Aakar Patel, the former head of Amnesty India, was stopped at Bengaluru Airport.


"The Constitution permits me to...": Trinamool MP Slams Meat Shop Ban



Updated IPL 2022 Points Table: Orange Cap, Purple Cap List Following SRH vs LSG


Fans are dissatisfied with on-field umpiring in the first Test between South Africa and Bangladesh.

IPL 2022: Kaviya Maran's photos go viral, and fans claim she "deserves better."

Poonam Pandey Keeps Her Promise To Take Off Her T-Shirt In Front Of The Camera; What's The Catch?



Day 11 of the RRR box office collection: Jr NTR and Ram Charan's film earns Rs 900 crore worldwide.







Why are HDFC Limited and HDFC Bank merging, and what will the consequences be?

With a rise of 89 points on the SGX Nifty, the wider index in India is off to a solid start.

Debris from a 'China Rocket' was discovered in a Maharashtra village.


The mayors of South and East Delhi have ordered that meat shops be closed during Navratri.

Petrol and diesel prices have risen by over ten percent in just two weeks.





Updates on the Russia-Ukraine Conflict: Biden Outlines Russia's Next Phase Of Sanctions


With a new ball, he always bowled brilliantly and swung it both ways: Arora, Vaibhav

'I'm sure he'll come good,' says skipper Jadeja after a crushing defeat to Punjab Kings.

Alyssa Healy's journey from tragedy to success


IPL 2022: Ravi Shastri Calls Gujarat Titans Star "One Of The Most Talented Players In World Cricket"




BTS performs "Smooth as Butter" on the Grammy Awards stage in 2022.

Pregnant In a white saree, Sonam Kapoor flaunts her baby bump.



The Sensex rises 1,400 points to 60,000; the Nifty reaches 18,000; and HDFC shares rise 14 percent.

The SGX Nifty has dropped 35 points; here's what happened in the market while you slept.

In Q4FY22, HDFC Bank's advances increased by 21%.


Police in Delhi bust a Chinese loan app fraud ring, arresting eight people.


Taking a Closer Look: A Market Makeover

The Hindu Mahapanchayat at Burari Ground has sparked a fight, prompting the police to intervene.

Pawar says he has no desire to lead the UPA.






Petrol and diesel prices have risen for the 12th time in two weeks.

Army has begun court martial proceedings against an officer engaged in the Amshipora encounter.

Since the peak of the third wave in January 17-23, weekly Covid cases have been declining.


Following the dissolution of Parliament, Pakistan's Prime Minister Imran Khan was denotified.

Sri Lanka's economic crisis has resulted in the resignation of all 26 cabinet ministers.


What should Supertech homebuyers do if they've filed an RERA complaint?





Day 1 Box Office Estimate: The John Abraham-starrer gets off to a poor start, with Rs. 3 crore.


This March was the hottest in India in 122 years.

Was Shashi Tharoor a child actor in this film? A member of the Congress clarifies

Day 8 of the RRR Box Office (Hindi) Early Trends: Attacks 'Attack' & Recovers


Bulldozers arrive at the doorsteps of criminal suspects in Uttar Pradesh, forcing them to surrender.

NCB witness Prabhakar Sail died of a heart attack in the Aryan Khan case.



After The Kashmir Files, RRR Box Office Day 6 (Hindi) Is Set To Be Yet Another 200 Crore Club Hit.


"Thank You For Your Resilience," says Chris Rock, in response to Will Smith's slap.

Explained: Why are e-scooters so popular in India?

The Congress 'worships' cylinders, while Rahul Gandhi roasts the BJP over fuel prices.

GAIL announced a Rs. 1,083 crore buyback programme at a price of Rs. 190 per share.

Near Delhi, a Driver Deliberately Runs Over an Elderly Man on Camera

Karnataka Chief Minister to Investigate "Serious Objections" to Halal Meat

"I'm willing to die for the country," Arvind Kejriwal said in response to BJP vandalism at home.

Mamata protests the BJP's request for a CBI investigation, calling it "intervention."

Shivpal Yadav talks with the Uttar Pradesh Chief Minister, sparking curiosity about his next move.

When most people turned their backs, a good Samaritan put his life on the line – and died.

Doctor commits suicide after being charged with murder in the death of a patient.

"Those who drink are Mahapaapi, not Indians," Nitish Kumar says.

The Support of Muslim Vendors by Two Karnataka BJP Leaders for Election Victory

IE100: India's Most Powerful People in 2022

Imran loses the bulk of his votes and is about to be knocked out.




Australia advances to the seventh final thanks to Healy's 129 and Haynes' 85 points.

IPL 2022: Ravi Shastri on Sanju Samson: "Has Power To Clear Any Ground In The World"


Dia Mirza wished her stepdaughter Samaira a birthday like this.


Today's Leo horoscope for March 30th, 2022: Your career will take off!

Will Smith's Mother On The Actor Slapping Chris Rock At The Oscars: "First Time In His Lifetime"


In one of the largest crypto heists ever, hackers stole almost $600 million.

After 'encouraging' Russia-Ukraine talks, stocks are up and oil is down.

Citibank is expected to sell its consumer banking division in India to Axis Bank today.

Petrol and diesel prices have risen by about a rupee for the eighth time in nine days.



The National School of Drama's 'Abhijnanashakuntalam' is a theatrical retelling of a classic.


Summer power shortages are expected due to Haryana's allocation of 728MW capacity.

The Weather Department has issued a warning for a "severe heat wave" in Delhi today and tomorrow.


Nilim Mahanta and Ebo Mili have been granted bail by the court.



BJP on Didi's bid: A ploy to shift focus away from Birbhum

Assam and Meghalaya sign a border-resolution accord in six sites; Amit Shah calls it "historic."



Journalist Rana Ayyub Was Stopped From Flying To London At Mumbai Airport

Two Lashkar terrorists were killed in J&K, one of whom had a "Press Card," according to police.





Mitchell Marsh will miss the first ODI due to injury; Inglis tests positive for Covid

Hardik Pandya admits being out to brother Krunal Pandya 'hurt worse' in IPL 2022, WATCH








Day 4 of the RRR box office collection: Jr NTR and Ram Charan's film continues to do well.


Today's Stock Market: Everything You Need To Know Before Trading On March 29

EV fire in Ola, Okinawa: The centre dispatches a team of professionals to investigate the situation.

Tata Power, Aurobindo, SBI Life, Power Grid, and Other Stocks to Watch Today



Imran accuses foreigners of causing his problems.

The "Butcher" remark made by Biden against Putin is "certainly alarming," according to the Kremlin.

Imran Khan is on the defensive after a minister resigns and the BAP joins the opposition.

According to a report, a Russian billionaire and Ukraine peace negotiators were poisoned.




Child Rights Organization Has Concerns About Arvind Kejriwal's Budget Remarks in Delhi


Five BJP MLAs have been suspended after fists were raised in Bengal House over the Birbhum deaths.

In the midst of the Ukraine crisis, Russia's foreign minister may visit India this week.

For India, the United States Relaxes Its Covid Travel Rating. The Implications

The hottest day of the year has arrived in Delhi, with temperatures 10 degrees above normal.

Why is Amit Shah's relocation to Chandigarh a red zone for Punjab?




What Mithali Raj Said About No-Ball In The Women's World Cup Final Against South Africa



'Mithali Raj and Jhulan Goswami should be at the end of their tether.'



Yash and Sanjay Dutt face battle in a 'storey written in blood' in KGF Chapter 2 trailer.

The 94th Academy Awards Winners List: Coda Wins Best Picture, Will Smith Wins Best Actor


Ashneer Grover regrets not investing in Sippline's 'glass ka mask' on Shark Tank India.

Day 17 Box Office Estimate: The Kashmir Files is expected to gross Rs. 9 crore.

Traffic cops will be worried for hours thanks to Zomato's 10-minute delivery.

Today's Stock Market: Everything You Need To Know Before Trading On March 28

On March 28th, compare gold prices for 22 carat and 24 carat gold.

PVR, Inox, Wipro, GAIL India, Emami, and Other Stocks to Watch Today

Petrol and diesel prices have risen for the sixth time in a week.

These are the top 10 things to know before the market opens today.

Ranjit Manchanda, a UK-based doctor, has been appointed to the AIIMS Infosys Oncology Chair.

UP: Police said two Muslim youths were killed by their neighbours for celebrating the BJP's victory.

Victims' families hope to return to Birbhum to rebuild their lives and houses.

Covid tolls are at 2-year lows: daily tolls are below 10, and weekly tolls are below 100.




AAP's Sisodia said the BJP is afraid after Amit Shah's 'positive news' in Chandigarh.

Banking Services Have Been Affected Partially by the 2-Day Bharat Bandh: 10 Points



IMD anticipates a heat wave in various areas during the next five days.

India has resumed regular international flights after a two-year hiatus.

Chris Woakes has gone from being a school bully to a bearded brawler.

After the Covid disaster, the Indian Premier League enters a vital season.

'Soft dismissals were the reason we lost,' says Babar Azam.

Despite the rebel attack, the Saudi Arabian Grand Prix will go on "as planned." The


IPL 2022: New CSK skipper Ravindra Jadeja is unconcerned about having MS Dhoni around.



'Kashmir Files Ne Meri Film Ko Bhi Duba Diya,' says Akshay Kumar; Vivek Agnihotri reacts.

'RRR' Day 1 Box Office Collections: SS. Rajamouli's film earns over Rs 240 crores; find out more!

Anil Ambani has resigned as the director of RInfra and RPower.


The government is committed to reducing taxes: FM Nirmala Sitharaman is an Indian politician.

In India, the price of 10 grammes of 24 carat and 22 carat gold has increased by Rs 70.

Prices of gasoline and diesel have risen for the fourth time in five days.

SC: It's not acceptable to shut down units because of a minor technical issue.

Sanjay Kumar of GAIL has been named IGL's new CEO.


Hate speech case: If spoken with a smile, there is no illegality, according to the HC.

Navjot Singh Sidhu is chastised by the Supreme Court, which has reserved its decision.

Arvind Kejriwal's remark on "The Kashmir Files" is mocked by Vivek Agnihotri.

Uddhav Thackeray on ED move against his brother-in-law: "Jail me, but don't tarnish my family."


With a proposal at Delhi Airport, an India-Ukraine love storey comes to a happy conclusion.

In the 52-strong Yogi ministry, 32 freshmen were appointed, while one deputy CM was dismissed.

With the PLA dragging its feet, resolving the India-China border is proving difficult.

Ex-MLA Bhagwant Mann's Clean Sweep On Pension Formula

Putin's conflict threatens to suffocate India's $81 billion economy in the region.

Live Updates On The Russia-Ukraine Crisis: Russia Indicates Less Ambitious Goals In The Ukraine War

Shane Watson Explains Why He Couldn't Say No To Coaching The Delhi Capitals In IPL 2022

Sunil Gavaskar names one team that is unlikely to win the IPL in 2022 as "no impact player."

MS Dhoni's CSK Captaincy Stint In Numbers In IPL 2022

Day 14 box office collection for The Kashmir Files: Vivek Agnihotri's film collects Rs 207 crore

Mom-To-Be In a yellow gown, Sonam Kapoor flaunts her pregnancy glow.

The Kapil Sharma Show will be taken off the air for a while.

For Her Bengali 'Godh Bharai' Ceremony, 'Saadh,' Debina Bonnerjee Wears A Red Suit And 'Sindoor'

Priyanka Chopra hosts an event honouring South Asian nominees ahead vof the Oscars in 2022.


In India, the price of 10 grammes of 24 carat and 22 carat gold has increased by Rs 180.

Russian firms on the West's blacklist are avoided by Indian banks.

Before the market begins today, here are 10 things to know.

From next week, the weather in Delhi will become considerably hotter.

Pandits in J&K have petitioned the Supreme Court to stop what they call "genocide."


The Pegasus panel is seeking public input.

National anthem recitals are now required in all madrasas in Uttar Pradesh.


Arvind Kejriwal on the Delhi Polls: "A Phone Call From The PMO Was Made"

Police file a FIR after a Kashmiri businessman is denied a hotel room; OYO delists it.

In Maharashtra, a man was killed by a teen who had broken his phone.

There's no link between hijab and exams, so don't make a big deal out of it: CJI




The Indian government's stance on the Ukraine issue is based on six principles: Jaishankar, S


Mamata Banerjee, the Chief Minister of Bengal, visits Birbhum village and arrests a TMC neta.

Ten New Developments in Ukraine's War



According to BCCI secretary Jay Shah, IPL revenue is reinvested in the game.

The Kolkata Knight Riders put their faith in Iyer's ability.

Shane Watson's Response When Asked To Compare MS Dhoni And Rishabh Pant In IPL 2022

IPL 2022: The rest of the world catches up


Sonam Kapoor's mother-in-law expresses her joy following the actress' pregnancy announcement.



Today's horoscope: March 23, 2022 astrological forecast

Salman Khan has been summoned by the court for reportedly misbehaving with a journalist.

Ankur Tewari remembers Gully Boy rapper MC Tod Fod as "amazing."

Today's gold prices for 24 carat and 22 carat gold may be seen here.
The bond sell-off has pushed money back into stocks.

Get some popcorn because the Mukesh Ambani vs. Gautam Adani feud is heating up.




The Ashram underpass in Delhi has been opened to the public.

From Noida to Ghaziabad, these are the world's most polluted cities in India.

A car driver was killed in a crash on the Delhi-Meerut highway, and the BMW owner was arrested.

The EOW of the Delhi Police has filed a charge against eight real estate firms over a land issue.


Vaccine manufacturers have petitioned the Supreme Court to prevent the release of trial data.

AAP's Rajya Sabha Nominations Punjab's "Betrayal": Navjot Sidhu

Sonia Gandhi makes contact with the G-23 leaders.





3 to 1: MCD Merger Set to Change Power Dynamics in Delhi Ahead of 'Civic War'





11 Bihar workers were killed in a massive godown fire in Secunderabad, Telangana.

Before receiving the Padma Shri, Yoga Guru 125 bows to the Prime Minister and President.

Australia's excellent 272 chase is aided by Meg Lanning's magnificent hundred.

Virat Kohli Joins Royal Challengers Bangalore Squad Ahead Of IPL 2022 "King Arrives"

Dhoni and CSK: How the IPL hero's image grew beyond the confines of the city

FACTS AND STATS: Ferrari ends a 45-race winless streak in Bahrain, with Zhou scoring on his debut.

Naseem Shah said he had to "bowl our hearts out" to bring the Lahore field back to life.



Ajay Devgn explains why he wouldn't have made Runway 34 if Amitabh Bachchan had said no.

Zoom Backs Youth Leaders' Expansion of the Digital Champions Program for Active Citizenship

Biden cautions US businesses about possible Russian cyberattacks.


Due to high energy prices, Fitch reduces India's growth forecast for FY23 to 8.5 percent.

What is the controversy surrounding the Shivaji statue in Telangana's Bodhan town?

The mother of a baby girl found dead inside a microwave oven is being investigated by Delhi Police.

AIMIM MP claims Aurangzeb is not our icon and wants to meet Uddhav with an alliance offer.

Why was Nitin Gadkari dubbed "Spiderman" by a BJP MP in the Lok Sabha?




The G-23 Congress and Rahul Gandhi have both made similar requests. So, what exactly is the issue?

Sitharaman in the Rajya Sabha: For the past ten years, there have been no defence acquisitions.




Threats against Karnataka High Court judges are condemned by the Tamil Nadu Bar Council.

MP IAS officer suggests donating 'The Kashmir Files' earnings to charity; Vivek Agnihotri answers.






Rajamouli reveals his secret to success

Today's horoscope is for Capricorn, Aquarius, Pisces, Leo, and other signs born on March 21, 2022.

The Kashmir Files Day 10 Box Office (Early Trends): Its 'Out Of This World' Run Continues!


Ashneer Grover, a judge on Shark Tank India, reacts to Ashish Chanchlani's spoof of the show.

How the NSE was enveloped in scandal by India's trading queen and mysterious guru

A startup has been accused of committing a $400 million fraud.


The acquisition of Sintex by Reliance Industries has been approved by the CoC.

Changes to the Land Act are being worked on as quickly as possible.

Waste-to-fuel-to-waste: Delhi's firms are having a hard time finding buyers for RDF.

The Academic Council of DU will evaluate CUET for admissions beginning in the next academic session.

The ED will examine West Bengal Chief Minister Mamata Banerjee's nephew.

After a four-year search, a 27-year-old woman wanted for murder and kidnapping is arrested.

A 19-year-midnight old's run near Delhi becomes viral, garnering more than 4 million views.







On AIMIM's suggestion of a tie-up, NCP president Sharad Pawar says, "It's not our party's decision."

"Censoring Kashmir Files Is An Assault On Freedom," says New Zealand's former Deputy Prime Minister.

Cyclone Asani: Heavy rain and strong winds in the Andaman and Nicobar Islands - 10 updates



Ghulam Nabi Azad hints at a political'retirement,' but adds civil society has a big role to play.

Turkey claims Moscow and Kyiv are closer on critical issues, as a school gets bombed

Ferrari, according to Horner, "may be the car to beat."

Gayatri Gopichand and Treesa Jolly win the All England Open 2022 for the first time.

England is frustrated by Kraigg Brathwaite's and Jermaine Blackwood's twin tonnes.





A new Rs 16,635-crore plan will promote textile capex in the downstream value chain.

The West hasn't stopped buying Russian oil, but it is pressuring India to impose trade restrictions.

Oil-scarce countries do not need to advise on Russian imports, according to India.

SHIBA INU'S PRICE HAS INCREASED BY 10%, WHILE BITGERT HAS INCREASED BY 10%.

In just one day, these two stocks helped Rakesh Jhunjhunwala increase his wealth by Rs 861 crore.

Petrol and diesel prices in Mumbai, Delhi, Chennai, and other cities on March 19th.

Karnataka proposes to teach the Bhagavad Gita after Gujarat; the Congress thinks it isn't required.


BJP's jab at Omar Abdullah over Kashmir Files: 'That his father abandoned Hindus?'


With a deal that "avoids third parties," Iran is ready to meet India's energy needs.


Akshay Kumar and Kriti Sanon deliver bhaukaal wali Holi in Bachchhan Paandey.

First Covid deaths in China since January 2021; global daily cases up 12% | Top news

'India has the right to import Russian oil,' says the Indian government.



Kareena Kapoor Is Getting Ready To Make Her OTT Debut. She writes, "And So It Begins."



Oil dips below $100, easing the strain on gas stations.


The Federal Reserve is expected to hike interest rates in order to put inflation under control.

Zomato's stock is up 2% as a result of a possible merger with Blinkit.

Today's gold price in India for 10 grammes of 24 carat and 22 carat gold is

The temperature in Delhi may increase much more following the short Holi holiday.





Bhagwant Mann And "Rang De Basanti" At Bhagat Singh's Village

PM warns BJP netas against dynastic politics, calling it "undemocratic."



Police said a militant has been killed in the ongoing gunfire in Nowgam.

The Kashmir Files wins Rs 60.20 crore on Day 5 at the box office, besting pre-COVID blockbusters.


Sonia Gandhi has asked five state presidents to resign, and the G-23 will convene today.

Today is Bhagwant Mann's inauguration as Punjab Chief Minister at a historic village.

After reading Radhe Shyam's negative reviews, Prabhas' ardent fan commits suicide?



Alia Bhatt's meteoric rise: from SOTY's stilted Shanaya to the colossal Gangubai Kathiawadi

Brahmastra's First Look: Alia Bhatt as Isha


Rishabh Pant is ready to embrace anything comes his way: Rohit Sharma is an Indian cricketer.



Perry, Gardner, and Haynes defeat the West Indies to maintain Australia's unblemished record.



No comments from the RBI on data sharing or the Chinese perspective: Vijay Shekhar Sharma

These are the top ten things to know before the market opens today.

Future, Reliance On "Fraud" Accused In Amazon's "Public Notice" Newspaper Ads


Sameer Gehlaut, Indiabulls Housing Finance's Non-Executive Director, has resigned.

Four banks have agreed to invest in Amrapali housing developments.

Kavita Prakash takes a nature walk for nature lovers in Delhi.


AAP demands that the BJP announce poll dates within a week of the municipal elections in Delhi.


Delhi's highest maximum temperature this season is 33 degrees.


Outcry over modifications to the J&K delim draught




When BJP MPs chant 'Modi, Modi, Modi' as the PM enters the Lok Sabha, the opposition is taken aback.

China and India are under pressure to separate themselves from Russia.



Sonia Gandhi Admits "Shielding" Amarinder Singh Was a Bad Decision: Sources
COVID-19 vaccination is available for children aged 12 to 14 years old starting March 16th.



Karnataka: Today's HC hijab judgement, Sec 144 in Bengaluru and other parts of the state


Virat Kohli's teammates can't stop laughing as he tries to imitate Jasprit Bumrah's bowling action.


Cricket Twitter was envious of Virat Kohli's 'best view' of Rishabh Pant's sixes.


Vikram, Kamal Haasan's film, has a release date.

Day 3 Box Office Collection For Prabhas' Romantic Thriller Radhe Shyam: Fantastic Sunday!



Day 3 box office collection for The Kashmir Files: Vivek Agnihotri's film earns Rs 15.10 crore.

Aamir Khan has denied rumours that he divorced Kiran Rao because of an alleged infidelity.


According to a report, India is considering Rupee payments for trade with Russia.

Paytm's stock has dropped 13% to $685, or 70% from its issue price of $2150.

Before the market opens today, here are the top ten things to know.


Jyotiraditya Scindia's comments on the Madhya Pradesh elections in 2023, Congress

'No Yadav Belt, No Government': Why Akhilesh Yadav's SP Couldn't Take the BJP's Seat

The interest rates on non-withdrawable FDs at HDFC Bank have been revised; see the new rates here.

PM Narendra Modi gives a big thumbs up to Yogi Adityanath's leadership in Uttar Pradesh.

Cyber police question Fadnavis over a 'leak' of classified information.

For the first time, the 100-year-old RIMC has opened its doors to female students.

According to Congress politician Balbir Sidhu, the party authored its own defeat in Punjab.

In a crash near Toronto, Canada, five Indian students died.
Highlights of the Coronavirus: 3,116 New COVID-19 Cases in India

Three guys have been arrested after a group of men sexually harassed women in public.

Imran Khan on Pakistani Inflation: "I'm not in politics for aloo, tamatar rates."





The Delhi High Court explains why electronic voting machines (EVMs) are not used in civic elections.

Kejriwal asks to Prime Minister Modi to allow civic elections in Delhi to go place.

The Supreme Court has appointed ex-judge Sikri to lead the Char Dham project panel.



"Stop crying like the Congress," Manish Sisodia said of the BJP's stance on the Delhi election.

In Kulgam, south Kashmir, a Sarpanch was killed.


With 6 MLAs in Manipur, JDU has a sliver of hope.

Mayawati explains why some of her party's supporters voted for the BJP.
The Hindu-CSDS-Lokniti post-poll survey will be conducted in 2022.

After Dhami's defeat in Uttarakhand, there is uncertainty on who would be the next Chief Minister.

Respect voters, not arrogance: Punjab CM-elect Bhagwant Mann urges AAP MLAs

India claims a technical glitch caused the missile to land in Pakistan.

Akhilesh Yadav Ally on UP Loss: "Fell Short, Will Go Back To People"


All 403 Assembly seats in Uttar Pradesh were contested by the AAP, however none were won.



When the G23 leaders of Congress convene, they may decide to target party leadership.

The BJP is stuck between Dhami and Biren.

PM Modi pays a visit to his mother in Gujarat, a day after the BJP's massive victory in four states.

A major fire in Delhi's Gokalpuri has killed seven people and destroyed 60 huts.

5 reasons why the AAP is on track for a landslide victory in Punjab

BJP wipes out Akhilesh Yadav's hopes in Uttar Pradesh, with well over 250 votes.

Djokovic Withdraws From Indian Wells & Miami

On March 30, Shane Warne's state memorial will be held at the MCG.

Real Madrid rally behind Karim Benzema's hat trick to knock PSG out of the Champions League.




A fan offers Kartik Aaryan Rs 20 crore to marry her, but the actor's response wins hearts.




Ashneer Grover and his wife Madhuri Jain have paid an advance tax of Rs 8.2 crore.


Today's gold price falls by Rs 410 per 10 gramme for 24 carat and Rs 410 for 22 carat gold.

A DU college class aimed at "rewriting history" has raised some concerns.


Encroachments have been cleared from a major stretch leading to Greater Kailash-II in Delhi.

Any speech critical of government policy is not seditious, according to Justice Nageswara Rao.


An ex-IB officer is accused of raping a 17-year-old girl in Delhi.


CM N Biren Singh Leads In Heingang Constituency In Manipur Result 2022 LIVE:


Key developments in the ongoing confrontation between Russia and Ukraine


Indian students are on their way to Poland, where they will be flown home today.





Tejasswi Prakash Gives Beau Karan Kundrra a Glimpse of Quality Time






Petrol and diesel prices in Mumbai, Delhi, Chennai, and other cities on March 9th.

Sun Pharma, TCS, Indigo, Indus Towers, and Other Stocks to Watch Today

An ex-Intelligence Bureau officer is on the run after being accused of raping a Delhi minor.

The Supreme Court has asked the Centre to state its position on Abu Salem's prison sentence.

Armed forces MacGregor medal for four

Have provided women with the confidence to file complaints: The first lady in central Delhi DCP


Latest developments in the Russia-Ukraine conflict

I-T raids Aaditya's aide's home, and Sena claims it was an attack from Delhi.

After a two-year hiatus, international flights will resume on March 27.



Akhilesh claims EVM theft in UP polls, while poll panel maintains it was used for training.

PM Modi meets with his Dutch counterpart, Mark Rutte, to discuss the situation in Ukraine.

Akhilesh Yadav accused the BJP of stealing the votes of the people.

The clogged restrooms at Istanbul's airport could be linked to India.


Russia Proposes A Ceasefire In Ukraine In Order To Evacuate: 5 New Developments


Behind the scenes: two phone calls, twelve buses, and a difficult journey with Red Cross assistance

Ukraine will never be a victory for Vladimir Putin, according to Joe Biden.

Virat Kohli's Royal Challengers Bangalore will name a new captain on THIS day in the IPL 2022.

Despite Maroof's 78*, Australia's spinners restrict Pakistan to 190 runs.

During the Pakistan-Australia match, a fan has a special message for Virat Kohl Test of Rawalpindi

Sunil Gavaskar regrets his ill-advised remark on Shane Warne "In addition,

Karan Kundrra Is Charging a Bomb for Kangana Ranaut's Arrest; Find Out How Much He Makes



At This Mini Dostana Reunion, Shilpa Shetty And John Abraham Did The Obvious


With Gal Gadot, Alia Bhatt is about to make her big Hollywood debut. This Isn't a Test


The rupee could fall to an all-time low of Rs 80 per dollar, according to a report.

There are five reasons why Ukraine has been able to hold off Russian aggression.

As votes close, a Rs 6 increase in petrol costs is expected, with oil testing $139.

Russia issues a warning to the West, threatening $300 per barrel oil and gas supply cuts to the EU.


The drama of the Ashram underpass in Delhi is coming to an end.

The Supreme Court has ordered that Balwant Singh Rajoana's appeal be heard.

Modi's visit to Pune is a shot in the arm for the BJP ahead of important municipal elections.


How Punjab's politicians are dealing with the lull before the storm

Allowing additional attempts in Civil Service exams is a tough issue: UPSC to SC

Students in Sumy board buses and are forced to disembark.



Kuwait airlift to 'Operation Ganga': India's largest evacuation operation

Wishes Images, Whatsapp Messages, Status, Quotes, and Greetings for International Women's Day 2022

Live Updates on the Ukraine-Russia War: The Humanitarian Crisis Is Worsening As Attacks Continue


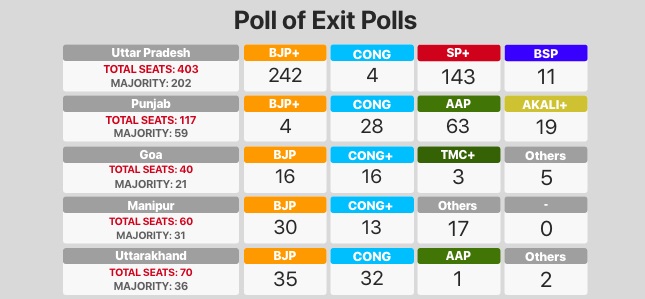
The BJP Wins UP Again, while the AAP Wins Punjab - Exit Polls

'Thinking of you,' says tennis great Novak Djokovic, as do football clubs.

IPL Schedule 2022: Dates, Times, Fixtures, Teams, and Venue Information Announced,

Ravichandran Ashwin Reveals How "Magnanimous" India Is Against Sri Lanka


Deepika Padukone teases a 'new house' in Spain as she posts photographs from Pathaan's session.



Did you know that Naseeruddin Shah has a disorder known as 'Onomatomania'?

Saba Azad, Hrithik Roshan's rumoured girlfriend, responded by calling him a "extraordinary human."


Will India's payment systems be able to withstand sanctions similar to those imposed by Russia?

The cost of gasoline and diesel is rising. Check out the rates in your area.

For the Reserve Bank of India, the oil shock might turn into a "nightmare."

Due to rising petroleum prices, the rupee is trading at a record low of 76.96 per dollar.

Former NSE MD Chitra Ramkrishna has been detained in connection with the NSE affair.


The Supreme Court has ruled that trial courts cannot convert a "life sentence" to "life in prison."

Candidates' spending limits have been raised to Rs 8 lakh in the Delhi civic elections.

Burari inhabitants avoid driving on Raahgiri Day to encourage fitness.


In 2.5 years, seven big cats perished at the Delhi Zoo, the most of them owing to 'kidney failure.'

J&K journalist Fahad Shah was detained for the third time just hours after being released on bail.


The IBP route connects the Brahmaputra to the Ganga.


Kerala Man Arrested In 6 Cases For 'Raped Me Holding Tattoo Needle To Spine'

Police have filed a UAPA complaint against ten suspects in the death of a Bajrang Dal worker.

BJP men climb atop AAP minister’s car, Arvind Kejriwal says party of lumpens

A grenade blast in Srinagar killed one civilian and injured 23 others.

Before evacuating an injured Indian student, Minister VK Singh reassures him.



India's representative in Ramallah, Mukul Arya, has died.


Shane Warne's final tweet, in which he pays tribute to Rod Marsh, has gone viral.

The cricket community is'shocked and heartbroken' by Shane Warne's death.



Jhund Day 1 Box Office (Early Trends): Amitabh Bachchan's Film Is Off To A Slow Start




'Are You So Dumb That...' says Sonakshi Sinha in response to wedding rumours with Salman Khan.

Bhavik Koladiya's shareholding in BharatPe has sparked a new feud.

A 'fortress economy' is seductive as globalisation is bombed, but India must understand its limits.

In Ukraine, Russia has taken control of Europe's largest nuclear power plant: 10 updates

Explained | How does the West intend to economically hurt Russia through sanctions?

Russia's oil consumers are running out.

We require world-class research institutes: Pinarayi Vijayan, Kerala's Chief Minister

The Delhi High Court has established three new courts to expedite the trial of pending UAPA cases.

Delhi: Is it true that children are not permitted to park in your area? DCPCR should be contacted.


"Be Strong," says the Indian Embassy to students in Sumy, Ukraine.

Exams for traditional courses will be held online at Mumbai University.

The Bone and Joint Hospital is engulfed in flames.

A Raipur journalist has been arrested for writing satires critical of the government.


Today, a skywalk connecting New Delhi Railway Station and a metro station nearby opens.


Prior to the last phase of the UP elections, Rahul and Priyanka give a 'hindutva' push.


Russia Closes In On Ukraine's Second Nuclear Plant: 5 Updates



For a healthy stomach, there are some dos and don'ts to follow.

World Obesity Day 2022: How to Lose Weight at Home Quickly and Safely

Sukhjeet, an Indian striker, aims to establish his place in the national squad.

Team India faces a plethora of issues in the Women's World Cup.

Sourav Ganguly goes against the grain and the regulations to attend selection sessions.





At Sanah Kapur's wedding, only Shahid Kapoor and Mira Rajput looked adorable.


'I don't care, it's innate,' Neelam Kothari said of being teased for her 'twang' accent.


Commodities are soaring as fears of supply shortages grow as a result of the war.

Everything You Need To Know Before Trading On March 4th

Vedanta, Vodafone Idea, IEX, and Other Stocks to Watch on March 4
PM Modi sees an opportunity to reduce budget gaps while also reducing oil use and emissions.

For investors, the NSE IFSC provides a myriad of options.

Before the market opens today, these are the 10 most popular things to know.




Russian troops attacked a nuclear power plant in Ukraine, but the fire was extinguished: 10 points

After abstentions, the US encourages India to distance itself from Russia in the Ukraine situation.

IAF avoids Pakistani airspace when transporting nationals from Ukraine's neighbouring countries.


R Priya will be the city's youngest and first Dalit woman mayor.

How Kerala's new push for higher education is a watershed moment for the ruling CPI (M)

The ED accepts Nawab Malik's payment of Rs 5 lakh, not Rs 55 lakh, as a typo.



PM to Students Returning From Ukraine: "Wouldn't Have Had To Go Abroad If..."



Union Minister: An Indian student was shot while attempting to flee Kyiv, Ukraine.

Minister Smriti Irani extends a warm welcome to Indians who have been evacuated from Ukraine.

India against. Sri Lanka: Team India's Test vice-captain Jasprit Bumrah

Mohali will allow a 50% audience for Virat Kohli's 100th Test match against Sri Lanka.

India against Sri Lanka: Virat Kohli is 38 runs away from becoming an India legend.

The 2022 Women's World Cup: Everything You Need to Know

'He was putting up hundreds every other innings,' Gavaskar says about Kohli ahead of the 100th Test.

Sporting legends in Russia have spoken out against the conflict.









Conflict between Russia and Ukraine: Things may not turn out as well for India as they appeared.

"Undesirable Colors To My Appointment": Why Did Air India's CEO Refuse the Position?

TMC wins 12 municipalities and leads in 34 in West Bengal Municipal Elections 2022.


Delhi News Live: 344 new Covid-19 cases are reported in Delhi, with a positive rate of 0.80%.

India tells Russia that international law and sovereignty must be respected.


On arrival at IGI airport in Delhi, a gangster who had been hiding in Thailand was arrested.

The C-17 airplane of the Indian Air Force takes off for Romania to transport Indians back to India.

How the conflict between Russia and Ukraine may affect MSME exporters to the region



LIVE: Russia assaults key Ukrainian cities, violence erupts near a military hospital in Kharkiv.

The Supreme Court decides to consider the Ministry firm's appeal.


Alumni are honoured at an event hosted by IIMC.

'Students from SOL are unable to attend classes.'

Mayank Agarwal has been selected the new captain of the Punjab Kings.

"Knocks of 18, 19, 20, and 30 are not enough," says ex-Pakistan captain Sanju Samson of India.

South Africa ties the series with a win thanks to Kagiso Rabada, Marco Jansen, and Keshav Maharaj.


It's illegal to play a dot ball in T20s: Iyer, Shreyas


Day 4 box office receipts for Bheemla Nayak: Pawan Kalyan's film is a hit






Russia is reeling under sanctions, with Putin branding the West as a "empire of falsehoods."

TCS will introduce a new operating structure that will be divided into four unique business groups.

Madhabi Puri Buch has been appointed as the new Sebi chairperson.


The ED discovers a Rs 200 million plot in Mumbai linked to Malik's son.

The CPM will seal 'pragmatism,' and the Vijayan authorities will attend the meeting.


Exams are the focus in Bengaluru's schools, which claim that hijab isn't an issue.

When Sonia Gandhi looked up Stalin's age on Google, she came up with a surprising result.

Is Mahashivratri a public holiday on March 1?


The cost of a commercial LPG cylinder has increased by Rs 105.

India is sending supplies to Ukraine today, with four ministers assisting in the evacuation.

Happy Maha Shivratri 2022: Wishes, Messages, Quotes, Images, Facebook & Whatsapp status


India demands an urgent end to the violence in Ukraine at a UNSC meeting.

LIVE NEWS FROM RUSSIA AND UKRAINE | Russia is bringing troops from the Far East closer to Europe.

In India, Google has launched its Play Pass subscription service, which includes over 1,000 apps.

Madhuri Dixit: I Never Lost Myself in Trying to Be the On-Screen Star | Exclusive


The winners of the SAG Awards in 2022 have been announced.

Day 3 box office earnings for Bheemla Nayak: Pawan Kalyan's flick surpasses Rs 100 crore

Munawar Faruqui tells Kangana Ranaut at the launch of 'Lock Upp,' "Dhamkiyan mat dijiye."

Samantha Ruth Prabhu on Alia Bhatt's Gangubai Kathiawadi: "A Masterpiece"

India's crude price has surpassed $100, implying a significant increase in fuel prices.

Vladimir Putin raises the ante on nuclear deterrence, while the West squeezes Russia's economy.




Mahashivratri 2022: These States' Banks Will Be Closed on Maha Shivratri, See the Complete List


Raja Bhaiya says the SP will not form a government in Uttar Pradesh.


As the Telangana Assembly Election approaches, Prashant Kishor begins groundwork for the TRS.

PM Modi's hit at Akhilesh Yadav: 'When people prayed for my death...'


The Mumbai power outage has been ordered to be investigated.


Headmistress is being held responsible for a student's suicide in Faridabad.

'Can't forsake our own people,' Rahul Gandhi says in response to a video showing Indians in Ukraine.


IND vs. SL: A World Record Path

Students are facing backlash in Ukraine as a result of India's UN move.

Today, Manipur voters will decide on 38 seats in the first phase of the election: 10 Facts

The US condemns Putin's nuclear directive as "unacceptable" and weighs more penalties.

The West has cut Russia off from SWIFT, and India is looking into other options.


India abstains on another UN vote on Ukraine and expresses support for the Moscow-Kyiv talks.


Hrithik Roshan becomes a supporter of rumoured girlfriend Saba Azad and writes her first post.


Day 2 of the Valimai box office collection: Ajith's record-breaking flick maintains its dream run.
Alia Bhatt's Gangubai Kathiawadi: Karan Johar's "Unbiased" Review

Lauren Down, a New Zealand batter, has been ruled out of the ICC Women's World Cup 2022.


Weekly Sports Newsletter: Why did Wriddhiman Saha receive support whereas Wasim Jaffer did not?


As South Africa seizes control, Kagiso Rabada devours New Zealand's openers.


The Anti-Corruption Bureau raids BBMP offices in large numbers.


The Haryana government advises realtors to pay EDC on time and concentrate on smaller projects.


Round 2 seat allotment results for NEET UG 2021 are now available.

Hemananda Biswal, Odisha's first tribal chief minister, has died.

A man jumps to his death from the 27th storey of a Parel high-rise in Mumbai.

SDMC directive states that no'religious apparel' is permitted in schools.



Develop indigenous weapons to stun enemies: PM

'Do not move to border posts without cooperation,' India advises Ukrainian citizens.

Why do Indians study medicine in Ukraine?

On Day 2 of the invasion, Ukrainian soldiers fight Russian troops in Kiev.




'We are all here...': Ukraine's President answers to Russian pressure in a video from Kyiv.

India explains its decision to abstain from voting on the Ukrainian invasion at the United Nations.



Jadeja Earns "Ravindra Pushpa" Tag After Celebrating Wicket With Allu Arjun Move

Ishan Kishan and Shreyas Iyer capture the day in the first T20I between India and Sri Lanka.


Here's the real storey of Gangubai Kathiawadi, as told by Alia Bhatt in her new film.




The collapse of the Russian stock market has wiped approximately $250 billion in value.


India is among Asia's worst losers as a result of the Russia-Ukraine crisis, according to a report.

The CBI has arrested an ex-NSE officer who was allegedly appointed on the advice of a yogi.

'I've lived through two wars,' says Anand Mahindra of the Russia-Ukraine crisis.


Yogi Adityanath is expected to run for governor of Uttar Pradesh in 2022.

Bikram Majithia surrenders in a drug case and is detained in Patiala.

A day before Gangubai's release, the Supreme Court denies a petition to stop it.

'Malik is intimidating officials, and the ED is likely to summon his son,' says a source.

As Russian invaders advance, Ukraine's president vows to remain in Kyiv.

PM Contacts Putin, Urging "Immediate Cessation Of Violence" in Ukraine

India specifies evacuation land routes as a result of late-night calls from the minister to Ukraine.



Lakshmibai College at DU intends to build a yagyashala where regular havans would be held.

T1 at Delhi Airport's new arrivals terminal is set to open today.

India against Sri Lanka: Is vice-captain Jasprit Bumrah a future skipper for Team India?

Franchises object to Mumbai Indians playing at Wankhede Stadium in IPL 2022.

Former India Selector Says Rahul Dravid Didn't Say Anything Wrong To Wriddhiman Saha

India wins a consolation match thanks to Mandhana, Harmanpreet, and Raj fifties.



Deepika Padukone was Alia Bhatt's plus one during the Gangubai Kathiawadi screening.




With a cheerful photo, Shabana Azmi welcomes Shibani Dandekar to the Akhtar family.

According to India Ratings, GDP advance estimates could be lowered to 8.6%.



Stocks rise as a result of the impact of Russian sanctions.

According to Priyanka Gandhi Vadra, the BJP wants the people of Uttar Pradesh to remain poor.

India's defence connections will not be affected by the tariffs, according to Russia.

During the Ukraine campaign, AI 1947 avoided risky airspace.

Joe Biden severs Moscow's financial ties with the West.

In the 4th phase of the UP elections, voter turnout was 60%, which was lower than in 2017.

RSS does not agree with the government on marriage age: it is a social, not a legal issue.
Nawab Malik's political trajectory from Sanjay Gandhi to Sharad Pawar

According to a Danish study, reinfection with distinct Omicron subtypes is conceivable.
How to Take Care of Your Eyes When You Have Diabetes



Afghanistan chooses to bat first, and Yasir Ali makes his debut for Bangladesh.






Why are economic sanctions on Russia expected to be even less successful in 2022?




In Delhi, a scholastic effort has been begun for 70 daycare houses.

The board will make a decision soon on the CBSE 10, 12 term 1 results.

Ukraine Live Updates: The United States has sanctioned Russia for "starting" an invasion of Ukraine.

In a case linked to the Mumbai underworld, a Maharashtra minister is being questioned.

A Shashi Tharoor Remark on Imran Khan's "TV Debate Challenge" to PM Modi

The purchase of Predator drones has been placed on hold.

In parts of Karnataka, more girls are removing their headscarf for school; protests continue.






5 micronutrients for women with gestational diabetes


Vijay Deverakonda Finally Reacts to Rashmika Mandanna's Pushpa Wedding Rumors


Today, the price of 24 carat and 22 carat gold in India falls by Rs 120.

Russian Roulette At The Opening Bell: 10 Things To Know

Delhi: Azadi Ka Amrit Mahotsav celebrations include a three-day craft expo.


Burari murder: victim was strangled before being raped.


At Delhi University's School of Open Learning, students demonstrate against "hooliganism."


Demolition crew lands initial strikes in countdown to fall of Noida twin buildings

The BJP's concern is that as the UP elections progress eastward, so does a herd of stray cattle.




Violence erupts in Karnataka a day after a Bajrang Dal leader is killed.

Covid cases number 13,405 in India, with 235 deaths; the active count has dropped below 2 lakh.

"We can't afford a military escalation," India tells the UN about the tensions in Ukraine.



The Delhi stadium has been added to the list of assets that can be sold.


Coronavirus symptoms: 6 signs of 'Stealth Omicron'


Everyone liked Suryakumar Yadav's 'Namaste' after a fiery half-century.

Sunil Gavaskar Evaluates Rohit Sharma's Time With Team India


Samantha steals the show in the first trailer for Shakuntalam.

Who was Gangubai Kathiawadi, and why is Alia Bhatt's film about her in trouble with the law?


Saba Azad, Hrithik's rumoured girlfriend, spent Sunday with the Roshans. See the image

Everything You Should Know Before Entering The Market On February 21


Before the market opens today, these are the 10 leading things to know.


Mekapati Goutham Reddy, an Andhra Pradesh minister, died of a heart attack.


The TRS chief meets with Thackeray and Pawar.

After the 'murder' of a Bajrang Dal worker in Shivamogga, Karnataka, security has been intensified.


Punjab voter turnout may not reach 2017 levels, and AAP strongholds are among the lowest.



Delhi's university ecosystem is buzzing, and there are high prospects for a comeback.

On the keto diet, there are five frequent mistakes that people make.

India against. West Indies: India dominates in the second T20I to clinch the series.



New Zealand takes a 387-run lead with to Nicholls 105 and Blundell 96.
Hrithik Roshan gives his uncommon blood type, and a 'Krrish' connection is discovered by a fan.


Samantha performs a sultry dance at the airport to Thalapathy Vijay's Halamithi Habibo song -



Reliance Industries, Future Retail, telecoms, Indian Oil, and ONGC are stocks to keep an eye on.



Top 10 things to know before the market opens today

Delhi takes issue with Singapore PM Lee Hsien Loong's remark on 'tainted' Indian lawmakers.

'Karhal is worth 300 seats,' says a rival for the Yadav fort.

The Supreme Court has upheld the minister's son's bail in the UP killings case.

LIVE UGC NET 2021 Result Updates: The NTA is expected to reveal the UGC-NET results today.

Why Was Amarinder Singh Removed As Punjab Chief Minister, According To Rahul Gandhi


Charanjit Singh Channi clarifies under fire, while Priyanka Gandhi uses her marriage as a shelter.

Over a Sedition Charge Against a Karnataka Minister, the Congress spends the night in the Assembly.


Singapore's Prime Minister's Remarks on Indian Members of Parliament: MEA summons envoy

The Congress sleeps over in the Assembly over a sedition charge against a Karnataka minister.

Punjab: Protest success does not translate into campaign victory; farmers have little clout.

Suspects loaded 3 kg of explosives into a Delhi flat using fake papers.

Plans for the Delhi Metro are on hold for the time being.

Police say a thief was apprehended and thrashed by residents of Noida, and he died as a result.

Some universities choose to provide students with a more flexible learning environment.

The rise and collapse of Gujarat's ABG Shipyard, which is now being investigated for fraud.


Rana Kapoor has been granted bail in the YES Bank fraud case.

Before the market begins today, here are ten things you should know.
Antibodies become increasingly powerful months after a COVID shot, according to a new study.

Google honours the developer of the chickenpox vaccine with a doodle




Bappi Lahiri's Mortal Remains Taken to Crematorium for Last Rites; Daughter Rema Devastated

Afghanistan's wider consequences for Central Asia: India at the United Nations

Russia's military withdrawal is being questioned by the West.


Priyanka Gandhi's slam at Himanta Biswa over Channi's 'bhaiyas' remark: 'Politics of tukde tukde...'


Official: EU loops India on Ukraine issues, warns of "huge consequences":

Keep 'UP, Bihar, and Delhi de bhaiye' out, says Charanjit Singh Channi.




Three clues that your headache is caused by an Omicron infection



The failure of Paytm and Zomato has forced Oyo Hotels and Delhivery to postpone their IPOs.

Is India's newest chip venture just a piece of paper?


With a swing of 3,500 points, the Sensex has experienced its sixth largest two-day swing.


Sandhya Mukhopadhyay, the singer who turned down the Padma Shri award, has died.

Deep Sidhu, an actor-activist suspected of the Red Fort riots, died in a car accident.

Resident Welfare Associations to Delhi LG: Relax orders for confinement zones and Covid families.

A timeline of Lalu Prasad Yadav's conviction in the fifth and last case of the fodder scam.


Punjab has a change vote, and AAP is the sole contender.


1st T20I between India and West Indies: The focus is fully on the T20 World Cup.


Retail inflation is at a seven-month high, and the WPI is in double digits for the tenth month.

Cryptocurrency prohibition is the'most prudent choice,' according to the RBI deputy governor.

LIC uses roadshows to entice worldwide investors to its IPO.

Ways to care for a child's mental health during treatment on International Childhood Cancer Day 2022
How does Covid contribute to stillbirths? Scientists have discovered
Is a re-infection of COVID associated with milder symptoms?

Randhir Kapoor wanted Kareena Kapoor and Karisma Kapoor to adopt him because they were wealthy.



Outstation final-year students at DU are pleading for a hybrid mode.

Muslim women have decided to support the BJP, according to Prime Minister Narendra Modi.

Covid-19: The first batch of Corbevax vaccine will be delivered to the centre today.

Hundreds of people cried as they said their goodbyes to Rahul Bajaj, from workers to ministers.


Allow us to wear hijab that matches the uniform colour: Karnataka HC students from Udupi




Arjun Tendulkar Attracts Two Bidders for IPL 2022 Auction, Eventually Bought By Mumbai Indians


Hijab will safeguard Muslim women from being raped, according to a Karnataka Congress MLA.

Support for political parties is divided along community lines in the Muslim belt.

During Modi's reign, there were Rs 5 billion in bank frauds, according to Rahul.


Rahul Bajaj, a prominent industrialist, was cremated in Pune with full state honours.



On board the PSLV C-52, ISRO successfully launched three satellites into orbit.

Strongholds of the Samajwadi Party in Uttar Pradesh, Uttarakhand, and Goa Vote: 10 Points

"If...," says Congress Leader Navjot Sidhu, "Navjot Sidhu will be appointed Super Chief Minister."

Stop it or we'll fining CAA protests: Supreme Court tells Team Yogi


LIVE CBSE Term 1 Result 2022: How and Where to Check Class 10, 12 Result

"Will remove PM Modi from power and install a government that will...", says KCR.
In India, there are now 50,407 new Covid cases, down 13% from yesterday.

At the Quad Meeting, India flags China's border moves as "when a large country..."

According to a local official, Chinese troops entered Indian land and drove away herders.

On Shivaji Park Memorial Row, Lata Mangeshkar's brother says, "Not Our Demand."


He describes Shibani Dandekar as "beautiful and exquisite" at his wedding.

Madhuri Jain of BharatPe may sue the auditor over a leaked report.

IRDAI clears draught papers for LIC Public Issue | Filing with SEBI is likely today.

The RBI chairman warns Indians against cryptocurrency, claiming that tulips are more valuable.

Madras High Court: Which is more important, country or religion?


‘We exist’: Rahul Gandhi's 'Union of cultures' tweet attacked by Northeast CMs

In reaction to a joke on Twitter, Ramdas Athawale points out an error in Shashi Tharoor's message.

In the first phase of the Uttar Pradesh election, 60% of eligible voters cast ballots.

Judge who issued the 'no skin-to-skin contact' order resigns after being denied elevation.

The son of a Union minister, Ashish Mishra, has been granted bail in the Lakhimpur Kheri violence.

One person is killed and another is trapped as the roof of a Gurgaon apartment building collapses.


Dhruv, Vikram's real-life son, lends his support to Vikram's star turn in Mahaan. Vikram
Deepika Padukone's "Biggest Cheerleader" is Ranveer Singh.

A businessman from Uttar Pradesh and his wife attempt suicide on Facebook Live, blaming the GST.

Two Dornier aircraft are being discussed with India: Sri Lankan radio station


The Congress's five MLAs joining the BJP-backed Meghalaya Alliance is described as "shocking."


Aaditya Thackeray of Sena says, "There Is No Place For Any Other Dress..." Row of Hijab

The second term of CBSE Class 10, 12 board exams will begin on April 26.

Here's what we know about the controversy surrounding the Chinese-made Equality Statue.

A three-judge panel, including the Chief Justice of Karnataka, will hear the hijab case.

Today's Mega Vote In The Heart Of The Farmers' Protest In Uttar Pradesh: 10 Points


Pat Cummins Breaks Silence On Justin Langer's Exit As Australia Head Coach: "To All Past Players..."




The AIMIM chairman takes a 'beti bachao, beti padhao' jab at Prime Minister Narendra Modi.


Arunachal Pradesh's Army has confirmed the deaths of seven soldiers died in an avalanche.

Here's everything you need to know about the CBSE Term 1 test 2022 results.


Domino's has now apologised for a "unsolicited social media message" about Kashmir.

Prime Minister Narendra Modi's remarks towards Telangana and Maharashtra have sparked outrage.

Man from Kerala is stranded on a mountain for two days without food or water.

'Indian officials must cease marginalising Muslim women,' Malala said of the Karnataka hijab issue.



Amit Shah will unveil the BJP manifesto in Lucknow today, ahead of the UP election.


PM Modi calls Congress's head a "tukde tukde gang" who relies on "division and rule."

As it becomes used to 'the new,' unfulfilled promises from Congress feed Malwa's rage.



Shah Rukh Khan's 'Dua' for Lata Mangeshkar elicits emotional reactions on Twitter.


In Andhra Pradesh, 9 people were killed in a car-truck collision.

How Rahul Gandhi Handled Difficult Situations Announcement of Navjot Sidhu vs. CS Channi

JNU will begin offering offline classes on Monday, but DU will remain online.

Amit Shah is likely to appear in Parliament about the recent attack on Owaisi's convoy.

Shah Rukh Khan, Ranbir Kapoor, Javed Akhtar, and others attend Lata Mangeshkar's burial.


As the number of Covid cases drops, schools and colleges in Delhi and four other states reopen.


Touching Navjot Sidhu's Feet, Charanjit Channi Sends A Message

India takes on England in the ICC U-19 World Cup Final, with everything going their way.

'Aluuuuu can't wait,' Katrina Kaif said of Alia Bhatt's Gangubai Kathiawadi.


Journalist held for anti-national social media posts: J&K Police

NEET-PG will be postponed by 6-8 weeks, according to the Centre.

Official Covid toll data are "not true," thus don't refuse ex gratia.

We Are Willing To Enact Even Tougher Social Media Regulations: Government

Navjot Sidhu Strikes As Rival CS Channi's Nephew Is Arrested

The Karnataka government insists on a uniform dress code and tells colleges to stick to it.

Two TRF militants were killed in a gunfight in Zakura, Srinagar, according to authorities.

After the earthquake in Afghanistan, strong tremors were felt in Delhi, Noida, and J&K.

In Galwan, China, 38 soldiers "panicked into retreat," according to a report.

Mayawati's electoral campaign in Uttar Pradesh is a curious instance.

MGNREGA wages worth Rs. 3,358 crore are yet to be paid.

On February 6, Rahul Gandhi is expected to announce his nominee for Punjab Chief Minister.

No one should attend school to practise their religion, according to a Karnataka minister.

Six people were murdered when a slab at a building under construction in Pune collapsed.

India's Covid Tally Drops Again with 1.49L Cases, according to Omicron LIVE Updates

Months of planning, then a devastating blast: Inside the US raid on ISIS's commander.

Anti-Hindu emotions harmed those who fired at Owaisi's car: police

A Central Agency Arrests Charanjit Channi's Nephew Ahead Of Punjab Elections

MS Dhoni Teases the Cover of His Graphic Novel "Atharva"

Priyanka Chopra's First Post After Giving Birth

According to Prime Minister Modi, the digital rupee will change fintech and create opportunities.

Before the market opens today, these are the top ten things to know.

UN Calls For An "Immediate" End To Violence In Myanmar

It is not advisable to label every man a rapist: Irani, Smriti

Amit Shah is accused of 'indecency' by Rahul Gandhi, to which Himanta Biswa Sarma responds.

BJP Leader Slams Priyanka Gandhi for Being "Hypocrite" After Her Husband Was Denied Ticket


According to an Australian newspaper, China sustained more casualties than previously stated.


Radhe Shyam, starring Prabhas and Pooja Hegde, will hit theatres on March 11th.

Myanmar's death toll has surpassed 1,500, with approximately 8,800 people detained by the UN.

It's no longer a 'experiment' after 16 female fighter pilots.

Aparna Yadav and Rita Bahuguna's son are among the BJP's Lucknow candidates.
Omicron Sub-Variant Found In 57 Countries Could Be More Infectious: WHO

Budget 2022: Politics at a distance, government banks on the ebb, and income trickles down


Congress Apologizes for Nirmala Sitharaman's "UP-Type" Remark

Covid-19 cases in Delhi have reduced to 2,779, with 38 deaths.

Before the opening bell on February 1st, there are ten things you should know.

This month is expected to be rainy than usual in north India.

Hindustani Bhau, a YouTuber, was detained in Dharavi, Mumbai, for 'instigating' pupils.

Make the Budget session productive, and don't let the polls affect it: Prime Minister Narendra Modi

Experts informed the SC panel that the digital trail points to Pegasus and the role of the state.

LIVE BLOG: Union Budget 2022: Cabinet Meeting Ends; Sensex Nears 59,000



India announces 2.09 lakh new Covid-19 cases, which is fewer than the number reported on Sunday.

Cyber experts tell the SC panel that there is proof of Pegasus use on phones.


Shweta Tiwari has been charged with injuring religious emotions with her 'bra' remark.

The Bengali Wedding of Mouni Roy and Suraj Nambiar


Bharti Airtel will receive a $1 billion investment from Google.

The S-400 Missile Deal with India "Shines Light On Russia's Destabilizing Role," according to the US

Why Is Mathura So Important For The BJP In Uttar Pradesh Elections?

Former Union Minister CM Ibrahim resigns from the Congress, claiming that the party "ignored me."

Prime Minister says he shares Central Asia's concerns about Afghanistan and terrorism.



An Indian family discovered dead along the US-Canada border has been recognised.


After a complaint from a filmmaker, a case has been filed against Google's Sundar Pichai in Mumbai.


Air India Will Offer This Service For The First Time Since Tata's Takeover: Report




Delhi is suffering from a chilly period, with relief uncertain today.

Women officers steal the show on R-Day, says Rafale pilot to contingent commanders

After regular market approval, Covishield and Covaxin could cost about 275 per dose.

India's positivity rate has risen to 19.5 percent, with 2.86 million new cases.



Swiggy is now a decacorn, with a valuation of $10.7 billion in a new investment round.
More selling is possible if the Nifty falls below the 17,000 level of support.

Nifty Trades Below 17,050: 10 Points; Sensex Recovers Partially, Down Over 500 Points


Suspended IPS officer wants transfer of case in multi-crore heist in Gurgaon


List of websites and apps to download marksheets for CBSE Class 10, 12 Term 1 results 2022


Delhi is experiencing a 'cold day,' with a yellow alert in effect for today and tomorrow.


Coronavirus LIVE Updates: India has 2,55,874 new Covid cases, with a 15.5 percent positivity rate.

Navjot Sidhu called the ED raids a "political vendetta," but adds that no one is off the hook.

Noida: By May, the supertech twin skyscrapers may be demolished.
With nearly 50k new COVID-19 cases in a single day, Karnataka has the greatest single-day increase.

Today's Petrol and Diesel Prices: Check Rates in Delhi, Mumbai, and Other Cities

China 'finds' an Arunachal youngster, and the process of identifying him is underway.

Manohar Parrikar's son, who left the BJP, is being urged to "reconsider his decision" in Goa.

Criminals, not people, are leaving Uttar Pradesh now, says chief minister Yogi Adityanath

'Long overdue historical correction,' Jaishankar says of Subhas Chandra Bose's statue at India Gate.


Cryptocurrency News: 6 Coins Gain Up To 674 Percent In One Day; Price List Here

Thich Nhat Hanh, a well-known Zen Buddhist monk, has died at the age of 95.

The first formal conversations between Biden and Kishida focus on China and nuclear weapons.

Trump Ordered US Defense Staff to Seize Voting Machines After Loss: Report

Delhi Experiences Light Rain, but Air Quality Remains 'Very Poor'

Amit Shah will campaign door-to-door in Kairana today for the UP elections.

'We are ecstatic,' Priyanka Chopra said of her and Nick Jonas' surrogate baby.

'For authentic shradhanjali to martyrs, Amar Jawan Jyoti merged.'

Today, PM Modi will meet with DMs from several districts.

The Delhi government wants to relax Covid restrictions, but the L-G claims it isn't ready yet.


After a major fire at a Mumbai high-rise, two people were killed and several others were injured.

RIL, Asian Paints, HUL, BoM, PNB Housing, VodaIdea, and Biocon are stocks to keep an eye on.

Should you buy, sell, or hold HUL after the third-quarter results?

Aparna Yadav visits Mulayam after joining the BJP and receives his blessings.


Girls sparked the Karnataka hijab row, which has been going on for three decades.

"Cracking on Clubhouse Chats," says Sena MP after three people are arrested for hate speech.

Bulli Bai case: Mumbai cops detain an Odisha MBA graduate.

Rahul Gandhi on the Amar Jawan Jyoti row: 'Some people don't comprehend patriotism.'


In just 24 hours, India has reported 3,47,254 new Covid cases, with 703 deaths.

Delhi suggests that the weekend curfew be lifted and that market restrictions be lifted.

Why Did Akhilesh Yadav Pick Karhal, a "Safe" Socialist Stronghold, Instead of a Family Seat?

The flame of the Amar Jawan Jyoti will be fused with that of the National War Memorial. As a result,

Cash worth Rs 4.72 lakh was discovered stashed in washing powder cartons in Noida.


In a Delhi home, a woman and her four children were discovered dead, with the stove on for heat.

The Karnataka government is expected to relax the weekend curfew.

China's PLA kidnaps a 17-year-old Indian boy in Arunachal Pradesh.

Akhilesh Yadav said Mulayam tried to persuade Aparna Yadav not to join the BJP.

According to reports, the poster girl for Priyanka Gandhi's flagship campaign may join the BJP.
SC: Don't wait for claims to contact kin for Covid ex gratia.


Everything You Need To Know About AGS Transact Technologies' Initial Public Offering

In the midst of the 5G rollout, Air India has cancelled some US flights.

Wednesday's day trading guide: 6 stocks to buy or sell today - 19th January

Raima Islam Shimu, a Bangladeshi actress, has confessed to her murder.



Bhagwant Mann is the AAP's Punjab CM-in-waiting, according to a phone poll.




Five Things to Expect From Delhi's New Electric Buses

Rain, dense fog, and hail are expected in these states and UTs, according to the IMD.
Omicron Tested Positive? Isolation at Home: Follow These Steps

The Uttarakhand Congress is split over Harak Singh Rawat's return.

After Tamil Nadu's tableau was rejected, CM M K Stalin wrote to Prime Minister Narendra Modi.

Mumbai Cops To Court In 'Bulli Bai' App Case Accused Of Being Involved In Sulli Deals







Pune: A conman defrauds a woman of Rs 62 lakh by creating a false profile on a matchmaking website.

70% of adults are twin-jabbed, and younger children may be included.


Pandit Birju Maharaj, the legendary Kathak maestro, has died at the age of 83.

'A 24-bomb Pak shipment was transported at Ghazipur.'

Election Commission Meeting Today On Punjab's Request To Defer Polls


Police said a religious leader was arrested for misogyny, not for a hate speech in Haridwar.

Arvind Kejriwal's Swipe In Goa 'Proved Delhi Government Most Honest': Arvind

The CBI has detained Gail's director in a bribery case.

The Indian Army's New Combat Uniform Has Been Revealed: Here Are The Top 5 Things To Know

In an early morning fire, the Secunderabad Club was completely destroyed.

Covid-19: India's one-year immunisation campaign comes to a close.

India has 2.71 lakh Covid cases, with 314 deaths in the first 24 hours; Omicron has 7,743 cases.


Bhim Army Chief Says No To UP Alliance: "Akhilesh Doesn't Want Dalit Support"






Stand behind the party, urges former BJP minister Maurya's daughter, who is also an MP




From October 2022, the government proposes mandating six airbags in all new cars.

Muslims are 'most secure and happiest' under BJP administration, according to the RSS.

UP Polls: Will the BJP's Leader Exodus Hurt It or Can It Still Rely on Caste Arithmetic?



TCS buyback offer will include Tata Sons and Tata Investment Corporation.

Kamal Khan, a 61-year-old veteran journalist, has died, and tributes have poured in.

"You'll all die," says a man accused of making a hate speech in Haridwar after his first arrest.

At Delhi's Flower Market, a bomb was discovered in an abandoned bag and detonated.

The budget session will begin on January 31 and will be divided into two parts: Report


In a case involving a nun rape, Bishop Franco was found not guilty.



Ananya Panday's lovely present from Gauri Khan has a Hollywood connection!


After a huge drop on Tuesday, Vodafone Idea stock is surging today. What do the brokers have to say?

Punjab's AAP chief ministerial candidate will be announced next week: Kejriwal


In April-May, a US F-18 fighter op-demonstration was held in Goa for the INS Vikrant.



After Navjot Sidhu's warning, Chief Minister Channi makes a suggestion to the Congress.

Due to the Covid spike, private offices in Delhi have been required to convert to WFH.

Day 25 of the Pushpa box office collection: Allu Arjun's film is showing no signs of slowing down.

Harbhajan Singh Makes a Big Statement About Virat Kohli In South Africa vs. India


Man poses as IPS officer, cheats Gurugram firm of Rs 37 crore

"Why Are You Laughing?" explains Yogi Adityanath's "80 vs 20."

On February 1-2, three Rafale fighter jets with Indian modifications would arrive.

Who are Trads, the criminal group to whom Bulli Bai's founder belongs?

Restaurants say it's the final blow after Delhi demands a shutdown.
India records 1.68 million new covid cases, down 6.4 percent from the previous day.

In just 24 hours, India has added 1.79 lakh Covid-19 cases, resulting in 146 deaths.

Goa Minister Resigns Ahead of Elections: "BJP Is No Longer The People's Party"

Hate Speeches in Haridwar: The Supreme Court Says It Will Hear The Case

PM Modi sends 100 pairs of jute slippers to workers at Kashi Vishwanath Dham.


Maharashtra government allows salons and gyms to open at 50% capacity the next day."



As actor Sonu Sood enters politics, his position as Punjab's state icon is withdrawn.

In a road accident, 4 people were killed and 6 others were injured. Bangalore


HC gives its approval, the Gangasagar Mela begins today, bringing thousands of people to Bengal.


Overnight in Delhi and nearby areas, heavy rain and thunderstorms


Caught on Camera: A Bihar woman healthcare worker is brutally attacked for missing a day of work.


The Rafale fighter will be demonstrated for the Navy in Goa, followed by the F/A-18 in March.


The ICMR chief is concerned about the safety of molnupiravir.
A Bihar man receives the Covid-19 vaccine 11 times and describes it as a "wonderful thing."



In a dawn fire in Delhi's Chandni Chowk Market, 60 shops were burned.

Pilots' 'loss of situational awareness,' according to the CDS crash investigation

Punjab Probe Panel on PM Security Breach; Supreme Court Hearing Tomorrow

Bulli Bai: According to Mumbai cops, an Uttarakhand girl was in contact with Vishal Kumar Jha.

Today, the Air Force will brief Defence Minister Gen. Rawat on the Chopper Crash.

Saffron scarves are worn by Karnataka college students in protest of hijab in the classroom.

All international passengers must have RT-PCR Covid tests in Mumbai.


Uttar Pradesh's night curfew has been extended by two hours.

3 Jaish terrorists, including a Pakistani national, were killed in the Pulwama encounter

Weekend Curfew in Delhi; Full Capacity for Buses and Metro

Police in Noida said a 3-year-old girl was slain and raped by her grandmother's lover.

The Delhi government intends to construct twin skyscrapers at ITO.

As cases spiral, 23 junior doctors from a Delhi hospital test Covid+ in a week.

Katrina Kaif's "Vibe" With Her Brother-In-Law The Instagram Exchange of Sunny Kaushal

Are foreign institutional investors (FIIs) re-entering the Indian market? Deepak Shenoy responds.


As Punjab colleges become Covid hotspots, 100 students test positive.


Cong demands the resignation of Ajay Mishra's son, who is named in the Lakhimpur Kheri chargesheet.

'Bulli Bai' case: Mumbai cops detain a 21-year-old Bengaluru student.

In my dreams, Lord Krishna tells me that I will establish Ram Rajya: Akhilesh Yadav

Arvind Kejriwal Tested Positive For Covid, Claims "Mild Symptoms"


Khatija Rahman, AR Rahman's eldest daughter, married in a private ceremony and shares a photo.


Priyanka Chopra and Nick Jonas 'celebrated life' aboard a yacht as they rang in the new year.
India Inc. predicts a surge in investment and is preparing capital expenditure plans.


Meghalaya Governor Meets With Prime Minister Modi Over Farm Laws, Calls Him "Egotistical"

Bengal shuts schools and institutions, but allows two-day flights from Mumbai and Delhi.

PM Modi, speaking at a sports varsity, claims that goons had free reign in previous UP governments.

Mrunal Thakur reports she has mild symptoms after testing positive for Covid-19.

Mamaearth is the first unicorn of 2022, having raised $52 million with a $1.2 billion valuation.


Manjinder Sirsa has resigned as president of the DGSMC.


How is Omicron growing so quickly in Delhi and Mumbai? Here's what the data tells us.

'Hate speech against Muslims is on the rise in India,' according to a Supreme Court petition.
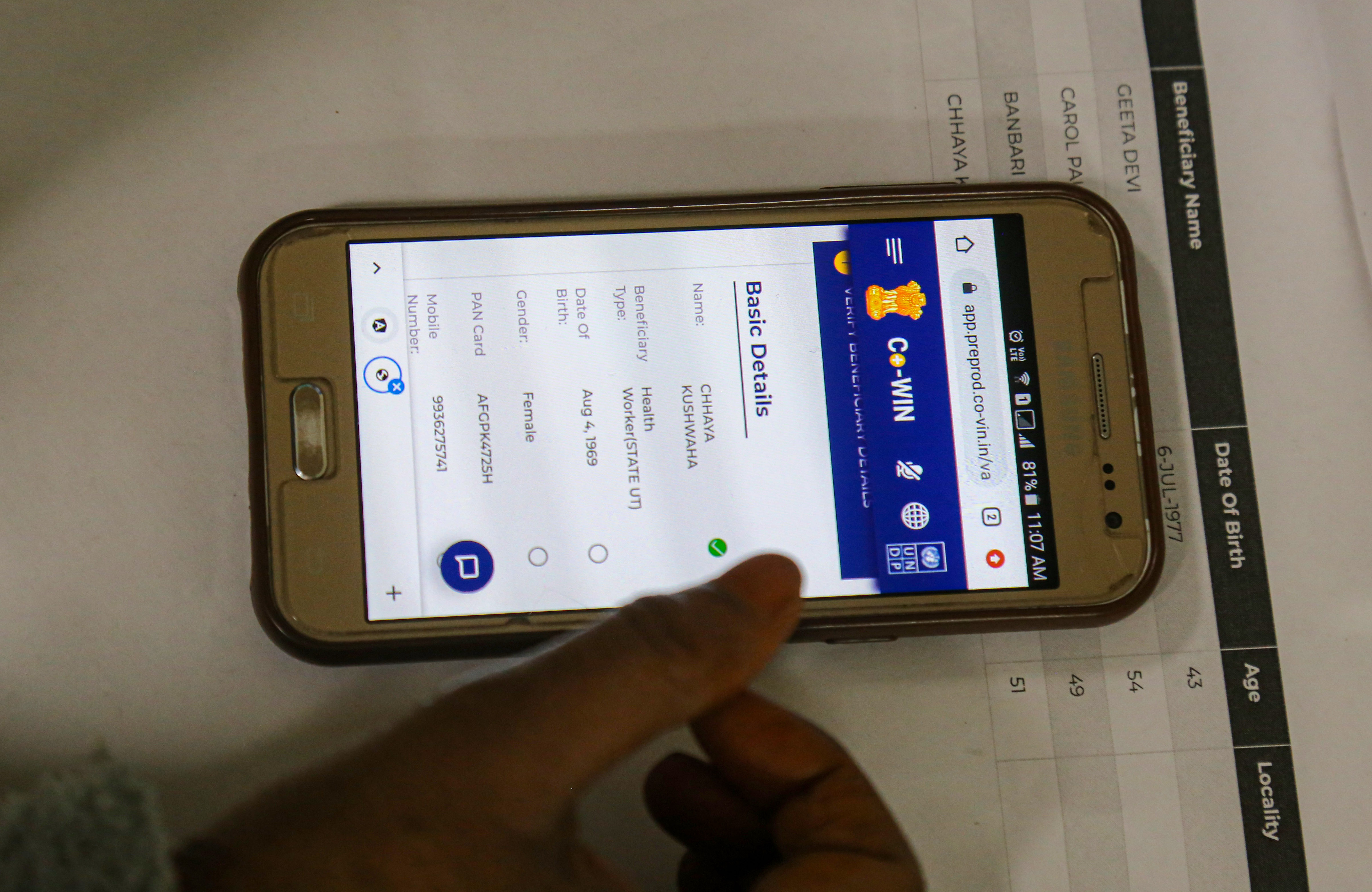
CoWin registration for the 15-18 age category is set to begin today.

Covid cases in Maharashtra and Mumbai have doubled in the last 48 hours; the third wave has arrived.

Several people are afraid they would be trapped when a landslide burys mining vehicles in Haryana.




In the high-octane action thriller Valimai, Ajith is a man on a mission.

Vijay Deverakonda evolves into an MMA champion in Mumbai's Slumdog Millionaire.


Faradion Limited is now 100% owned by Reliance New Energy Solar.

AFSPA has been prolonged in Nagaland notwithstanding the cancellation of the call.

NEET-PG aspirants are stuck in time, facing an uncertain future.
1,270 Omicron Cases, with the majority (450) in Maharashtra; 16,764 Fresh Covid Cases

In a shootout in Srinagar, three terrorists were killed, one of whom was a JeM operative.

AAP has filed a complaint against BJP leaders for their treatment in a "dangerous" hospital.

Due to worries about Covid-19, the India Art Fair has been postponed.

In Delhi, yellow level curbs would be maintained: LG Baijal, Anil


After a car chase, Yash Dasgupta and Nusrat Jahan managed to hide from the reporters.

Stocks in Asia are down, but oil companies are having a great year.




After commenting against Gandhi and Islam, a Hindu religious leader was arrested.

Journalists must avoid being "coopted by ideology or the state," according to CJI Ramana.



Today's horoscope: December 29, 2021.

Mohammed Shami on the "Emotional Celebration" for his 200th Test Wicket: "It Was For My Father"


Day 12 (Early Trends): Pushpa Box Office Remains Stable, On Track To Reach 200 Crores

For the first time as RIL chairman, Mukesh Ambani discusses succession.

Tax Department provides relief ahead of the ITR application deadline



Maharashtra Board Students Say Cancel Exams After Varsha Gaikwad Tests Positive

As Aaditya Thackeray enters the Maha Assembly, BJP MLA Nitesh Rane exclaims, "Meow, Meow, Meow!"



Dengue fever has claimed the lives of 23 people in Delhi this year, 15 of them were youngsters.




Next year, 5G will be available in metros and larger cities.


The arrest of SFJ Multani in Germany is a result of the Modi government's diplomatic pressure.






Analysts say the Nifty must stay over 17,000 to continue to rise.

RBL Bank's stock is down today. What are the opinions of the brokerages on the stock?


"Misoperation". On the Boiler Explosion That Killed 7 People in Bihar

Maharashtra School Seals 52 Students Infected With Covid

The suspect in the Ludhiana blast was in contact with a drug lord, according to phone records.


Tamil Nadu reports 33 new Omicron cases, bringing the total number of cases to 34.

Priyanka Gandhi's Children's Instagram Not Hacked: Sources After Probe

As the risk of Omicron looms, Delhi Govt has banned Christmas and New Year celebrations.

CRPF Women Commandos To Provide Security To Amit Shah, Sonia Gandhi, Others


Sourav Ganguly On India Player's Surprise Inclusion In T20 World Cup Squad

In ‘message’ to Didi, Sonia Gandhi’s competition meet shuns TMC
Dalit, Adivasi Christians the Most Attacked in Karnataka

Last batch of farmers to depart UP Gate protest web site at Ghazipur today

Opinions divided over Kashi Vishwanath corridor

Congress steps up attack on govt over SIT report on Lakhimpur Kheri violence

After 21 Years India's Harnaaz Sandhu Brings Home Miss Universe Crown
Tega Industries stocks see stellar debut with 68% list premium. What should investor do ?

Protest ends, clean-up begins at farmers protest sites

Women's Asian Champions Trophy: Indian hockey player tests +ve, match against Korea cancelled

Delhi Records Season's Lowest Temperature At 6.4 Degrees Celsius

India desires rule of Hindus said Rahul Gandhi

20-yr-old international traveler becomes Chandigarh's first Omicron case

Sonia Gandhi slams ‘blatantly misogynist’ question in CBSE Class 10 exam, demands apology from govt

India sends life-giving drugs to Afghanistan in resource

Gen Bipin Rawat’s daughters collect ashes, to immerse them in Haridwar

'I Go to Rajya Sabha When I Feel Like,' Says Ex CJI Ranjan Gogoi

PM Modi to release Saryu canal project these days. 10 Points

farmers end protest after government accepts demands, Cops To Remove Barricades From Protest Sites

Naidu urges Govt, Opposition to stop impasse in Rajya Sabha, conduct business

Four Arrivals From "At-Risk" Nations Test Covid Positive At Delhi Airport: Report

‘Incredible mind-set’, says Congress after Mamata’s ‘what UPA?’


CBSE term-1 board tests 2022: Here’s Class 10 social Science know-how paper analysis

Mamata Banerjee Meets Shiv Sena's Aaditya Thackeray, Sanjay Raut In Mumbai

2 union leaders say time to speak about dharna quit

Assam lynching: Main accused Neeraj Das dies in accident

Due to pollutants: NCR states on 48-hour be aware
Can RT-PCR check detect upon infection with Omicron version of Covid-19?

Here are the allegation towards 12 Rajya Sabha suspended MPs

Without a debate Parliament clears Farm Laws Repeal Bill


Tirumala temple OSD Dollar Seshadri passed away of cardiac arrest in Visakhapatnam

Amid farm protests, Govt moved from ‘no’ to ‘written guarantee’: Explained

PM Modi To Lay Foundation Stone Of Noida International Airport Today

What is INS Vela, the submarine that Indian Navy is set to commission? Explained

India’s fertility price below replacement stage

Anti-Corruption Bureau raid on Karnataka PWD junior engineer Money spills out from pipe

UPA should have hit again after 26/eleven, says Tewari

What AAP’s ‘Rs 1,000 to all women’ will value Punjab. Explained

Union IT Minister: Personal Data Protection invoice will in addition beef up privacy


The Hinduja Family Feud Puts Century-Old Business Empire In Jeopardy

PM Modi to Unveil Asia's Largest Airport at Jewar Tomorrow

Farm legal guidelines repeal: Cabinet approval probably these days, bill listed for winter session

Courtesy Supreme Court’s push, this Dalit scholar movements closer to his IIT dream
‘No biological proof…’: ICMR chief on need for booster pictures of Covid-19 jabs

Param Bir Singh case: A look lower back at different pinnacle law enforcement officials at the run

Group Captain Abhinandan Varthaman conferred Vir Chakra by President Ram Nath Kovind

TMC MPs meet Amit Shah on Tripura, Mamata to call on PM

5 International Airports For UP Ahead Of Polls: 10 Points

Farm laws repeal: Govt starts efforts for House consensus

Delhi pollutants: Construction can resume, colleges stay close; truck entry ban prolonged

Capt Amarinder to Fight Punjab Polls from Family Bastion Patiala, Says ‘Won't Run Away’


'I may not prevent, right here to do my job': Vir Das on 'Two Indias' controversy

Withdraw CAA Like farm law : AIMIM leader Asaduddin Owaisi tells PM Modi

In Delhi Today Mamata Banerjee Meeting Sonia Gandhi

Sack MoS, make MSP legitimate right: farmers positioned six needs to PM Modi

Grenade assault near Army camp in Pathankot, probe underway


Shamita Shetty returns toBigg Boss 15: grills Nishant Bhat of stepping on relationships

Here's What Amitabh Bachchan Wrote After Watching Son Abhishek's Bob Biswas Trailer


Reliance Industries, Saudi Aramco to re-examine proposed investment deal in O2C enterprise

India reviews 10,302 Covid cases, 267 deaths; Kerala debts for 204 fatalities

Today, In Lucknow PM Modi, Amit Shah, Ajit Doval going To Meet Top Cops From All States

At least 17 Dead, 100 Missing After Heavy Rain In Tripurti, Andhra Pradesh

Farm laws scrapped: What's in the back of the timing?

India vs New Zealand, 2d T20I: India Look To Keep Winning Combination In Ranchi

Stock market holiday: BSE, NSE to remain closed today on account of Guru Nanak Jayanti

Paytm CEO's Fixation On provide A Record IPO Led To A First-Day Flop

Odd-Even Scheme In Haryana Likely From Next Week, Say Sources

Javed Akhtar responds to Kangana Ranaut's 'bheekh' remark, here's what he tweeted

Under fireplace over Srinagar killings, J&K admin orders probe, exhumes 2 our bodies

Manipur ambush no longer privy to presence of woman, child: Revolutionary People’s Front

KCR protests against Centre as paddy difficulty receives ‘extreme’, turns up the heat on BJP

Life saved as employees, lawyers stepped in, Bihar judge to HC on police attack


PM Says three Farm Laws To Be neutralize


Delhi air pollution: Schools, schools to stay closed until similarly orders

India refutes allegations on ‘weakening’ Glasgow weather p.C.


From ''Ten Thousand'' income to billionaire, Paytm's Vijay Shekhar Sharma says his dream is…

Twitter money owed of Govt critics, activists centered by Tripura police

Nations Must Ensure Crypto Not In Wrong Hands, "Can Spoil Our Youth": PM

"Debates On TV Causing More Pollution": Chief Justice Amid Blame Game


Coronavirus India Live Updates: India Records 10,197 New COVID-19 Cases, 15% inflate Than Yesterday

Gujarat CM says ‘no problem’ with non-veg meals whilst road carriers cry out for help

Petrol, Diesel Price Drop: Fuel Rates Fall Sharply After Tax Cut; Check Price Here

Delhi air pollutants: NCR School, Collages Shut production at a halt until November 21

850 new swanky liquor shops open in Delhi these days. Know fees, timings and facilities

Purvanchal Expressway: Modi Hails Yogi Govt, Slams Earlier Regimes for 'Punishing People

Kartarpur Sahib Corridor returns for explorers today: All you really want to know

Kangana Ranaut Now Targets Mahatma Gandhi said "One Can Only Get Bheekh..."

IB, home, RAW, defence officials residency can be reached out for 2 yrs Modi govt issues warning

Delhi govt to present proposition on lockdown to SC today to handle air contamination

Coronavirus: Delhi records 36 new cases, 1 passing
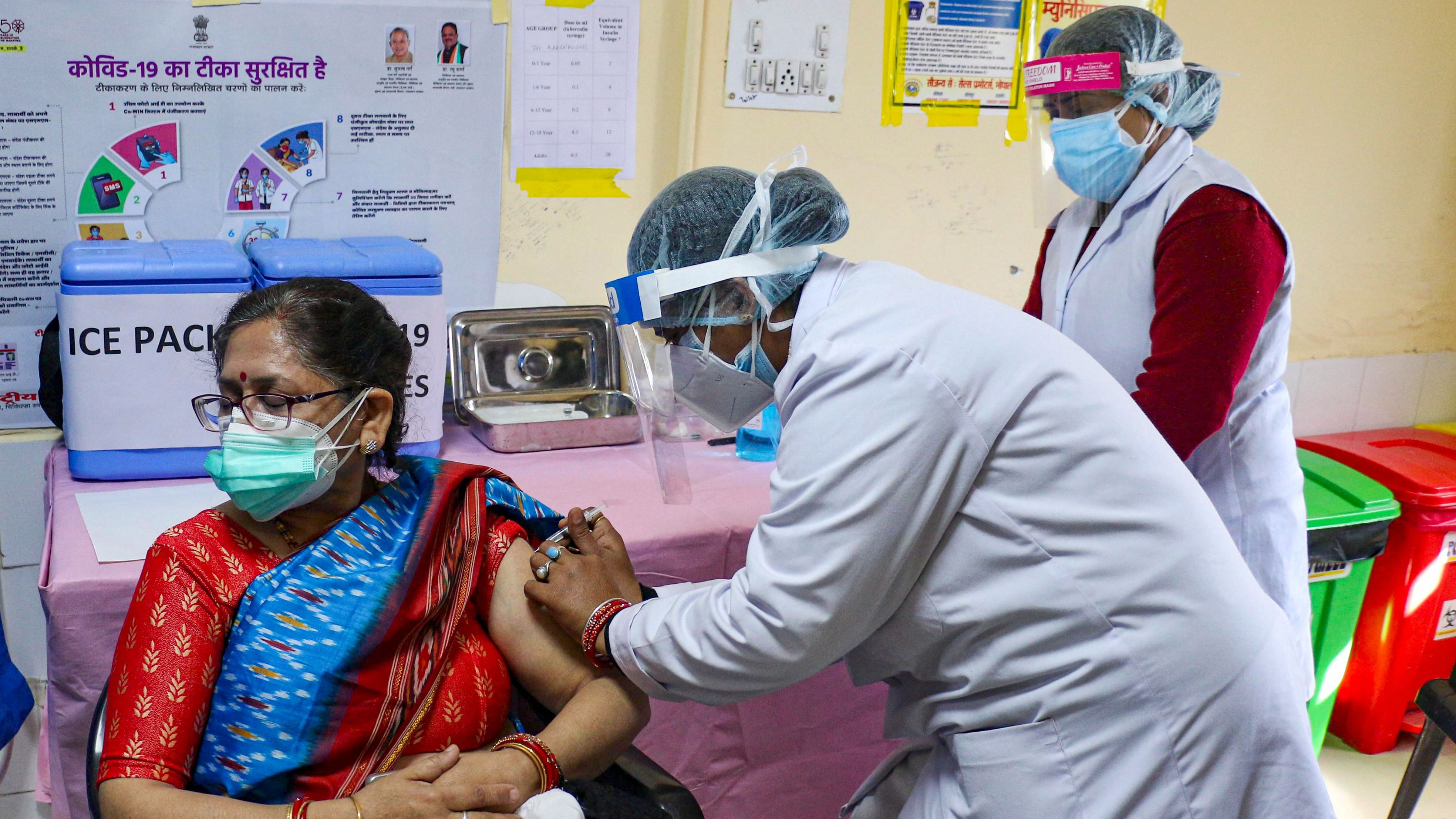
Coronavirus Vaccine Tracker: Over 30.2 lakh jabs on November 14

Two journalists getting back from Tripura confined by Assam police

Curfew in Maharashtra's Amravati extended to four towns as BJP stages protest

Gujarat ATS holds onto 120 kg of heroin worth crores in Morbi, 3 captured

Padma Vibhushan Babasaheb Purandare pass away at 99; PM Modi says, 'tormented indeed's

YouTuber, Who Made His Dog "Fly", Arrested For Filming Sacred UP Site

Amit Shah-led meet of southern states heads settle 40 of 51 forthcoming issues

Disgraceful to stigmatize the whole Hindu religion: RK Singh on Salman Khurshid's new book


Woman assaulted by chaser loses vision in both her eyes

Purvanchal Expressway: PM Modi to land in IAF's Super Hercules for inaugural

Hinduism isnt about disdain, Hindutva is, says Rahul Gandhi

Interjection in Sameer Wankhede's authentication: Bombay HC tests Nawab Malik

TN governor toward PM Modi, Amit Shah, meets with CM

Chennai Rains : How control rooms go to rain-related grievances, alleviate public fear

Narendra Modi to launch RBI Retail Direct Scheme today: All you want to know about new Scheme

Petrol, Diesel Price fall: Fuel Rates Cheaper With Excise Duty Cut. See Fuel Prices

Delhi: Covid cases cross 50 imprint following 2 months


Amit Shah in Varanasi on Nov 12, may debate on UP polls strategy with Bhartiya Janta Party workers

Nykaa makes a heavenly presentation, stock records at Rs 2,018 with 79% premium

BJP MP says 'panga' with PM Modi implies political demise of KCR

India hosts NSA-level summit on Afghanistan; 7 nations in attendance

High Alert For Heavy Rainfall For Today, Tomorrow In Flood-Hit Tamil Nadu

Delhi Govt Deploys Boats to take off Froth from Yamuna with Ropes

HC directs Delhi government to settle on request over 'missing' trees in six weeks

Delhi news live: Air quality remaining parts 'extremely poor'; 33 new Covid-19 cases reported

Delhi news features: BJP can celebrate Chhath Puja at Yamuna bank if L-G, cops permit it, says AAP

Emergency landing of Air India flight in Assam because of obstacle in landing gear


Madhya Pradesh: 4 kids dead in fire at childrens' ward of hospital in Bhopal


We are made to weep for water and die on in water: Madras HC to Chennai Corporation


BJP Attacks Congress Over New Rafale Report

Tamil Nadu Braces For Very Heavy Rainfall

"Just Mamata Banerjee Got It Spot On": Derek O'Brien On Demonetization


4 CRPF soldiers killed, 3 injured in Sukma after a jawan starts shooting

Fisherman killed in Pakistan open firing off Gujarat

BJP not family-run party, says PM Modi; spotlights 'phenomenal' gains in bypoll results

Five years after demonetization, cash being used now at record 14.5% of GDP


Anil Deshmukh's ED custody closes today, will be delivered under the steady gaze of PMLA court

Ex-SBI supervisor's arrest part of bid to stall resolution, show documents

Huge search launched in J&K's Khablan forest after reports of militants in area

After Navjot Sidhu's Ultimatum to congress, Top Punjab Government Lawyer's Attack at sidhu

Black Diwali' for teachers of Delhi govt-funded DU colleges

Human brains cells not impacted by COVID-19 infection: Study
Low platelet count? Foods to expand your platelet count after dengue

"My Inner Voice Said Kedarnath Will Rise Again": PM Modi On 2013 Devastation

Muhurat Trading: Samvat 2078 starts on a happy note
India logs 12,729 new Covid-19 cases, active caseload ascends to 148,922

Bangalore News Live: Houses, streets flooded after heavy rains in

After Center, BJP-ruled Haryana, Himachal cut fuel Prices further

'Priyanka Gandhi's women-driven guarantees just implied for UP polls'

Delhi's air quality drops to 'extreme' as Capital imprints Diwali with firecrackers

Delhi auto driver shot dead, police researching

300 party leaders to show up before PM Modi at BJP's mega meeting in Delhi

Why ex-SBI chief Pratip Chaudhuri's arrest has baffled banking industry
Delhi records 1,200 Dengue cases in October, most noteworthy in 4 years

Anti-CAA protest secular, charge sheet communal': Umar Khalid in Delhi riots connivance case

PM Modi gets back to Delhi after G20 summit, COP26 climate conference

24-year-old arrested in the wake of leasing 300 bank accounts to the phishing capital of India


IT division appends properties worth Rs 1,000 crore supposedly having a place with Ajit Pawar

Infighting in Punjab Congress proceeds, Sidhu slams Channi for promising 'candies'

Chennai nerd hits pothole on Anna Salai, slips, goes under wheel of MTC transport

Bihar bye-surveys: Initial round of counting shows mixed outcomes for RJD, JD

BJP countinues on lead in MP; TMC starts celebrations in Bengal

Boris Johnson acknowledges Modi's invite to India, to plan visit soon


INS Visakhapatnam to be commissioned on Nov 18, followed by INS Vela sub

China re-forces new lockdowns in the midst of rising Covid-19 cases

Gujarat gets its first plane café. Peruse subtleties here

Tips for rearranging your kitchen and restroom this bubbly season

Enjoy some time off From the City Life and Visit These 5 Beautiful Delhi-NCR Forests

CVC's Ahmedabad offered forecasts well for BCCI and the IPL

Mumbai zonal head Sameer Wankhede arrives at NCB office in Delhi

Zee Entertainment Enterprises drops October 27 executive gathering

Fair review must for versatile economy: RBI

During Delhi's Master Plan 2041 formal proceeding, DDA's 'flurry' draws fire

'Have donated for Ram Mandir,' says Kejriwal subsequent to offering supplications at Ram Janambhoomi

Delhi BJP pioneer Manoj Tiwari to hold exceptional immunization drive for Chhath Puja

India should focus more on its most troublesome neighbor

Aryan Khan's Lawyer Files Counter Affidavit After NCB Opposes Bail Plea

Obstinate stubble: Delhi's anguish set to return

Nehru Place resembles a ghetto, says Delhi High Court hearing supplication on road merchants

Authorize standards to try not to jam in business sectors: Delhi HC

Delhi: Fire kills four in Seemapuri region

1 Billion Vaccinations and what it Mean for Metro Ridership?
Eye donation: Important things you should remember

Delhi now near winning fight against dengue, says Kejriwal

Gautam Gambhir's fourth 'Jan Rasoi' to open in Laxmi Nagar on Saturday

17 killed in fire at Russian modern explosives production line

Fortify your immune system with these day by day propensities

Secy Youth Affairs, Sports partakes in Clean India Program in Delhi

Clarified: Here's Why Modi Govt's High Taxes on Fuel Don't Just Affect 5% of India

Canada eliminates warning on trivial travel; Here's how it affects India

OTTs to make their introduction on International Film Festival of India

DDMA to settle on Chhath festivities one week from now in Delhi as legislative issues increases

'Ghee' is code word for explosives, claims NIA; Delhi court says no proof
Coronavirus immunization portions in Delhi inside contacting distance of 2 crore

Priyanka Gandhi meets group of Agra man who passed on in police guardianship

Coherence of twofold motor govt required for Goa's turn of events, says PM Modi

Focus in Delhi HC protects IT rule requiring WhatsApp to follow originator of message

India, UK back worldwide way to deal with drawing in Taliban

Brush Vroom: From Priyanka to Kejriwal to Modi, Humble Jhadu is Sweepstake in Indian Politics

Arvind Kejriwal to begin his two-day Punjab visit today

'Go to Delhi if Not Satisfied': BJP's Gujarat Chief Says Party to Field 100 New Faces in 2022 Polls

The travel industry support: Vistadome mentors in Shatabdi trains on 3 courses

Delhi BJP says will observe Chhath, yet with Covid conventions

High court excuses request testing Rakesh Asthana's arrangement as Delhi Police magistrate

'Blackout may occur in Delhi': Satyendar Jain focuses fingers at Center

Sonam Kapoor acclaims Kim Kardashian's 'SNL' facilitating debut

Delhi Police Registers Case After Actress Accuses YouTuber Of Alleged Harrasment

Delhi-based umpire Sumit Bansal dies at 46


Durga Puja 2021: All you need to think about importance, date, history and time

Biodiversity COP from Monday, equivocalness about India's objectives accomplished

Pulverizing individuals under SUVs isn't BJP's legislative issues: UP unit boss

Uttarakhand transport serve Yashpal Arya stops BJP, joins Congress alongside MLA child Sanjeev


South Delhi Municipal Corporation clinics get PSA oxygen plants

Delhi: Fire at Paper godown in Harsh Vihar; no setbacks

Delhi govt misdirecting individuals over power supply: Adesh Gupta

Man shoots spouse, mother-in-law in west Delhi, captured


Delhi air terminal sees record number of travelers after second Covid-19 wave
Coronavirus visa, antibody segregation new exchange boundaries: India to WTO

Charu Parashar commends birthday with her young lady posse

Akshay Kumar gets nostalgic as he goes for Raksha Bandhan in Delhi: 'Exquisite to hear the prattle'

NCB captures Nigerian public with ecstacy pills in Mumbai sting operation case; produces in court

One more notification glued external Ashish Mishra's home over Lakhimpur Kheri viciousness
Unvaccinated govt staff will not be permitted to go to office from Oct 16: DDMA

Clear skies, moderate air quality for Delhi on Friday

Akbar Road billboard vandalized in Delhi, Hindu Sena claims liability

Delhi air terminal to continue flight activities at T1 terminal from Oct 31

New government school to give equivalent heave to classes, sports

Delhi reports 44 Covid-19 cases, zero passing; positivity rate 0.07%

Engineer killed in hit-and-run at Rajiv Chowk underpass

Previous India pacer Abhimanyu Mithun resigns

Anshu Malik leaves a mark on the world, arrives at World Championships last

India's GDP development in 2021-22 seen at 8.3%, says World Bank report

India to re-open for foreign tourists from 15 Oct, yet with riders. Subtleties here

Fasting this Navratri? Remember these fundamental tips

Compelling Ayurvedic tips for normal defecation, obstruction help

Delhi: Arvind Kejriwal declares activity intend to battle air contamination in winters

Blended sentiments over ranchers impeding Ghazipur line

Delhi: 16 players captured from farmhouse gambling club


Delhi CM Kejriwal launches one-stop app for tourists

NSCN(IM) Invited to Delhi to Continue Talks on Naga Issue, Says Nagaland CM

Bullets rain again, gangster's aide shot in southwest Delhi

Amarinder in Delhi today on his first visit subsequent to stopping as Punjab CM


5 fundamental indicative tests for ladies more than 30
Lung wellbeing: Nutritionist shares cautioning signs you ought not overlook

Delhi: Salon advised to pay Rs 2 crore for 'bungling' model's hair style

IPL 2021: All eyes on Rishabh Pant's advancement as pioneer as Delhi Capitals restart crusade

"Not Surprised": Kevin Pietersen's Brutal Assessment Of David Warner's Duck Against Delhi Capitals

Delhi: DDA to assemble international sports complex in Dwarka

Toward the west of India, an extraordinary realm of opium is rising. New Delhi needs to act, quick

Vistara To Operate Flights Between Delhi, Paris From November 7


Hate speech case: Delhi HC awards bail to coordinator of Jantar Mantar occasion

Sushmita Sen-starrer 'Aarya' named at International Emmy Awards

Sonu Sood, being examined for tax avoidance, says 'a ton of it is my compensation'

Delhi financial specialist looks for FIR against Raj Kundra, Shilpa Shetty over extortion

Punjab CM Leaves For Delhi, May Discuss Cabinet Expansion With Cong Leadership


PM holds bilateral meetings with his Japanese and Australian partners

TMC's Arpita Ghosh resigns as Rajya Sabha MP

Criminal Jitender Gogi shot dead inside Delhi's Rohini court

Delhi gazes at alcohol lack as private distributes shut shop

Ex-Delhi police chief, who dealt with Batla House case, dies at 70

Delhi to plan bio-decomposer arrangement from today to battle stubble consuming
